Entry Database : PDB / ID : 4ktsTitle Bovine trypsin in complex with microviridin J at pH 8.5 Cationic trypsin Microviridin Keywords / / / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Microcystis aeruginosa MRC (bacteria)Bos taurus (cattle)Method / / / Resolution : 1.3 Å Authors Quitterer, F. / Groll, M. / Hertweck, C. / Dittmann, E. Journal : Angew.Chem.Int.Ed.Engl. / Year : 2014Title : Harnessing the evolvability of tricyclic microviridins to dissect protease-inhibitor interactions.Authors : Weiz, A.R. / Ishida, K. / Quitterer, F. / Meyer, S. / Kehr, J.C. / Muller, K.M. / Groll, M. / Hertweck, C. / Dittmann, E. History Deposition May 21, 2013 Deposition site / Processing site Revision 1.0 Apr 2, 2014 Provider / Type Revision 1.1 Apr 16, 2014 Group Revision 1.2 Sep 20, 2023 Group Data collection / Database references ... Data collection / Database references / Derived calculations / Refinement description Category chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model / pdbx_struct_conn_angle / struct_conn / struct_conn_type / struct_ref_seq_dif / struct_site Item _database_2.pdbx_DOI / _database_2.pdbx_database_accession ... _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _pdbx_struct_conn_angle.ptnr1_auth_comp_id / _pdbx_struct_conn_angle.ptnr1_auth_seq_id / _pdbx_struct_conn_angle.ptnr1_label_atom_id / _pdbx_struct_conn_angle.ptnr1_label_comp_id / _pdbx_struct_conn_angle.ptnr1_label_seq_id / _pdbx_struct_conn_angle.ptnr3_auth_comp_id / _pdbx_struct_conn_angle.ptnr3_auth_seq_id / _pdbx_struct_conn_angle.ptnr3_label_atom_id / _pdbx_struct_conn_angle.ptnr3_label_comp_id / _pdbx_struct_conn_angle.ptnr3_label_seq_id / _pdbx_struct_conn_angle.value / _struct_conn.conn_type_id / _struct_conn.id / _struct_conn.pdbx_dist_value / _struct_conn.pdbx_leaving_atom_flag / _struct_conn.ptnr1_auth_asym_id / _struct_conn.ptnr1_auth_comp_id / _struct_conn.ptnr1_auth_seq_id / _struct_conn.ptnr1_label_asym_id / _struct_conn.ptnr1_label_atom_id / _struct_conn.ptnr1_label_comp_id / _struct_conn.ptnr1_label_seq_id / _struct_conn.ptnr2_auth_asym_id / _struct_conn.ptnr2_auth_comp_id / _struct_conn.ptnr2_auth_seq_id / _struct_conn.ptnr2_label_asym_id / _struct_conn.ptnr2_label_atom_id / _struct_conn.ptnr2_label_comp_id / _struct_conn.ptnr2_label_seq_id / _struct_conn_type.id / _struct_ref_seq_dif.details / _struct_site.pdbx_auth_asym_id / _struct_site.pdbx_auth_comp_id / _struct_site.pdbx_auth_seq_id
Show all Show less
 Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information Microcystis aeruginosa MRC (bacteria)
Microcystis aeruginosa MRC (bacteria)
 X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.3 Å
MOLECULAR REPLACEMENT / Resolution: 1.3 Å  Authors
Authors Citation
Citation Journal: Angew.Chem.Int.Ed.Engl. / Year: 2014
Journal: Angew.Chem.Int.Ed.Engl. / Year: 2014 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 4kts.cif.gz
4kts.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb4kts.ent.gz
pdb4kts.ent.gz PDB format
PDB format 4kts.json.gz
4kts.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads 4kts_validation.pdf.gz
4kts_validation.pdf.gz wwPDB validaton report
wwPDB validaton report 4kts_full_validation.pdf.gz
4kts_full_validation.pdf.gz 4kts_validation.xml.gz
4kts_validation.xml.gz 4kts_validation.cif.gz
4kts_validation.cif.gz https://data.pdbj.org/pub/pdb/validation_reports/kt/4kts
https://data.pdbj.org/pub/pdb/validation_reports/kt/4kts ftp://data.pdbj.org/pub/pdb/validation_reports/kt/4kts
ftp://data.pdbj.org/pub/pdb/validation_reports/kt/4kts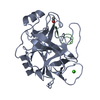

 Links
Links Assembly
Assembly
 Components
Components
 Type: Cyclic peptide / Class: Enzyme inhibitor / Mass: 1724.867 Da / Num. of mol.: 1
Type: Cyclic peptide / Class: Enzyme inhibitor / Mass: 1724.867 Da / Num. of mol.: 1 Microcystis aeruginosa MRC (bacteria) / Gene: mdnA / Production host:
Microcystis aeruginosa MRC (bacteria) / Gene: mdnA / Production host: 
 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  SLS
SLS  / Beamline: X06SA / Wavelength: 1 Å
/ Beamline: X06SA / Wavelength: 1 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller













 PDBj
PDBj





