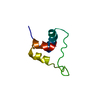
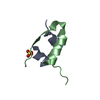
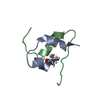
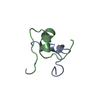
Entry Database : PDB / ID : 1uz9Title Crystallographic and solution studies of N-lithocholyl insulin: a new generation of prolonged-acting insulins. (INSULIN) x 2 Keywords / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species HOMO SAPIENS (human)Method / / / Resolution : 1.6 Å Authors Whittingham, J.L. / Jonassen, I. / Havelund, S. / Roberts, S.M. / Dodson, E.J. / Verma, C.S. / Wilkinson, A.J. / Dodson, G.G. Journal : Biochemistry / Year : 2004Title : Crystallographic and Solution Studies of N-Lithocholyl Insulin: A New Generation of Prolonged-Acting Human InsulinsAuthors : Whittingham, J.L. / Jonassen, I. / Havelund, S. / Roberts, S.M. / Dodson, E.J. / Verma, C.S. / Wilkinson, A.J. / Dodson, G.G. History Deposition Mar 8, 2004 Deposition site / Processing site Revision 1.0 Mar 3, 2005 Provider / Type Revision 1.1 May 7, 2011 Group Revision 1.2 Jul 13, 2011 Group Revision 1.3 Dec 13, 2023 Group Data collection / Database references ... Data collection / Database references / Derived calculations / Refinement description Category chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model / pdbx_struct_conn_angle / struct_conn Item _database_2.pdbx_DOI / _database_2.pdbx_database_accession ... _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _pdbx_struct_conn_angle.ptnr1_auth_comp_id / _pdbx_struct_conn_angle.ptnr1_auth_seq_id / _pdbx_struct_conn_angle.ptnr1_label_asym_id / _pdbx_struct_conn_angle.ptnr1_label_atom_id / _pdbx_struct_conn_angle.ptnr1_label_comp_id / _pdbx_struct_conn_angle.ptnr1_label_seq_id / _pdbx_struct_conn_angle.ptnr1_symmetry / _pdbx_struct_conn_angle.ptnr3_auth_comp_id / _pdbx_struct_conn_angle.ptnr3_auth_seq_id / _pdbx_struct_conn_angle.ptnr3_label_asym_id / _pdbx_struct_conn_angle.ptnr3_label_atom_id / _pdbx_struct_conn_angle.ptnr3_label_comp_id / _pdbx_struct_conn_angle.ptnr3_label_seq_id / _pdbx_struct_conn_angle.ptnr3_symmetry / _pdbx_struct_conn_angle.value / _struct_conn.pdbx_dist_value / _struct_conn.pdbx_leaving_atom_flag / _struct_conn.ptnr1_auth_comp_id / _struct_conn.ptnr1_auth_seq_id / _struct_conn.ptnr1_label_asym_id / _struct_conn.ptnr1_label_atom_id / _struct_conn.ptnr1_label_comp_id / _struct_conn.ptnr1_label_seq_id / _struct_conn.ptnr1_symmetry / _struct_conn.ptnr2_auth_comp_id / _struct_conn.ptnr2_auth_seq_id / _struct_conn.ptnr2_label_asym_id / _struct_conn.ptnr2_label_atom_id / _struct_conn.ptnr2_label_comp_id / _struct_conn.ptnr2_label_seq_id / _struct_conn.ptnr2_symmetry Revision 1.4 Nov 20, 2024 Group / Category / pdbx_modification_feature / Item
Show all Show less
 Yorodumi
Yorodumi Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information HOMO SAPIENS (human)
HOMO SAPIENS (human) X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.6 Å
MOLECULAR REPLACEMENT / Resolution: 1.6 Å  Authors
Authors Citation
Citation Journal: Biochemistry / Year: 2004
Journal: Biochemistry / Year: 2004 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 1uz9.cif.gz
1uz9.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb1uz9.ent.gz
pdb1uz9.ent.gz PDB format
PDB format 1uz9.json.gz
1uz9.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/uz/1uz9
https://data.pdbj.org/pub/pdb/validation_reports/uz/1uz9 ftp://data.pdbj.org/pub/pdb/validation_reports/uz/1uz9
ftp://data.pdbj.org/pub/pdb/validation_reports/uz/1uz9
 Links
Links Assembly
Assembly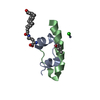

 Components
Components HOMO SAPIENS (human) / References: UniProt: P01308
HOMO SAPIENS (human) / References: UniProt: P01308 HOMO SAPIENS (human) / References: UniProt: P01308
HOMO SAPIENS (human) / References: UniProt: P01308







 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  SRS
SRS  / Beamline: PX9.6 / Wavelength: 0.87
/ Beamline: PX9.6 / Wavelength: 0.87  Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller






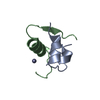



















































 PDBj
PDBj




















