+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1ben | ||||||
|---|---|---|---|---|---|---|---|
| Title | INSULIN COMPLEXED WITH 4-HYDROXYBENZAMIDE | ||||||
 Components Components | (HUMAN INSULIN) x 2 | ||||||
 Keywords Keywords | HORMONE / INSULIN / GLUCOSE METABOLISM | ||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of glycogen catabolic process / : / negative regulation of fatty acid metabolic process / Signaling by Insulin receptor / negative regulation of feeding behavior / IRS activation / Insulin processing / regulation of protein secretion / positive regulation of peptide hormone secretion / positive regulation of respiratory burst ...negative regulation of glycogen catabolic process / : / negative regulation of fatty acid metabolic process / Signaling by Insulin receptor / negative regulation of feeding behavior / IRS activation / Insulin processing / regulation of protein secretion / positive regulation of peptide hormone secretion / positive regulation of respiratory burst / negative regulation of acute inflammatory response / Regulation of gene expression in beta cells / alpha-beta T cell activation / Synthesis, secretion, and deacylation of Ghrelin / negative regulation of protein secretion / positive regulation of dendritic spine maintenance / negative regulation of gluconeogenesis / fatty acid homeostasis / positive regulation of glycogen biosynthetic process / positive regulation of insulin receptor signaling pathway / Signal attenuation / FOXO-mediated transcription of oxidative stress, metabolic and neuronal genes / negative regulation of respiratory burst involved in inflammatory response / negative regulation of lipid catabolic process / positive regulation of lipid biosynthetic process / negative regulation of oxidative stress-induced intrinsic apoptotic signaling pathway / nitric oxide-cGMP-mediated signaling / transport vesicle / regulation of protein localization to plasma membrane / positive regulation of nitric-oxide synthase activity / Insulin receptor recycling / COPI-mediated anterograde transport / negative regulation of reactive oxygen species biosynthetic process / positive regulation of brown fat cell differentiation / insulin-like growth factor receptor binding / NPAS4 regulates expression of target genes / neuron projection maintenance / endoplasmic reticulum-Golgi intermediate compartment membrane / positive regulation of mitotic nuclear division / Insulin receptor signalling cascade / positive regulation of glycolytic process / endosome lumen / acute-phase response / positive regulation of cytokine production / positive regulation of D-glucose import across plasma membrane / insulin receptor binding / positive regulation of long-term synaptic potentiation / positive regulation of protein secretion / positive regulation of cell differentiation / wound healing / Regulation of insulin secretion / hormone activity / positive regulation of neuron projection development / negative regulation of protein catabolic process / regulation of synaptic plasticity / positive regulation of protein localization to nucleus / Golgi lumen / cognition / vasodilation / glucose metabolic process / insulin receptor signaling pathway / cell-cell signaling / regulation of protein localization / glucose homeostasis / PI5P, PP2A and IER3 Regulate PI3K/AKT Signaling / positive regulation of cell growth / protease binding / secretory granule lumen / positive regulation of canonical NF-kappaB signal transduction / positive regulation