[English] 日本語
 Yorodumi
Yorodumi- PDB-1qiy: HUMAN INSULIN HEXAMERS WITH CHAIN B HIS MUTATED TO TYR COMPLEXED ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1qiy | ||||||
|---|---|---|---|---|---|---|---|
| Title | HUMAN INSULIN HEXAMERS WITH CHAIN B HIS MUTATED TO TYR COMPLEXED WITH PHENOL | ||||||
 Components Components |
| ||||||
 Keywords Keywords | HORMONE / GLUCOSE METABOLISM / DIABETES / INSULIN MUTANT | ||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of glycogen catabolic process / : / negative regulation of fatty acid metabolic process / Signaling by Insulin receptor / negative regulation of feeding behavior / IRS activation / Insulin processing / regulation of protein secretion / positive regulation of peptide hormone secretion / positive regulation of respiratory burst ...negative regulation of glycogen catabolic process / : / negative regulation of fatty acid metabolic process / Signaling by Insulin receptor / negative regulation of feeding behavior / IRS activation / Insulin processing / regulation of protein secretion / positive regulation of peptide hormone secretion / positive regulation of respiratory burst / negative regulation of acute inflammatory response / Regulation of gene expression in beta cells / alpha-beta T cell activation / Synthesis, secretion, and deacylation of Ghrelin / negative regulation of protein secretion / positive regulation of dendritic spine maintenance / negative regulation of gluconeogenesis / fatty acid homeostasis / positive regulation of glycogen biosynthetic process / positive regulation of insulin receptor signaling pathway / Signal attenuation / FOXO-mediated transcription of oxidative stress, metabolic and neuronal genes / negative regulation of respiratory burst involved in inflammatory response / negative regulation of lipid catabolic process / positive regulation of lipid biosynthetic process / negative regulation of oxidative stress-induced intrinsic apoptotic signaling pathway / nitric oxide-cGMP-mediated signaling / regulation of protein localization to plasma membrane / positive regulation of nitric-oxide synthase activity / transport vesicle / Insulin receptor recycling / COPI-mediated anterograde transport / positive regulation of brown fat cell differentiation / negative regulation of reactive oxygen species biosynthetic process / insulin-like growth factor receptor binding / NPAS4 regulates expression of target genes / neuron projection maintenance / endoplasmic reticulum-Golgi intermediate compartment membrane / positive regulation of mitotic nuclear division / Insulin receptor signalling cascade / positive regulation of glycolytic process / endosome lumen / acute-phase response / positive regulation of cytokine production / positive regulation of D-glucose import across plasma membrane / insulin receptor binding / positive regulation of long-term synaptic potentiation / positive regulation of protein secretion / positive regulation of cell differentiation / wound healing / Regulation of insulin secretion / hormone activity / positive regulation of neuron projection development / negative regulation of protein catabolic process / positive regulation of protein localization to nucleus / regulation of synaptic plasticity / Golgi lumen / cognition / vasodilation / glucose metabolic process / insulin receptor signaling pathway / cell-cell signaling / regulation of protein localization / glucose homeostasis / PI5P, PP2A and IER3 Regulate PI3K/AKT Signaling / positive regulation of cell growth / protease binding / secretory granule lumen / positive regulation of canonical NF-kappaB signal transduction / positive regulation of MAPK cascade / positive regulation of phosphatidylinositol 3-kinase/protein kinase B signal transduction / positive regulation of cell migration / endoplasmic reticulum lumen / G protein-coupled receptor signaling pathway / Amyloid fiber formation / receptor ligand activity / Golgi membrane / negative regulation of gene expression / positive regulation of cell population proliferation / positive regulation of gene expression / regulation of DNA-templated transcription / : / extracellular region / identical protein binding Similarity search - Function | ||||||
| Biological species |  HOMO SAPIENS (human) HOMO SAPIENS (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 2.3 Å MOLECULAR REPLACEMENT / Resolution: 2.3 Å | ||||||
