登録情報 データベース : PDB / ID : 2wo1タイトル Crystal Structure of the EphA4 Ligand Binding Domain EPHRIN TYPE-A RECEPTOR キーワード / / / / / 機能・相同性 分子機能 ドメイン・相同性 構成要素
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 Homo sapiens (ヒト)手法 / / / 解像度 : 1.85 Å データ登録者 Bowden, T.A. / Aricescu, A.R. / Nettleship, J.E. / Siebold, C. / Rahman-Huq, N. / Owens, R.J. / Stuart, D.I. / Jones, E.Y. ジャーナル : Structure / 年 : 2009タイトル : Structural Plasticity of Eph-Receptor A4 Facilitates Cross-Class Ephrin Signalling著者 : Bowden, T.A. / Aricescu, A.R. / Nettleship, J.E. / Siebold, C. / Rahman-Huq, N. / Owens, R.J. / Stuart, D.I. / Jones, E.Y. 履歴 登録 2009年7月21日 登録サイト / 処理サイト 改定 1.0 2009年10月27日 Provider / タイプ 改定 1.1 2011年7月13日 Group / Version format compliance改定 1.2 2011年11月23日 Group 改定 1.3 2018年2月28日 Group / カテゴリ Item _entity_src_gen.gene_src_common_name / _entity_src_gen.host_org_common_name ... _entity_src_gen.gene_src_common_name / _entity_src_gen.host_org_common_name / _entity_src_gen.pdbx_gene_src_scientific_name / _entity_src_gen.pdbx_host_org_cell_line / _entity_src_gen.pdbx_host_org_scientific_name / _entity_src_gen.pdbx_host_org_strain 改定 1.4 2023年12月20日 Group Data collection / Database references ... Data collection / Database references / Derived calculations / Other / Refinement description カテゴリ chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / pdbx_database_status / pdbx_initial_refinement_model / struct_site Item _database_2.pdbx_DOI / _database_2.pdbx_database_accession ... _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _pdbx_database_status.status_code_sf / _struct_site.pdbx_auth_asym_id / _struct_site.pdbx_auth_comp_id / _struct_site.pdbx_auth_seq_id
すべて表示 表示を減らす Remark 700 SHEET THE SHEET STRUCTURE OF THIS MOLECULE IS BIFURCATED. IN ORDER TO REPRESENT THIS FEATURE IN ... SHEET THE SHEET STRUCTURE OF THIS MOLECULE IS BIFURCATED. IN ORDER TO REPRESENT THIS FEATURE IN THE SHEET RECORDS BELOW, TWO SHEETS ARE DEFINED.
 データを開く
データを開く 基本情報
基本情報 要素
要素 キーワード
キーワード 機能・相同性情報
機能・相同性情報 Homo sapiens (ヒト)
Homo sapiens (ヒト) X線回折 /
X線回折 /  シンクロトロン /
シンクロトロン /  分子置換 / 解像度: 1.85 Å
分子置換 / 解像度: 1.85 Å  データ登録者
データ登録者 引用
引用 ジャーナル: Structure / 年: 2009
ジャーナル: Structure / 年: 2009 構造の表示
構造の表示 Molmil
Molmil Jmol/JSmol
Jmol/JSmol ダウンロードとリンク
ダウンロードとリンク ダウンロード
ダウンロード 2wo1.cif.gz
2wo1.cif.gz PDBx/mmCIF形式
PDBx/mmCIF形式 pdb2wo1.ent.gz
pdb2wo1.ent.gz PDB形式
PDB形式 2wo1.json.gz
2wo1.json.gz PDBx/mmJSON形式
PDBx/mmJSON形式 その他のダウンロード
その他のダウンロード 2wo1_validation.pdf.gz
2wo1_validation.pdf.gz wwPDB検証レポート
wwPDB検証レポート 2wo1_full_validation.pdf.gz
2wo1_full_validation.pdf.gz 2wo1_validation.xml.gz
2wo1_validation.xml.gz 2wo1_validation.cif.gz
2wo1_validation.cif.gz https://data.pdbj.org/pub/pdb/validation_reports/wo/2wo1
https://data.pdbj.org/pub/pdb/validation_reports/wo/2wo1 ftp://data.pdbj.org/pub/pdb/validation_reports/wo/2wo1
ftp://data.pdbj.org/pub/pdb/validation_reports/wo/2wo1 リンク
リンク 集合体
集合体
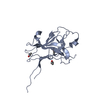

 要素
要素 Homo sapiens (ヒト) / プラスミド: PHLSEC / 細胞株 (発現宿主): HEK293T / 発現宿主:
Homo sapiens (ヒト) / プラスミド: PHLSEC / 細胞株 (発現宿主): HEK293T / 発現宿主:  Homo sapiens (ヒト) / 参照: UniProt: P54764, receptor protein-tyrosine kinase
Homo sapiens (ヒト) / 参照: UniProt: P54764, receptor protein-tyrosine kinase X線回折 / 使用した結晶の数: 1
X線回折 / 使用した結晶の数: 1  試料調製
試料調製 シンクロトロン / サイト:
シンクロトロン / サイト:  ESRF
ESRF  / ビームライン: BM14 / 波長: 0.977
/ ビームライン: BM14 / 波長: 0.977  解析
解析 分子置換
分子置換 ムービー
ムービー コントローラー
コントローラー
















 PDBj
PDBj