of MAPK cascade / positive regulation of phosphatidylinositol 3-kinase/protein kinase B signal transduction / positive regulation of cell migration / G protein-coupled receptor signaling pathway / endoplasmic reticulum lumen / Amyloid fiber formation / receptor ligand activity / Golgi membrane / negative regulation of gene expression / positive regulation of cell population proliferation / positive regulation of gene expression / regulation of DNA-templated transcription / : / extracellular region / identical protein binding Similarity search - Function | ||||||
| Method |  X-RAY DIFFRACTION / Resolution: 1.4 Å X-RAY DIFFRACTION / Resolution: 1.4 Å | ||||||
 Authors Authors | Smith, G.D. / Ciszak, E. / Pangborn, W. | ||||||
 Citation Citation |  Journal: Protein Sci. / Year: 1996 Journal: Protein Sci. / Year: 1996Title: A novel complex of a phenolic derivative with insulin: structural features related to the T-->R transition. Authors: Smith, G.D. / Ciszak, E. / Pangborn, W. #1:  Journal: Structure / Year: 1995 Journal: Structure / Year: 1995Title: Role of C-Terminal B-Chain Residues in Insulin Assembly: The Structure of Hexameric Lysb28Prob29-Human Insulin Authors: Ciszak, E. / Beals, J.M. / Frank, B.H. / Baker, J.C. / Carter, N.D. / Smith, G.D. #2:  Journal: Biochemistry / Year: 1994 Journal: Biochemistry / Year: 1994Title: Crystallographic Evidence for Dual Coordination Around Zinc in the T3R3 Human Insulin Hexamer Authors: Ciszak, E. / Smith, G.D. #3:  Journal: Proc.Natl.Acad.Sci.USA / Year: 1994 Journal: Proc.Natl.Acad.Sci.USA / Year: 1994Title: The Structure of a Complex of Hexameric Insulin and 4'-Hydroxyacetanilide Authors: Smith, G.D. / Ciszak, E. #4:  Journal: Proteins / Year: 1992 Journal: Proteins / Year: 1992Title: The Structure of a Rhombohedral R6 Insulin/Phenol Complex Authors: Smith, G.D. / Dodson, G.G. #5:  Journal: Philos.Trans.R.Soc.London,Ser.B / Year: 1988 Journal: Philos.Trans.R.Soc.London,Ser.B / Year: 1988Title: The Structure of 2Zn Pig Insulin Crystals at 1.5A Resolution Authors: Baker, E.N. / Blundell, T.L. / Cutfield, J.F. / Cutfield, S.M. / Dodson, E.J. / Dodson, G.G. / Hodgkin, D.C. / Hubbard, R.E. / Isaacs, N.W. / Reynolds, C.D. / Sakabe, K. / Sakabe, N. / Vijayan, N.M. #6:  Journal: Proc.Natl.Acad.Sci.USA / Year: 1984 Journal: Proc.Natl.Acad.Sci.USA / Year: 1984Title: Structural Stability in the 4-Zinc Human Insulin Hexamer Authors: Smith, G.D. / Swenson, D.C. / Dodson, E.J. / Dodson, G.G. / Reynolds, C.D. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1ben.cif.gz 1ben.cif.gz | 55.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1ben.ent.gz pdb1ben.ent.gz | 41.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1ben.json.gz 1ben.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/be/1ben https://data.pdbj.org/pub/pdb/validation_reports/be/1ben ftp://data.pdbj.org/pub/pdb/validation_reports/be/1ben ftp://data.pdbj.org/pub/pdb/validation_reports/be/1ben | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 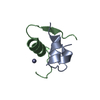
| |||||||||||||||||||||
| 2 | 
| |||||||||||||||||||||
| 3 | 
| |||||||||||||||||||||
| 4 | 
| |||||||||||||||||||||
| 5 | 
| |||||||||||||||||||||
| 6 | 
| |||||||||||||||||||||
| 7 | 
| |||||||||||||||||||||
| 8 | 
| |||||||||||||||||||||
| 9 | 
| |||||||||||||||||||||
| 10 | 
| |||||||||||||||||||||
| 11 | 
| |||||||||||||||||||||
| 12 | 
| |||||||||||||||||||||
| 13 | 
| |||||||||||||||||||||
| Unit cell |
| |||||||||||||||||||||
| Components on special symmetry positions |
