[English] 日本語
 Yorodumi
Yorodumi- EMDB-3851: Near-atomic resolution fibril structure of complete amyloid-beta(... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-3851 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Near-atomic resolution fibril structure of complete amyloid-beta(1-42) by cryo-EM | |||||||||
 Map data Map data | The density map was sharpened by a B-factor of -50 Ang^2 and filtered to 3.5 Ang. This density map was used for model building and refinement. | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | amyloid / fibril / aggregation / Alzheimer's disease / Protein fibril | |||||||||
| Function / homology |  Function and homology information Function and homology informationamyloid-beta complex / growth cone lamellipodium / cellular response to norepinephrine stimulus / collateral sprouting in absence of injury / growth cone filopodium / microglia development / Formyl peptide receptors bind formyl peptides and many other ligands / axo-dendritic transport / regulation of Wnt signaling pathway / axon midline choice point recognition ...amyloid-beta complex / growth cone lamellipodium / cellular response to norepinephrine stimulus / collateral sprouting in absence of injury / growth cone filopodium / microglia development / Formyl peptide receptors bind formyl peptides and many other ligands / axo-dendritic transport / regulation of Wnt signaling pathway / axon midline choice point recognition / regulation of synapse structure or activity / hippocampal neuron apoptotic process / astrocyte activation involved in immune response / NMDA selective glutamate receptor signaling pathway / regulation of spontaneous synaptic transmission / mating behavior / growth factor receptor binding / peptidase activator activity / Insertion of tail-anchored proteins into the endoplasmic reticulum membrane / PTB domain binding / positive regulation of amyloid fibril formation / Golgi-associated vesicle / astrocyte projection / Lysosome Vesicle Biogenesis / neuron remodeling / Deregulated CDK5 triggers multiple neurodegenerative pathways in Alzheimer's disease models / nuclear envelope lumen / dendrite development / TRAF6 mediated NF-kB activation / positive regulation of protein metabolic process / signaling receptor activator activity / negative regulation of long-term synaptic potentiation / Advanced glycosylation endproduct receptor signaling / transition metal ion binding / The NLRP3 inflammasome / regulation of multicellular organism growth / main axon / modulation of excitatory postsynaptic potential / intracellular copper ion homeostasis / ECM proteoglycans / response to insulin-like growth factor stimulus / regulation of presynapse assembly / positive regulation of T cell migration / neuronal dense core vesicle / Purinergic signaling in leishmaniasis infection / cellular response to manganese ion / positive regulation of chemokine production / Notch signaling pathway / swimming behavior / neuron projection maintenance / extracellular matrix organization / clathrin-coated pit / astrocyte activation / axonogenesis / positive regulation of mitotic cell cycle / Mitochondrial protein degradation / positive regulation of calcium-mediated signaling / ionotropic glutamate receptor signaling pathway / platelet alpha granule lumen / response to interleukin-1 / regulation of neuron apoptotic process / cellular response to cAMP / cellular response to copper ion / positive regulation of glycolytic process / endosome lumen / positive regulation of long-term synaptic potentiation / trans-Golgi network membrane / central nervous system development / dendritic shaft / positive regulation of interleukin-1 beta production / protein serine/threonine kinase binding / learning / adult locomotory behavior / Post-translational protein phosphorylation / serine-type endopeptidase inhibitor activity / locomotory behavior / microglial cell activation / cellular response to nerve growth factor stimulus / TAK1-dependent IKK and NF-kappa-B activation / positive regulation of non-canonical NF-kappaB signal transduction / synapse organization / visual learning / regulation of long-term neuronal synaptic plasticity / positive regulation of interleukin-6 production / recycling endosome / Golgi lumen / positive regulation of JNK cascade / response to lead ion / cognition / Regulation of Insulin-like Growth Factor (IGF) transport and uptake by Insulin-like Growth Factor Binding Proteins (IGFBPs) / cellular response to amyloid-beta / endocytosis / positive regulation of tumor necrosis factor production / neuron projection development / positive regulation of inflammatory response / calcium ion transport / Platelet degranulation / regulation of translation / heparin binding / regulation of gene expression Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
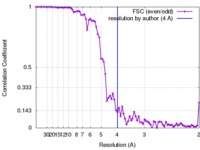
| Method | helical reconstruction / cryo EM / Resolution: 4.0 Å | |||||||||
 Authors Authors | Gremer L / Schoelzel D | |||||||||
