Entry Database : PDB / ID : 1msoTitle T6 Human Insulin at 1.0 A Resolution Insulin A-Chain Insulin B-Chain Keywords / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Method / / Resolution : 1 Å Authors Smith, G.D. / Pangborn, W.A. / Blessing, R.H. Journal : Acta Crystallogr.,Sect.D / Year : 2003Title : The structure of T6 human insulin at 1.0 A resolution.Authors : Smith, G.D. / Pangborn, W.A. / Blessing, R.H. History Deposition Sep 19, 2002 Deposition site / Processing site Revision 1.0 Mar 4, 2003 Provider / Type Revision 1.1 Apr 28, 2008 Group Revision 1.2 Jul 13, 2011 Group / Version format complianceRevision 1.3 Oct 11, 2017 Group / Category Revision 1.4 Oct 16, 2024 Group Data collection / Database references ... Data collection / Database references / Derived calculations / Refinement description / Structure summary Category chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / pdbx_entry_details / pdbx_initial_refinement_model / pdbx_modification_feature / pdbx_struct_conn_angle / software / struct_conn / struct_site Item _database_2.pdbx_DOI / _database_2.pdbx_database_accession ... _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _pdbx_struct_conn_angle.ptnr1_auth_comp_id / _pdbx_struct_conn_angle.ptnr1_auth_seq_id / _pdbx_struct_conn_angle.ptnr1_label_asym_id / _pdbx_struct_conn_angle.ptnr1_label_atom_id / _pdbx_struct_conn_angle.ptnr1_label_comp_id / _pdbx_struct_conn_angle.ptnr1_label_seq_id / _pdbx_struct_conn_angle.ptnr1_symmetry / _pdbx_struct_conn_angle.ptnr3_auth_comp_id / _pdbx_struct_conn_angle.ptnr3_auth_seq_id / _pdbx_struct_conn_angle.ptnr3_label_asym_id / _pdbx_struct_conn_angle.ptnr3_label_atom_id / _pdbx_struct_conn_angle.ptnr3_label_comp_id / _pdbx_struct_conn_angle.ptnr3_label_seq_id / _pdbx_struct_conn_angle.ptnr3_symmetry / _pdbx_struct_conn_angle.value / _software.name / _struct_conn.pdbx_dist_value / _struct_conn.ptnr1_auth_asym_id / _struct_conn.ptnr1_auth_comp_id / _struct_conn.ptnr1_auth_seq_id / _struct_conn.ptnr1_label_asym_id / _struct_conn.ptnr1_label_atom_id / _struct_conn.ptnr1_label_comp_id / _struct_conn.ptnr1_label_seq_id / _struct_conn.ptnr1_symmetry / _struct_conn.ptnr2_auth_asym_id / _struct_conn.ptnr2_auth_comp_id / _struct_conn.ptnr2_auth_seq_id / _struct_conn.ptnr2_label_asym_id / _struct_conn.ptnr2_label_atom_id / _struct_conn.ptnr2_label_comp_id / _struct_conn.ptnr2_label_seq_id / _struct_conn.ptnr2_symmetry / _struct_site.pdbx_auth_asym_id / _struct_site.pdbx_auth_comp_id / _struct_site.pdbx_auth_seq_id
Show all Show less
 Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 1 Å
MOLECULAR REPLACEMENT / Resolution: 1 Å  Authors
Authors Citation
Citation Journal: Acta Crystallogr.,Sect.D / Year: 2003
Journal: Acta Crystallogr.,Sect.D / Year: 2003 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 1mso.cif.gz
1mso.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb1mso.ent.gz
pdb1mso.ent.gz PDB format
PDB format 1mso.json.gz
1mso.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/ms/1mso
https://data.pdbj.org/pub/pdb/validation_reports/ms/1mso ftp://data.pdbj.org/pub/pdb/validation_reports/ms/1mso
ftp://data.pdbj.org/pub/pdb/validation_reports/ms/1mso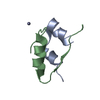
 Links
Links Assembly
Assembly

 Components
Components X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation ROTATING ANODE / Type: RIGAKU RU200 / Wavelength: 1.5418 Å
ROTATING ANODE / Type: RIGAKU RU200 / Wavelength: 1.5418 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller













 PDBj
PDBj




















