[English] 日本語
 Yorodumi
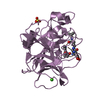
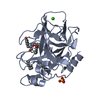
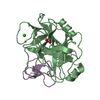
Yorodumi- PDB-1elg: NATURE OF THE INACTIVATION OF ELASTASE BY N-PEPTIDYL-O-AROYL HYDR... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1elg | ||||||
|---|---|---|---|---|---|---|---|
| Title | NATURE OF THE INACTIVATION OF ELASTASE BY N-PEPTIDYL-O-AROYL HYDROXYLAMINE AS A FUNCTION OF PH | ||||||
 Components Components | PORCINE PANCREATIC ELASTASE | ||||||
 Keywords Keywords | COMPLEX (HYDROLASE/INHIBITOR) / COMPLEX (HYDROLASE-INHIBITOR) / COMPLEX (HYDROLASE-INHIBITOR) complex | ||||||
| Function / homology |  Function and homology information Function and homology informationpancreatic elastase / serine-type endopeptidase activity / proteolysis / : / metal ion binding Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / Resolution: 1.65 Å X-RAY DIFFRACTION / Resolution: 1.65 Å | ||||||
 Authors Authors | Ding, X. / Rasmussen, B. / Demuth, H.-U. / Ringe, D. / Steinmetz, A.C.U. | ||||||
 Citation Citation |  Journal: Biochemistry / Year: 1995 Journal: Biochemistry / Year: 1995Title: Nature of the inactivation of elastase by N-peptidyl-O-aroyl hydroxylamine as a function of pH. Authors: Ding, X. / Rasmussen, B.F. / Demuth, H.U. / Ringe, D. / Steinmetz, A.C. #1:  Journal: Biochemistry / Year: 1994 Journal: Biochemistry / Year: 1994Title: Direct Structural Observation of an Acyl-Enzyme Intermediate in the Hydrolysis of an Ester Substrate by Elastase Authors: Ding, X. / Rasmussen, B.F. / Petsko, G.A. / Ringe, D. #2:  Journal: J.Enzyme Inhib. / Year: 1991 Journal: J.Enzyme Inhib. / Year: 1991Title: Inhibition of Proteases with Enkephalin-Analogue Inhibitors Authors: Demuth, H.-U. / Silberring, J. / Nyberg, F. #3:  Journal: J.Am.Chem.Soc. / Year: 1991 Journal: J.Am.Chem.Soc. / Year: 1991Title: Competing Redox and Inactivation Processes in the Inhibition of Cysteine Proteinases by Peptidyl O-Acylhydroxamates. 13C and 15N NMR Evidence for a Novel Sulfenamide Enzyme Adduct Authors: Robinson, V.J. / Coles, P.J. / Smith, R.A. / Krantz, A. #4:  Journal: J.Org.Chem. / Year: 1989 Journal: J.Org.Chem. / Year: 1989Title: N-O Bond Fission as the Rate-Determining Step in the Aqueous Conversion of N-Peptiyl-O-(P-Nitrobenzoyl)Hydroxylamines to P-Nitrobenzoic Acid and Peptidylhydroxamic Acids Authors: Demuth, H.-U. / Fischer, G. / Barth, A. / Schowen, R.L. #5:  Journal: J.Am.Chem.Soc. / Year: 1989 Journal: J.Am.Chem.Soc. / Year: 1989Title: Crystal Structure of the Covalent Complex Formed by a Peptidyl Difluoro Keto Amide with Porcine Pancreatic Elastase at 1.78 Angstroms Resolution Authors: Takahashi, L.H. / Radhakrishnan, R. / Rosenfield Junior, R.E. / Meyer Junior, E.F. / Trainor, D.A. | ||||||
| History |
| ||||||
| Remark 700 | SHEET THE TWO SEVEN STRANDED SHEETS IN THIS STRUCTURE ARE REALLY SIX STRANDED BETA BARRELS. THIS IS ...SHEET THE TWO SEVEN STRANDED SHEETS IN THIS STRUCTURE ARE REALLY SIX STRANDED BETA BARRELS. THIS IS DENOTED BY THE FIRST STRAND RECURRING AS THE LAST STRAND. |
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1elg.cif.gz 1elg.cif.gz | 64.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1elg.ent.gz pdb1elg.ent.gz | 46.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1elg.json.gz 1elg.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/el/1elg https://data.pdbj.org/pub/pdb/validation_reports/el/1elg ftp://data.pdbj.org/pub/pdb/validation_reports/el/1elg ftp://data.pdbj.org/pub/pdb/validation_reports/el/1elg | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 25928.031 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  |
|---|---|
| #2: Chemical | ChemComp-CA / |
| #3: Chemical | ChemComp-BAA / ( |
| #4: Water | ChemComp-HOH / |
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION X-RAY DIFFRACTION |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.15 Å3/Da / Density % sol: 42.75 % | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | *PLUS pH: 5 / Method: vapor diffusion, sitting drop | ||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Radiation | Scattering type: x-ray |
|---|---|
| Radiation wavelength | Relative weight: 1 |
| Reflection | Resolution: 1.65→10 Å / Num. obs: 23730 / % possible obs: 85 % / Observed criterion σ(I): 2 |
| Reflection | *PLUS Lowest resolution: 44 Å / Num. obs: 27890 / % possible obs: 100 % / Observed criterion σ(I): 0 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Rfactor Rwork: 0.17 / Rfactor obs: 0.17 / Highest resolution: 1.65 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Highest resolution: 1.65 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS Lowest resolution: 10 Å / Num. reflection all: 23730 / σ(I): 2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS |
 Movie
Movie Controller
Controller




















 PDBj
PDBj