 Authors Authors | Tang, L. / Whittingham, J.L. / Verma, C.S. / Caves, L.S.D. / Dodson, G.G. | ||||||
 Citation Citation |  Journal: Biochemistry / Year: 1999 Journal: Biochemistry / Year: 1999Title: Structural Consequences of the B5 Histidine --> Tyrosine Mutation in Human Insulin Characterized by X-Ray Crystallography and Conformational Analysis. Authors: Tang, L. / Whittingham, J.L. / Verma, C.S. / Caves, L.S.D. / Dodson, G.G. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1qiy.cif.gz 1qiy.cif.gz | 77.4 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1qiy.ent.gz pdb1qiy.ent.gz | 58.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1qiy.json.gz 1qiy.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/qi/1qiy https://data.pdbj.org/pub/pdb/validation_reports/qi/1qiy ftp://data.pdbj.org/pub/pdb/validation_reports/qi/1qiy ftp://data.pdbj.org/pub/pdb/validation_reports/qi/1qiy | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Details | THE ASYMMETRIC UNIT CONTAINS A 2ZN INSULIN HEXAMER,CONSISTING OF THREE EQUIVALENT DIMERS RELATED BY A NON-CRYSTALLOGRAPHIC 3-FOLD SYMMETRY AXIS. THE ZINC AND CHLORIDE IONS ARE LOCATED ON THIS 3-FOLD AXIS. |
- Components
Components
-Protein/peptide , 2 types, 12 molecules ACEGIKBDFHJL
| #1: Protein/peptide | Mass: 2383.698 Da / Num. of mol.: 6 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  HOMO SAPIENS (human) / Production host: HOMO SAPIENS (human) / Production host:  #2: Protein/peptide | Mass: 3458.980 Da / Num. of mol.: 6 / Mutation: YES Source method: isolated from a genetically manipulated source Source: (gene. exp.)  HOMO SAPIENS (human) / Production host: HOMO SAPIENS (human) / Production host:  |
|---|
-Non-polymers , 4 types, 101 molecules 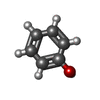






| #3: Chemical | ChemComp-IPH / #4: Chemical | #5: Chemical | #6: Water | ChemComp-HOH / | |
|---|
-Details
| Compound details | ENGINEERED RESIDUE IN CHAIN B, HIS 29 TO TYR ENGINEERED RESIDUE IN CHAIN D, HIS 29 TO TYR ...ENGINEERED |
|---|---|
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.5 Å3/Da / Density % sol: 50 % | |||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | pH: 6.8 Details: CRYSTALLISATION IN BATCH, 10 MG B5 TYR INSULIN DISSOLVED IN 2 ML 0.02M HCL. TO THIS ADDED 0.1 ML 0.12M ZINC ACETATE, 1.5 ML 0.2 M TRI-SODIUM CITRATE, 0.4 ML 2.5% (AQ.) PHENOL AND 120 MG NACL. ...Details: CRYSTALLISATION IN BATCH, 10 MG B5 TYR INSULIN DISSOLVED IN 2 ML 0.02M HCL. TO THIS ADDED 0.1 ML 0.12M ZINC ACETATE, 1.5 ML 0.2 M TRI-SODIUM CITRATE, 0.4 ML 2.5% (AQ.) PHENOL AND 120 MG NACL. PH ADJUSTED TO 6.5-7.8 . | |||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Method: batch method / PH range low: 7.8 / PH range high: 6.5 | |||||||||||||||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 289 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU RUH2R / Wavelength: 1.5418 ROTATING ANODE / Type: RIGAKU RUH2R / Wavelength: 1.5418 |
| Detector | Type: MULTIWIRE SIEMENS / Detector: AREA DETECTOR / Date: Jan 15, 1993 |
| Radiation | Monochromator: GRAPHITE(002) / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 2.26→36.8 Å / Num. obs: 13832 / % possible obs: 92 % / Redundancy: 1.8 % / Biso Wilson estimate: 34.8 Å2 / Rmerge(I) obs: 0.046 / Net I/σ(I): 10 |
| Reflection shell | Resolution: 2.2→2.3 Å / Redundancy: 1.6 % / Rmerge(I) obs: 0.107 / Mean I/σ(I) obs: 5 / % possible all: 95.5 |
| Reflection shell | *PLUS % possible obs: 95.5 % |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: R6 (NATIVE) INSULIN Resolution: 2.3→36.8 Å / σ(F): 0 Details: THE FOLLOWING SIDECHAINS HAVE BEEN ASSIGNED ZERO OCCUPANCIES DUE TO DISORDER, A4, A14, B13, B21, B29, B30, C4, D1, E5, F1, F21, F25, G4, H13, H21, J21, L1, L21, L30 THE FOLLOWING CHAIN ...Details: THE FOLLOWING SIDECHAINS HAVE BEEN ASSIGNED ZERO OCCUPANCIES DUE TO DISORDER, A4, A14, B13, B21, B29, B30, C4, D1, E5, F1, F21, F25, G4, H13, H21, J21, L1, L21, L30 THE FOLLOWING CHAIN TERMINAL RESIDUES HAVE BEEN ASSIGNED ZERO OCCUPANCIES DUE TO DISORDER, D29-D30, F29-F30, H29-H30, J29-J30
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 35.9 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.3→36.8 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj




















