+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1qng | ||||||
|---|---|---|---|---|---|---|---|
| Title | Plasmodium falciparum Cyclophilin complexed with Cyclosporin A | ||||||
 Components Components |
| ||||||
 Keywords Keywords | ISOMERASE/IMMUNOSUPPRESSANT / ISOMERASE-IMMUNOSUPPRESSANT COMPLEX / CYCLOPHILIN-CYCLOSPORIN COMPLEX / CYCLOSPORIN A / IMMUNOSUPPRESSANT / CYCLOPHILIN | ||||||
| Function / homology |  Function and homology information Function and homology informationcyclosporin A binding / peptidylprolyl isomerase / peptidyl-prolyl cis-trans isomerase activity / protein folding / cytoplasm Similarity search - Function | ||||||
| Biological species |   TOLYPOCLADIUM INFLATUM (fungus) TOLYPOCLADIUM INFLATUM (fungus) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 2.1 Å MOLECULAR REPLACEMENT / Resolution: 2.1 Å | ||||||
 Authors Authors | Peterson, M.R. / Hall, D.R. / Hunter, W.N. | ||||||
 Citation Citation |  Journal: J.Mol.Biol. / Year: 2000 Journal: J.Mol.Biol. / Year: 2000Title: The Three-Dimensional Structure of a Plasmodium Falciparum Cyclophilin in Complex with the Potent Anti-Malarial Cyclosporin A Authors: Peterson, M.R. / Hall, D.R. / Berriman, M. / Leonard, G.A. / Fairlamb, A.H. / Hunter, W.N. #1: Journal: Biochem.J. / Year: 1998 Title: Detailed Characterization of a Cyclophilin from the Human Malaria Parasite Plasmodium Falciparum Authors: Berriman, M. / Fairlamb, A.H. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1qng.cif.gz 1qng.cif.gz | 55.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1qng.ent.gz pdb1qng.ent.gz | 38.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1qng.json.gz 1qng.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/qn/1qng https://data.pdbj.org/pub/pdb/validation_reports/qn/1qng ftp://data.pdbj.org/pub/pdb/validation_reports/qn/1qng ftp://data.pdbj.org/pub/pdb/validation_reports/qn/1qng | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  1qnhSC S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 18845.375 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Production host:  |
|---|---|
| #2: Protein/peptide | 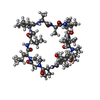  Type: Cyclic peptide / Class: Immunosuppressant / Mass: 1220.625 Da / Num. of mol.: 1 / Source method: isolated from a natural source Type: Cyclic peptide / Class: Immunosuppressant / Mass: 1220.625 Da / Num. of mol.: 1 / Source method: isolated from a natural sourceDetails: CYCLOSPORIN IS A CYCLIC UNDECAPEPTIDE. CYCLIZATION IS ACHIEVED BY LINKING THE N- AND THE C- TERMINI. Source: (natural)  TOLYPOCLADIUM INFLATUM (fungus) / References: NOR: NOR00033, Cyclosporin A TOLYPOCLADIUM INFLATUM (fungus) / References: NOR: NOR00033, Cyclosporin A |
| #3: Water | ChemComp-HOH / |
| Compound details | CYCLOSPORIN IS A CYCLIC UNDECAPEPTIDE. HERE, CYCLOSPORIN A IS REPRESENTED BY THE SEQUENCE (SEQRES) ...CYCLOSPORI |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.05 Å3/Da / Density % sol: 40 % | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | pH: 7.5 / Details: pH 7.50 | ||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Method: vapor diffusion, hanging drop | ||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU RUH3R / Wavelength: 1.5418 ROTATING ANODE / Type: RIGAKU RUH3R / Wavelength: 1.5418 |
| Detector | Type: R-AXIS IV / Detector: IMAGE PLATE / Details: MIRRORS |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 2.1→32.8 Å / Num. obs: 8101 / % possible obs: 92.3 % / Redundancy: 3 % / Biso Wilson estimate: 26.5 Å2 / Rmerge(I) obs: 0.035 / Net I/σ(I): 26.3 |
| Reflection shell | Resolution: 2.1→2.15 Å / Redundancy: 3 % / Rmerge(I) obs: 0.061 / Mean I/σ(I) obs: 15 / % possible all: 89.5 |
| Reflection | *PLUS Num. measured all: 61405 |
| Reflection shell | *PLUS % possible obs: 89.5 % |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 1QNH Resolution: 2.1→32.8 Å / Isotropic thermal model: RESTRAINED / Cross valid method: THROUGHOUT / σ(F): 0 Details: REFINEMENT AT EARLY STAGES UTILISED REFMAC. THE SIDE CHAIN OF ARG49 IS DISORDERED
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 21.8 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.1→32.8 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.1→2.2 Å / Total num. of bins used: 8
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Xplor file |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS Type: c_angle_deg / Dev ideal: 1.275 |
 Movie
Movie Controller
Controller







































 PDBj
PDBj