 Citation Citation |  Journal: Science / Year: 2017 Journal: Science / Year: 2017Title: Fibril structure of amyloid-β(1-42) by cryo-electron microscopy. Authors: Lothar Gremer / Daniel Schölzel / Carla Schenk / Elke Reinartz / Jörg Labahn / Raimond B G Ravelli / Markus Tusche / Carmen Lopez-Iglesias / Wolfgang Hoyer / Henrike Heise / Dieter ...Authors: Lothar Gremer / Daniel Schölzel / Carla Schenk / Elke Reinartz / Jörg Labahn / Raimond B G Ravelli / Markus Tusche / Carmen Lopez-Iglesias / Wolfgang Hoyer / Henrike Heise / Dieter Willbold / Gunnar F Schröder /   Abstract: Amyloids are implicated in neurodegenerative diseases. Fibrillar aggregates of the amyloid-β protein (Aβ) are the main component of the senile plaques found in brains of Alzheimer's disease ...Amyloids are implicated in neurodegenerative diseases. Fibrillar aggregates of the amyloid-β protein (Aβ) are the main component of the senile plaques found in brains of Alzheimer's disease patients. We present the structure of an Aβ(1-42) fibril composed of two intertwined protofilaments determined by cryo-electron microscopy (cryo-EM) to 4.0-angstrom resolution, complemented by solid-state nuclear magnetic resonance experiments. The backbone of all 42 residues and nearly all side chains are well resolved in the EM density map, including the entire N terminus, which is part of the cross-β structure resulting in an overall "LS"-shaped topology of individual subunits. The dimer interface protects the hydrophobic C termini from the solvent. The characteristic staggering of the nonplanar subunits results in markedly different fibril ends, termed "groove" and "ridge," leading to different binding pathways on both fibril ends, which has implications for fibril growth. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_3851.map.gz emd_3851.map.gz | 36.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-3851-v30.xml emd-3851-v30.xml emd-3851.xml emd-3851.xml | 15.9 KB 15.9 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_3851_fsc.xml emd_3851_fsc.xml | 7.7 KB | Display |  FSC data file FSC data file |
| Images |  emd_3851.png emd_3851.png | 147.8 KB | ||
| Filedesc metadata |  emd-3851.cif.gz emd-3851.cif.gz | 5.5 KB | ||
| Others |  emd_3851_half_map_1.map.gz emd_3851_half_map_1.map.gz emd_3851_half_map_2.map.gz emd_3851_half_map_2.map.gz | 13.5 MB 13.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-3851 http://ftp.pdbj.org/pub/emdb/structures/EMD-3851 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-3851 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-3851 | HTTPS FTP |
-Related structure data
| Related structure data |  5oqvMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_3851.map.gz / Format: CCP4 / Size: 38.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_3851.map.gz / Format: CCP4 / Size: 38.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | The density map was sharpened by a B-factor of -50 Ang^2 and filtered to 3.5 Ang. This density map was used for model building and refinement. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.935 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Half map: even half map.
| File | emd_3851_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | even half map. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: odd half map.
| File | emd_3851_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | odd half map. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Beta-amyloid protein 42 fibrils
| Entire | Name: Beta-amyloid protein 42 fibrils |
|---|---|
| Components |
|
-Supramolecule #1: Beta-amyloid protein 42 fibrils
| Supramolecule | Name: Beta-amyloid protein 42 fibrils / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Amyloid beta A4 protein
| Macromolecule | Name: Amyloid beta A4 protein / type: protein_or_peptide / ID: 1 / Number of copies: 9 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 4.520087 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: DAEFRHDSGY EVHHQKLVFF AEDVGSNKGA IIGLMVGGVV IA UniProtKB: Amyloid-beta precursor protein |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | helical reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Buffer | pH: 2 Component:
Details: in water | ||||||
|---|---|---|---|---|---|---|---|
| Grid | Model: UltrAuFoil R 1.2/1.3 Quantifoil / Material: GOLD / Mesh: 300 / Pretreatment - Type: GLOW DISCHARGE | ||||||
| Vitrification | Cryogen name: ETHANE / Instrument: FEI VITROBOT MARK IV Details: 2.5 microL sample was applied to the grid, blotted for 2.5 s before plunging.. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI ARCTICA |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Detector mode: INTEGRATING / Number grids imaged: 1 / Number real images: 2026 / Average exposure time: 2.0 sec. / Average electron dose: 24.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal magnification: 110000 |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
- Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: AB INITIO MODEL / Target criteria: Cross-correlation coefficient |
|---|---|
| Output model |  PDB-5oqv: |
 Movie
Movie Controller
Controller


























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)