+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: SASBDB / ID: SASDCW3 |
|---|---|
 試料 試料 | Proline utilization A from Legionella pneumophila 8 mg/mL
|
| 機能・相同性 |  機能・相同性情報 機能・相同性情報proline dehydrogenase / proline dehydrogenase activity / L-glutamate gamma-semialdehyde dehydrogenase / L-glutamate gamma-semialdehyde dehydrogenase activity / L-proline catabolic process to L-glutamate / : / cytoplasmic side of plasma membrane / DNA-binding transcription factor activity / DNA binding 類似検索 - 分子機能 |
| 生物種 |  Legionella pneumophila subsp. pneumophila (strain Philadelphia 1 / ATCC 33152 / DSM 7513) (バクテリア) Legionella pneumophila subsp. pneumophila (strain Philadelphia 1 / ATCC 33152 / DSM 7513) (バクテリア) |
 引用 引用 |  ジャーナル: FEBS J / 年: 2017 ジャーナル: FEBS J / 年: 2017タイトル: Biophysical investigation of type A PutAs reveals a conserved core oligomeric structure. 著者: David A Korasick / Harkewal Singh / Travis A Pemberton / Min Luo / Richa Dhatwalia / John J Tanner /  要旨: Many enzymes form homooligomers, yet the functional significance of self-association is seldom obvious. Herein, we examine the connection between oligomerization and catalytic function for proline ...Many enzymes form homooligomers, yet the functional significance of self-association is seldom obvious. Herein, we examine the connection between oligomerization and catalytic function for proline utilization A (PutA) enzymes. PutAs are bifunctional enzymes that catalyze both reactions of proline catabolism. Type A PutAs are the smallest members of the family, possessing a minimal domain architecture consisting of N-terminal proline dehydrogenase and C-terminal l-glutamate-γ-semialdehyde dehydrogenase modules. Type A PutAs form domain-swapped dimers, and in one case (Bradyrhizobium japonicum PutA), two of the dimers assemble into a ring-shaped tetramer. Whereas the dimer has a clear role in substrate channeling, the functional significance of the tetramer is unknown. To address this question, we performed structural studies of four-type A PutAs from two clades of the PutA tree. The crystal structure of Bdellovibrio bacteriovorus PutA covalently inactivated by N-propargylglycine revealed a fold and substrate-channeling tunnel similar to other PutAs. Small-angle X-ray scattering (SAXS) and analytical ultracentrifugation indicated that Bdellovibrio PutA is dimeric in solution, in contrast to the prediction from crystal packing of a stable tetrameric assembly. SAXS studies of two other type A PutAs from separate clades also suggested that the dimer predominates in solution. To assess whether the tetramer of B. japonicum PutA is necessary for catalytic function, a hot spot disruption mutant that cleanly produces dimeric protein was generated. The dimeric variant exhibited kinetic parameters similar to the wild-type enzyme. These results implicate the domain-swapped dimer as the core structural and functional unit of type A PutAs. ENZYMES: Proline dehydrogenase (EC 1.5.5.2); l-glutamate-γ-semialdehyde dehydrogenase (EC 1.2.1.88). DATABASES: The atomic coordinates and structure factor amplitudes have been deposited in the Protein Data Bank under accession number 5UR2. The SAXS data have been deposited in the SASBDB under the ...DATABASES: The atomic coordinates