+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7epe | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Crystal structure of mGlu2 bound to NAM563 | |||||||||||||||
 Components Components | Metabotropic glutamate receptor 2 | |||||||||||||||
 Keywords Keywords | MEMBRANE PROTEIN / GPCR | |||||||||||||||
| Function / homology | Flavodoxin domain / Rossmann fold / 3-Layer(aba) Sandwich / Alpha Beta / FLAVIN MONONUCLEOTIDE / Chem-J9R Function and homology information Function and homology information | |||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.5 Å MOLECULAR REPLACEMENT / Resolution: 2.5 Å | |||||||||||||||
 Authors Authors | Du, J. / Wang, D. / Lin, S. / Han, S. / Wu, B. / Zhao, Q. | |||||||||||||||
| Funding support |  China, 4items China, 4items
| |||||||||||||||
 Citation Citation |  Journal: Nature / Year: 2021 Journal: Nature / Year: 2021Title: Structures of human mGlu2 and mGlu7 homo- and heterodimers. Authors: Juan Du / Dejian Wang / Hongcheng Fan / Chanjuan Xu / Linhua Tai / Shuling Lin / Shuo Han / Qiuxiang Tan / Xinwei Wang / Tuo Xu / Hui Zhang / Xiaojing Chu / Cuiying Yi / Peng Liu / Xiaomei ...Authors: Juan Du / Dejian Wang / Hongcheng Fan / Chanjuan Xu / Linhua Tai / Shuling Lin / Shuo Han / Qiuxiang Tan / Xinwei Wang / Tuo Xu / Hui Zhang / Xiaojing Chu / Cuiying Yi / Peng Liu / Xiaomei Wang / Yu Zhou / Jean-Philippe Pin / Philippe Rondard / Hong Liu / Jianfeng Liu / Fei Sun / Beili Wu / Qiang Zhao /   Abstract: The metabotropic glutamate receptors (mGlus) are involved in the modulation of synaptic transmission and neuronal excitability in the central nervous system. These receptors probably exist as both ...The metabotropic glutamate receptors (mGlus) are involved in the modulation of synaptic transmission and neuronal excitability in the central nervous system. These receptors probably exist as both homo- and heterodimers that have unique pharmacological and functional properties. Here we report four cryo-electron microscopy structures of the human mGlu subtypes mGlu2 and mGlu7, including inactive mGlu2 and mGlu7 homodimers; mGlu2 homodimer bound to an agonist and a positive allosteric modulator; and inactive mGlu2-mGlu7 heterodimer. We observed a subtype-dependent dimerization mode for these mGlus, as a unique dimer interface that is mediated by helix IV (and that is important for limiting receptor activity) exists only in the inactive mGlu2 structure. The structures provide molecular details of the inter- and intra-subunit conformational changes that are required for receptor activation, which distinguish class C G-protein-coupled receptors from those in classes A and B. Furthermore, our structure and functional studies of the mGlu2-mGlu7 heterodimer suggest that the mGlu7 subunit has a dominant role in controlling dimeric association and G-protein activation in the heterodimer. These insights into mGlu homo- and heterodimers highlight the complex landscape of mGlu dimerization and activation. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7epe.cif.gz 7epe.cif.gz | 92 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7epe.ent.gz pdb7epe.ent.gz | 65.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7epe.json.gz 7epe.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ep/7epe https://data.pdbj.org/pub/pdb/validation_reports/ep/7epe ftp://data.pdbj.org/pub/pdb/validation_reports/ep/7epe ftp://data.pdbj.org/pub/pdb/validation_reports/ep/7epe | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  7epaC  7epbC  7epcC  7epdC  7epfC  1i1oS  4or2S S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 48496.578 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Production host: Homo sapiens (human) / Production host:  Baculovirus expression vector pFastBac1-HM Baculovirus expression vector pFastBac1-HM |
|---|---|
| #2: Chemical | ChemComp-FMN / |
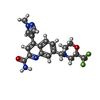
| #3: Chemical | ChemComp-J9R / |
| Has ligand of interest | Y |
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.53 Å3/Da / Density % sol: 51.36 % |
|---|---|
| Crystal grow | Temperature: 293.15 K / Method: lipidic cubic phase Details: 100 mM HEPES, pH 7.0, 100~200 mM NaCl, 100~200 mM MgCl2, and 20~30%(v/v) PEG 400 |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SPring-8 SPring-8  / Beamline: BL41XU / Wavelength: 1 Å / Beamline: BL41XU / Wavelength: 1 Å |
| Detector | Type: DECTRIS PILATUS3 6M / Detector: PIXEL / Date: May 8, 2019 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1 Å / Relative weight: 1 |
| Reflection | Resolution: 2.5→50 Å / Num. obs: 17691 / % possible obs: 99.2 % / Redundancy: 9.3 % / Biso Wilson estimate: 72.93 Å2 / Rmerge(I) obs: 0.144 / Net I/σ(I): 28.1 |
| Reflection shell | Resolution: 2.5→2.59 Å / Num. unique obs: 1681 / CC1/2: 0.514 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 4OR2, 1I1O Resolution: 2.5→32.82 Å / Cross valid method: FREE R-VALUE Stereochemistry target values: GeoStd + Monomer Library + CDL v1.2
| ||||||||||||||||||||||||
| Displacement parameters | Biso mean: 84.49 Å2 | ||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.5→32.82 Å
| ||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.5→2.66 Å
|
 Movie
Movie Controller
Controller

















 PDBj
PDBj