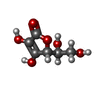
Entry Database : PDB / ID : 2ydgTitle Ascorbate co-crystallized HEWL. Lysozyme C Keywords / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Gallus gallus (chicken)Method / / / Resolution : 2 Å Authors De la Mora, E. / Carmichael, I. / Garman, E.F. Funding support Organization Grant number Country Consejo Nacional de Ciencia y Tecnologia (CONACYT)
Journal : J.Synchrotron.Radiat. / Year : 2011Title : Effective Scavenging at Cryotemperatures: Further Increasing the Dose Tolerance of Protein Crystals.Authors : De La Mora, E. / Carmichael, I. / Garman, E.F. History Deposition Mar 19, 2011 Deposition site / Processing site Revision 1.0 Jul 20, 2011 Provider / Type Revision 2.0 Jul 29, 2020 Group Atomic model / Data collection ... Atomic model / Data collection / Derived calculations / Other / Structure summary Category atom_site / chem_comp ... atom_site / chem_comp / entity / pdbx_chem_comp_identifier / pdbx_database_status / pdbx_entity_nonpoly / pdbx_struct_conn_angle / pdbx_struct_special_symmetry / struct_conn / struct_site / struct_site_gen Item _atom_site.auth_atom_id / _atom_site.label_atom_id ... _atom_site.auth_atom_id / _atom_site.label_atom_id / _chem_comp.name / _chem_comp.type / _entity.pdbx_description / _pdbx_database_status.status_code_sf / _pdbx_entity_nonpoly.name / _pdbx_struct_conn_angle.ptnr1_auth_comp_id / _pdbx_struct_conn_angle.ptnr1_auth_seq_id / _pdbx_struct_conn_angle.ptnr1_label_asym_id / _pdbx_struct_conn_angle.ptnr1_label_atom_id / _pdbx_struct_conn_angle.ptnr1_label_comp_id / _pdbx_struct_conn_angle.ptnr1_label_seq_id / _pdbx_struct_conn_angle.ptnr3_auth_comp_id / _pdbx_struct_conn_angle.ptnr3_auth_seq_id / _pdbx_struct_conn_angle.ptnr3_label_asym_id / _pdbx_struct_conn_angle.ptnr3_label_atom_id / _pdbx_struct_conn_angle.ptnr3_label_comp_id / _pdbx_struct_conn_angle.ptnr3_label_seq_id / _pdbx_struct_conn_angle.value / _struct_conn.pdbx_dist_value / _struct_conn.ptnr1_auth_comp_id / _struct_conn.ptnr1_auth_seq_id / _struct_conn.ptnr1_label_asym_id / _struct_conn.ptnr1_label_atom_id / _struct_conn.ptnr1_label_comp_id / _struct_conn.ptnr1_label_seq_id / _struct_conn.ptnr2_auth_comp_id / _struct_conn.ptnr2_auth_seq_id / _struct_conn.ptnr2_label_asym_id / _struct_conn.ptnr2_label_atom_id / _struct_conn.ptnr2_label_comp_id / _struct_conn.ptnr2_label_seq_id Description / Provider / Type Revision 3.0 Oct 4, 2023 Group Advisory / Atomic model ... Advisory / Atomic model / Author supporting evidence / Data collection / Database references / Derived calculations / Experimental preparation / Non-polymer description / Other / Refinement description / Source and taxonomy / Structure summary Category atom_site / atom_sites ... atom_site / atom_sites / chem_comp / chem_comp_atom / chem_comp_bond / database_2 / diffrn / diffrn_source / entity / entity_name_com / entity_src_nat / exptl_crystal_grow / pdbx_audit_support / pdbx_chem_comp_identifier / pdbx_contact_author / pdbx_database_status / pdbx_entity_nonpoly / pdbx_entry_details / pdbx_nonpoly_scheme / pdbx_struct_assembly_prop / pdbx_struct_conn_angle / pdbx_struct_sheet_hbond / pdbx_struct_special_symmetry / pdbx_validate_close_contact / pdbx_validate_rmsd_angle / pdbx_validate_torsion / refine / refine_hist / refine_ls_restr / refine_ls_shell / reflns / reflns_shell / software / struct / struct_conf / struct_conn / struct_ref / struct_sheet / struct_sheet_order / struct_sheet_range Item _atom_sites.fract_transf_matrix[2][1] / _atom_sites.fract_transf_matrix[3][2] ... _atom_sites.fract_transf_matrix[2][1] / _atom_sites.fract_transf_matrix[3][2] / _chem_comp.formula / _chem_comp.formula_weight / _chem_comp.id / _chem_comp.mon_nstd_flag / _chem_comp.name / _chem_comp.pdbx_synonyms / _chem_comp.type / _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _diffrn.pdbx_serial_crystal_experiment / _diffrn_source.pdbx_wavelength / _diffrn_source.pdbx_wavelength_list / _entity.formula_weight / _entity.pdbx_description / _entity.pdbx_number_of_molecules / _entity_name_com.name / _entity_src_nat.common_name / _entity_src_nat.pdbx_beg_seq_num / _entity_src_nat.pdbx_end_seq_num / _entity_src_nat.pdbx_organism_scientific / _exptl_crystal_grow.method / _exptl_crystal_grow.temp / _pdbx_contact_author.email / _pdbx_contact_author.id / _pdbx_contact_author.identifier_ORCID / _pdbx_contact_author.name_first / _pdbx_contact_author.name_salutation / _pdbx_contact_author.organization_type / _pdbx_database_status.SG_entry / _pdbx_entity_nonpoly.comp_id / _pdbx_entity_nonpoly.name / _pdbx_struct_conn_angle.ptnr1_auth_comp_id / _pdbx_struct_conn_angle.ptnr1_label_atom_id / _pdbx_struct_conn_angle.ptnr1_label_comp_id / _pdbx_struct_conn_angle.ptnr3_auth_comp_id / _pdbx_struct_conn_angle.ptnr3_label_atom_id / _pdbx_struct_conn_angle.ptnr3_label_comp_id / _pdbx_struct_conn_angle.value / _pdbx_struct_sheet_hbond.sheet_id / _pdbx_validate_torsion.phi / _pdbx_validate_torsion.psi / _refine.B_iso_mean / _refine.aniso_B[1][1] / _refine.aniso_B[2][2] / _refine.aniso_B[3][3] / _refine.correlation_coeff_Fo_to_Fc / _refine.correlation_coeff_Fo_to_Fc_free / _refine.details / _refine.ls_R_factor_R_free / _refine.ls_R_factor_R_work / _refine.ls_R_factor_obs / _refine.ls_d_res_low / _refine.ls_number_reflns_R_free / _refine.ls_number_reflns_obs / _refine.ls_percent_reflns_R_free / _refine.overall_SU_B / _refine.overall_SU_ML / _refine.pdbx_overall_ESU_R / _refine.pdbx_overall_ESU_R_Free / _refine.pdbx_solvent_ion_probe_radii / _refine.pdbx_solvent_shrinkage_radii / _refine.pdbx_solvent_vdw_probe_radii / _refine_hist.d_res_low / _refine_hist.number_atoms_solvent / _refine_hist.number_atoms_total / _refine_ls_restr.dev_ideal / _refine_ls_restr.dev_ideal_target / _refine_ls_restr.number / _refine_ls_shell.R_factor_R_free / _refine_ls_shell.R_factor_R_work / _refine_ls_shell.number_reflns_R_free / _refine_ls_shell.number_reflns_R_work / _reflns.B_iso_Wilson_estimate / _reflns_shell.d_res_low / _reflns_shell.number_unique_obs / _reflns_shell.percent_possible_all / _software.version / _struct.pdbx_CASP_flag / _struct_conn.pdbx_dist_value / _struct_conn.ptnr2_auth_comp_id / _struct_conn.ptnr2_label_atom_id / _struct_conn.ptnr2_label_comp_id / _struct_ref.pdbx_align_begin / _struct_ref.pdbx_seq_one_letter_code / _struct_sheet.id / _struct_sheet_order.sheet_id / _struct_sheet_range.sheet_id Description / Details / Provider / Type Revision 3.1 Dec 20, 2023 Group / Category Revision 3.2 Nov 6, 2024 Group / Category / pdbx_modification_feature / Item
Show all Show less
 Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information
 X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2 Å
MOLECULAR REPLACEMENT / Resolution: 2 Å  Authors
Authors Mexico, 1items
Mexico, 1items  Citation
Citation Journal: J.Synchrotron.Radiat. / Year: 2011
Journal: J.Synchrotron.Radiat. / Year: 2011 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 2ydg.cif.gz
2ydg.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb2ydg.ent.gz
pdb2ydg.ent.gz PDB format
PDB format 2ydg.json.gz
2ydg.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads 2ydg_validation.pdf.gz
2ydg_validation.pdf.gz wwPDB validaton report
wwPDB validaton report 2ydg_full_validation.pdf.gz
2ydg_full_validation.pdf.gz 2ydg_validation.xml.gz
2ydg_validation.xml.gz 2ydg_validation.cif.gz
2ydg_validation.cif.gz https://data.pdbj.org/pub/pdb/validation_reports/yd/2ydg
https://data.pdbj.org/pub/pdb/validation_reports/yd/2ydg ftp://data.pdbj.org/pub/pdb/validation_reports/yd/2ydg
ftp://data.pdbj.org/pub/pdb/validation_reports/yd/2ydg






 Links
Links Assembly
Assembly
 Components
Components
 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  ESRF
ESRF  / Beamline: ID14-4 / Wavelength: 0.939 Å
/ Beamline: ID14-4 / Wavelength: 0.939 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller



































































































































































































































































 PDBj
PDBj