and structure factor amplitudes have been deposited in the Protein Data Bank under accession number 5UR2. The SAXS data have been deposited in the SASBDB under the following accession codes: SASDCP3 (BbPutA), SASDCQ3 (DvPutA 1.5 mg·mL ), SASDCX3 (DvPutA 3.0 mg·mL ), SASDCY3 (DvPutA 4.5 mg·mL ), SASDCR3 (LpPutA 3.0 mg·mL ), SASDCV3 (LpPutA 5.0 mg·mL ), SASDCW3 (LpPutA 8.0 mg·mL ), SASDCS3 (BjPutA 2.3 mg·mL ), SASDCT3 (BjPutA 4.7 mg·mL ), SASDCU3 (BjPutA 7.0 mg·mL ), SASDCZ3 (R51E 2.3 mg·mL ), SASDC24 (R51E 4.7 mg·mL ), SASDC34 (R51E 7.0 mg·mL ). |
 登録者 登録者 |
|
- 構造の表示
構造の表示
| 構造ビューア | 分子:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-モデル
| モデル #1427 |  タイプ: dummy / ダミー原子の半径: 3.30 A / 対称性: P2 / カイ2乗値: 4.222  Omokage検索でこの集合体の類似形状データを探す (詳細) Omokage検索でこの集合体の類似形状データを探す (詳細) |
|---|---|
| モデル #1429 |  タイプ: dummy / ソフトウェア: (5.0 r6293) / ダミー原子の半径: 5.00 A / カイ2乗値: 4.222  Omokage検索でこの集合体の類似形状データを探す (詳細) Omokage検索でこの集合体の類似形状データを探す (詳細) |
- 試料
試料
 試料 試料 | 名称: Proline utilization A from Legionella pneumophila 8 mg/mL 試料濃度: 8 mg/ml |
|---|---|
| バッファ | 名称: 50 mM Tris-HCl, 50 mM NaCl, 0.5 mM EDTA, and 0.5 mM THP at pH 7.5. pH: 7.5 |
| 要素 #680 | 名称: LpPutA / タイプ: protein / 記述: Bifunctional protein PutA / 分子量: 119.178 / 分子数: 2 由来: Legionella pneumophila subsp. pneumophila (strain Philadelphia 1 / ATCC 33152 / DSM 7513) 参照: UniProt: Q5ZUU6 配列: HHHHHHGKPI PNPLLGLDST ENLYFQGMLE KQSIHLPEGL RAAINKAYRM DELSLITELS EQAALDPQQM MAIKTSATKL VQSVRSERKK STGIDSFLTE YALSSDEGIA LMCLAEALLR VPDNATIDNL IKDKLAGGDW GAHRGQSESF FVNATTWALM LTGKVLTPEK ...配列: HHHHHHGKPI PNPLLGLDST ENLYFQGMLE KQSIHLPEGL RAAINKAYRM DELSLITELS EQAALDPQQM MAIKTSATKL VQSVRSERKK STGIDSFLTE YALSSDEGIA LMCLAEALLR VPDNATIDNL IKDKLAGGDW GAHRGQSESF FVNATTWALM LTGKVLTPEK AENTLTKALL KLVNRSSEAV VRKAVDKAMR IMSKQFVMGR TINEALARAK KKEDRGYRYS YDMLGEAALT SADAARYFEA YKEAIISIGE KADKHSDVYR RPGISIKLSA LHPRYSEFQY ERVMAELPPK LLALSRLAKD YGIALTIDAE ESERLDLSLD VIEKVFTDES LQGWNGFGLA VQSYQKRAFY VLDWVAALAR SKQRRIMVRL IKGAYWDSEI KKTQMQGFSE YPVFTRKVFT DVSFQACAKK ILTMTDAIYP QFATHNAYSV AMILNLVGGY RDFEFQCLHG MGNELYEQIV PANCYGIPCR IYAPVGSHED LLPYLVRRLL ENGANSSFVN RIVDDKAPIS ELVEDPVAKS RSLLDKINKN IPLPEDIFLP VRKNSKGFDF TNRLERALLQ QELAKIESKE WQASPMIAGR KLSRDLLQTV MSPQQPAYAI GSVQQATLDD VEVALNQAKL AFELWSKKPV EERASCLNRF ADLLQANMAE LMVLTCREAG KTWSDGIAEV REAIDFCRYY AKKAQELMSS PQRFNGYTGE LNELSLHPRG TILCISPWNF PLAIFTGQVV AGLVTGNCVI AKPAEQTPLI AAYAVKLMHQ AGIPEGVIQL IPGAGETIGA ALVADKRIKA VLFTGSTDTA NLINRTLATR GGEIIPLIAE TGGQNAMIVD SSALLEQVVV DAVTSAFGSA GQRCSALRVL YVQEEVYPRT VELLKGAMAE LVVGDPQWLS TDVGPVIDKE ALSILKNHVE NMRKHHEILY QCTVDDEALS GYFMPPTAIA IDSISALEKE VFGPILHVIQ FKRKDLDKVI NQINQTSYGL TLGIHSRINE TVDYIRQRVH AGNCYVNRNM IGAVVGLQPF GGEGLSGTGP KAGGPNYLIR LCHERTYTVD TTAAGGNASL MSIPEEG |
-実験情報
| ビーム | 設備名称: Advanced Light Source (ALS) 12.3.1 (SIBYLS) / 地域: Berkeley, CA / 国: USA  / 線源: X-ray synchrotron / 線源: X-ray synchrotron | |||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 検出器 | 名称: MAR 165 CCD | |||||||||||||||||||||||||||||||||
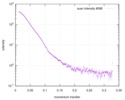
| スキャン | 測定日: 2010年4月20日 / 単位: 1/A /
| |||||||||||||||||||||||||||||||||
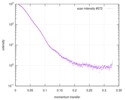
| 距離分布関数 P(R) |
| |||||||||||||||||||||||||||||||||
| 結果 |
|
 ムービー
ムービー コントローラー
コントローラー


 SASDCW3
SASDCW3