[English] 日本語
 Yorodumi
Yorodumi- PDB-2wv5: Crystal structure of foot-and-mouth disease virus 3C protease in ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2wv5 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of foot-and-mouth disease virus 3C protease in complex with a decameric peptide corresponding to the VP1-2A cleavage junction with a GLN to Glu substitution at P1 | ||||||
 Components Components |
| ||||||
 Keywords Keywords | HYDROLASE/PEPTIDE / 3C PROTEASE / HYDROLASE / VIRAL PROTEIN / HYDROLASE PEPTIDE COMPLEX / HYDROLASE-PEPTIDE complex | ||||||
| Function / homology |  Function and homology information Function and homology informationL-peptidase / symbiont-mediated perturbation of host chromatin organization / picornain 3C / T=pseudo3 icosahedral viral capsid / host cell cytoplasmic vesicle membrane / viral capsid / ribonucleoside triphosphate phosphatase activity / host cell / nucleoside-triphosphate phosphatase / channel activity ...L-peptidase / symbiont-mediated perturbation of host chromatin organization / picornain 3C / T=pseudo3 icosahedral viral capsid / host cell cytoplasmic vesicle membrane / viral capsid / ribonucleoside triphosphate phosphatase activity / host cell / nucleoside-triphosphate phosphatase / channel activity / clathrin-dependent endocytosis of virus by host cell / monoatomic ion transmembrane transport / RNA helicase activity / viral protein processing / host cell endoplasmic reticulum membrane / symbiont-mediated activation of host autophagy / RNA-directed RNA polymerase / cysteine-type endopeptidase activity / viral RNA genome replication / RNA-directed RNA polymerase activity / symbiont entry into host cell / DNA-templated transcription / virion attachment to host cell / host cell nucleus / structural molecule activity / proteolysis / RNA binding / ATP binding Similarity search - Function | ||||||
| Biological species |   FOOT-AND-MOUTH DISEASE VIRUS FOOT-AND-MOUTH DISEASE VIRUS | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.7 Å MOLECULAR REPLACEMENT / Resolution: 2.7 Å | ||||||
 Authors Authors | Zunszain, P.A. / Knox, S.R. / Sweeney, T.R. / Yang, J. / Roque-Rosell, N. / Belsham, G.J. / Leatherbarrow, R.J. / Curry, S. | ||||||
 Citation Citation |  Journal: J.Mol.Biol. / Year: 2010 Journal: J.Mol.Biol. / Year: 2010Title: Insights Into Cleavage Specificity from the Crystal Structure of Foot-and-Mouth Disease Virus 3C Protease Complexed with a Peptide Substrate. Authors: Zunszain, P.A. / Knox, S.R. / Sweeney, T.R. / Yang, J. / Roque-Rosell, N. / Belsham, G.J. / Leatherbarrow, R.J. / Curry, S. | ||||||
| History |
| ||||||
| Remark 700 | SHEET DETERMINATION METHOD: DSSP THE SHEETS PRESENTED AS "AB" IN EACH CHAIN ON SHEET RECORDS BELOW ... SHEET DETERMINATION METHOD: DSSP THE SHEETS PRESENTED AS "AB" IN EACH CHAIN ON SHEET RECORDS BELOW IS ACTUALLY AN 6-STRANDED BARREL THIS IS REPRESENTED BY A 7-STRANDED SHEET IN WHICH THE FIRST AND LAST STRANDS ARE IDENTICAL. THE SHEETS PRESENTED AS "BB" IN EACH CHAIN ON SHEET RECORDS BELOW IS ACTUALLY AN 6-STRANDED BARREL THIS IS REPRESENTED BY A 7-STRANDED SHEET IN WHICH THE FIRST AND LAST STRANDS ARE IDENTICAL. THE SHEETS PRESENTED AS "CB" IN EACH CHAIN ON SHEET RECORDS BELOW IS ACTUALLY AN -4-STRANDED BARREL THIS IS REPRESENTED BY A -3-STRANDED SHEET IN WHICH THE FIRST AND LAST STRANDS ARE IDENTICAL. THE SHEETS PRESENTED AS "DB" IN EACH CHAIN ON SHEET RECORDS BELOW IS ACTUALLY AN -2-STRANDED BARREL THIS IS REPRESENTED BY A -1-STRANDED SHEET IN WHICH THE FIRST AND LAST STRANDS ARE IDENTICAL. THE SHEET STRUCTURE OF THIS MOLECULE IS BIFURCATED. IN ORDER TO REPRESENT THIS FEATURE IN THE SHEET RECORDS BELOW, TWO SHEETS ARE DEFINED. |
