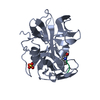
Entry Database : PDB / ID : 6ensTitle Structure of mouse wild-type RKIP Phosphatidylethanolamine-binding protein 1 Keywords / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Mus musculus (house mouse)Method / / / Resolution : 1.3 Å Authors Hirschbeck, M. / Koelmel, W. / Schindelin, H. / Lorenz, K. / Kisker, C. Funding support Organization Grant number Country National Institutes of Health GM087630 National Institutes of Health GM55694 German Research Foundation FZ82
Journal : Proc. Natl. Acad. Sci. U.S.A. / Year : 2017Title : Conserved salt-bridge competition triggered by phosphorylation regulates the protein interactome.Authors : Skinner, J.J. / Wang, S. / Lee, J. / Ong, C. / Sommese, R. / Sivaramakrishnan, S. / Koelmel, W. / Hirschbeck, M. / Schindelin, H. / Kisker, C. / Lorenz, K. / Sosnick, T.R. / Rosner, M.R. History Deposition Oct 6, 2017 Deposition site / Processing site Revision 1.0 Dec 13, 2017 Provider / Type Revision 1.1 Dec 20, 2017 Group / Category / citation_authorItem _citation.country / _citation.journal_abbrev ... _citation.country / _citation.journal_abbrev / _citation.journal_id_ASTM / _citation.journal_id_CSD / _citation.journal_id_ISSN / _citation.pdbx_database_id_PubMed / _citation.title / _citation_author.name Revision 1.2 Dec 27, 2017 Group / Category Item / _citation.page_first / _citation.page_lastRevision 1.3 Jan 17, 2024 Group / Database references / Refinement descriptionCategory chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model Item / _database_2.pdbx_database_accession
Show all Show less
 Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information
 X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.3 Å
MOLECULAR REPLACEMENT / Resolution: 1.3 Å  Authors
Authors United States,
United States,  Germany, 3items
Germany, 3items  Citation
Citation Journal: Proc. Natl. Acad. Sci. U.S.A. / Year: 2017
Journal: Proc. Natl. Acad. Sci. U.S.A. / Year: 2017 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 6ens.cif.gz
6ens.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb6ens.ent.gz
pdb6ens.ent.gz PDB format
PDB format 6ens.json.gz
6ens.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/en/6ens
https://data.pdbj.org/pub/pdb/validation_reports/en/6ens ftp://data.pdbj.org/pub/pdb/validation_reports/en/6ens
ftp://data.pdbj.org/pub/pdb/validation_reports/en/6ens

 Links
Links Assembly
Assembly
 Components
Components

 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  BESSY
BESSY  / Beamline: 14.1 / Wavelength: 0.91841 Å
/ Beamline: 14.1 / Wavelength: 0.91841 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller












 PDBj
PDBj