| |||||||||||||||||||||
| Details | THE CRYSTALLOGRAPHIC ASYMMETRIC UNIT OF INSULIN CONSISTS OF TWO INSULIN MONOMERS EACH CONSISTING OF TWO HETEROCHAINS. THE ENTRY PRESENTS COORDINATES FOR MONOMER I (CHAIN IDENTIFIERS A AND B) AND II (CHAIN IDENTIFIERS C AND D). APPLYING THE THREE-FOLD CRYSTALLOGRAPHIC SYMMETRY AXIS YIELDS A HEXAMER AROUND THE AXIS. THERE ARE TWO ZINC IONS AND ONE CHLORIDE ION PER INSULIN HEXAMER LOCATED ON THE THREE-FOLD AXIS AND HAVE OCCUPANCIES OF 0.33. WATERS HOH 18, HOH 54, AND HOH 59 ARE LOCATED ON THE THREE-FOLD AXIS. |
- Components
Components
-Protein/peptide , 2 types, 4 molecules ACBD
| #1: Protein/peptide | Mass: 2383.698 Da / Num. of mol.: 2 / Source method: obtained synthetically / Details: GIFT OF LILLY RESEARCH LABORATORIES / References: UniProt: P01308 #2: Protein/peptide | Mass: 3433.953 Da / Num. of mol.: 2 / Source method: obtained synthetically / Details: GIFT OF LILLY RESEARCH LABORATORIES / References: UniProt: P01308 |
|---|
-Non-polymers , 4 types, 173 molecules 






| #3: Chemical | | #4: Chemical | #5: Chemical | ChemComp-CL / | #6: Water | ChemComp-HOH / | |
|---|
-Details
| Compound details | THE CONFORMATIONS OF THE TWO MONOMERS ARE DIFFERENT AS THE RESULT OF A DIFFERENCE IN CONFORMATION ...THE CONFORMATI |
|---|---|
| Has protein modification | Y |
| Nonpolymer details | THE MODEL ALSO CONTAINS TWO ZINC IONS. THE FIRST ZINC ION (ZN B 31) IS COORDINATED BY THREE ...THE MODEL ALSO CONTAINS TWO ZINC IONS. THE FIRST ZINC ION (ZN B 31) IS COORDINATE |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 6 X-RAY DIFFRACTION / Number of used crystals: 6 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.03 Å3/Da / Density % sol: 33 % Description: DATA WERE MEASURED FROM SIX CRYSTALS, GROWN IN MICROGRAVITY ON SHUTTLE FLIGHT, STS-60. THE R-AXIS-IIC SOFTWARE WAS USED TO INTEGRATE EACH IMAGE PLATE; SCALING AND MERGING WAS ...Description: DATA WERE MEASURED FROM SIX CRYSTALS, GROWN IN MICROGRAVITY ON SHUTTLE FLIGHT, STS-60. THE R-AXIS-IIC SOFTWARE WAS USED TO INTEGRATE EACH IMAGE PLATE; SCALING AND MERGING WAS ACCOMPLISHED WITH THE DREAM PROGRAM PACKAGE (R.H. BLESSING, CRYSTALLOGR. REV. 1, 3-58 (1987). | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | Method: slow cooling / pH: 5.7 Details: 0.05M SODIUM CITRATE, 0.007M ZINC ACETATE, 0.06M 4-HYDROXYBENZAMIDE, 1.0M SODIUM CHLORIDE, PH=5.7. CRYSTALS GROWN BY SLOW COOLING., slow cooling | ||||||||||||||||||||||||||||||||||||
| Crystal | *PLUS | ||||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Method: unknown | ||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction source | Wavelength: 1.5418 |
|---|---|
| Detector | Type: RIGAKU RAXIS IIC / Detector: IMAGE PLATE / Date: Feb 14, 1994 |
| Radiation | Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 1.4→80 Å / Num. obs: 18002 / % possible obs: 76.6 % / Observed criterion σ(I): 2 / Redundancy: 8.6 % / Rmerge(I) obs: 0.066 |
| Reflection | *PLUS Num. measured all: 159339 |
- Processing
Processing
| Software | Name: PROFFT / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Resolution: 1.4→8 Å / σ(F): 2 /
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 27.4 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.4→8 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS σ(I): 17907 / Rfactor all: 0.176 / Rfactor obs: 0.154 / Rfactor Rwork: 0.183 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS |
 Movie
Movie Controller
Controller













 PDBj
PDBj




