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2wv5.cif.gz 2wv5.cif.gz | 161.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2wv5.ent.gz pdb2wv5.ent.gz | 127.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2wv5.json.gz 2wv5.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/wv/2wv5 https://data.pdbj.org/pub/pdb/validation_reports/wv/2wv5 ftp://data.pdbj.org/pub/pdb/validation_reports/wv/2wv5 ftp://data.pdbj.org/pub/pdb/validation_reports/wv/2wv5 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  2wv4C  2j92S S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 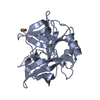
| ||||||||||||||||
| 2 | 
| ||||||||||||||||
| 3 | 
| ||||||||||||||||
| 4 | 
| ||||||||||||||||
| Unit cell |
| ||||||||||||||||
| Noncrystallographic symmetry (NCS) | NCS oper:
|
- Components
Components
| #1: Protein | Mass: 23049.654 Da / Num. of mol.: 4 / Mutation: YES Source method: isolated from a genetically manipulated source Source: (gene. exp.)   FOOT-AND-MOUTH DISEASE VIRUS / Strain: A10-61 / Production host: FOOT-AND-MOUTH DISEASE VIRUS / Strain: A10-61 / Production host:  #2: Protein/peptide | Mass: 1144.297 Da / Num. of mol.: 4 / Fragment: VP1-2A CLEAVAGE JUNCTION (P5-P5'), RESIDUES 29-38 / Mutation: YES / Source method: obtained synthetically / Source: (synth.)   FOOT-AND-MOUTH DISEASE VIRUS / References: UniProt: Q65050, UniProt: P03306*PLUS FOOT-AND-MOUTH DISEASE VIRUS / References: UniProt: Q65050, UniProt: P03306*PLUS#3: Water | ChemComp-HOH / | Compound details | ENGINEERED RESIDUE IN CHAIN A, CYS1744 TO LYS ENGINEERED RESIDUE IN CHAIN A, CYS1791 TO LEU ...ENGINEERED | Has protein modification | Y | Sequence details | CONTAINS ADDITIONAL GLY AS RESIDUE 1. LAST 6 RESIDUES AT C- TERM MISSING FROM OUR STRUCTURE. ...CONTAINS ADDITIONAL | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.25 Å3/Da / Density % sol: 45 % / Description: NONE |
|---|---|
| Crystal grow | pH: 8 / Details: SEE PAPER, pH 8 |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: ID14-2 / Wavelength: 0.934 / Beamline: ID14-2 / Wavelength: 0.934 |
| Detector | Type: ADSC CCD / Detector: CCD / Date: Dec 15, 2006 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.934 Å / Relative weight: 1 |
| Reflection | Resolution: 2.7→63.2 Å / Num. obs: 16981 / % possible obs: 76.1 % / Observed criterion σ(I): 0 / Redundancy: 2.4 % / Biso Wilson estimate: 45.5 Å2 / Rmerge(I) obs: 0.09 / Net I/σ(I): 8.6 |
| Reflection shell | Resolution: 2.7→2.85 Å / Redundancy: 2.6 % / Rmerge(I) obs: 0.39 / Mean I/σ(I) obs: 3.2 / % possible all: 60.8 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 2J92 Resolution: 2.7→56.33 Å / Rfactor Rfree error: 0.01 / Data cutoff high absF: 666675.7 / Data cutoff low absF: 0 / Isotropic thermal model: RESTRAINED / Cross valid method: THROUGHOUT / σ(F): 0 / Details: BULK SOLVENT MODEL USED
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Solvent model: FLAT MODEL / Bsol: 51.132 Å2 / ksol: 0.4 e/Å3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 32.1 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.7→56.33 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints NCS | NCS model details: RESTRAINTS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.7→2.87 Å / Rfactor Rfree error: 0.037 / Total num. of bins used: 6
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Xplor file |
|
 Movie
Movie Controller
Controller













 PDBj
PDBj


