[English] 日本語
 Yorodumi
Yorodumi- PDB-3gv6: Crystal Structure of human chromobox homolog 6 (CBX6) with H3K9 p... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3gv6 | ||||||
|---|---|---|---|---|---|---|---|
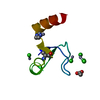
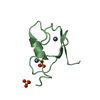
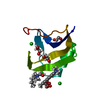
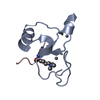
| Title | Crystal Structure of human chromobox homolog 6 (CBX6) with H3K9 peptide | ||||||
 Components Components |
| ||||||
 Keywords Keywords | Transcription/DNA binding protein / chromobox homolog / CBX6 / H3K9 peptide / SGC / Chromatin regulator / Nucleus / Phosphoprotein / Repressor / Transcription / Transcription regulation / Chromosomal protein / DNA-binding / Nucleosome core / Transcription-DNA binding protein COMPLEX / Structural Genomics / Structural Genomics Consortium | ||||||
| Function / homology |  Function and homology information Function and homology informationPRC1 complex / PcG protein complex / RUNX1 interacts with co-factors whose precise effect on RUNX1 targets is not known / heterochromatin / Regulation of PTEN gene transcription / structural constituent of chromatin / nucleosome / chromatin organization / Oxidative Stress Induced Senescence / single-stranded RNA binding ...PRC1 complex / PcG protein complex / RUNX1 interacts with co-factors whose precise effect on RUNX1 targets is not known / heterochromatin / Regulation of PTEN gene transcription / structural constituent of chromatin / nucleosome / chromatin organization / Oxidative Stress Induced Senescence / single-stranded RNA binding / nuclear body / protein heterodimerization activity / chromatin binding / chromatin / negative regulation of transcription by RNA polymerase II / DNA binding / nucleoplasm / nucleus Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 1.76 Å MOLECULAR REPLACEMENT / Resolution: 1.76 Å | ||||||
 Authors Authors | Dong, A. / Amaya, M.F. / Li, Z. / Loppnau, P. / Kozieradzki, I. / Edwards, A.M. / Arrowsmith, C.H. / Weigelt, J. / Bountra, C. / Bochkarev, A. ...Dong, A. / Amaya, M.F. / Li, Z. / Loppnau, P. / Kozieradzki, I. / Edwards, A.M. / Arrowsmith, C.H. / Weigelt, J. / Bountra, C. / Bochkarev, A. / Min, J. / Ouyang, H. / Structural Genomics Consortium (SGC) | ||||||
 Citation Citation |  Journal: J.Biol.Chem. / Year: 2011 Journal: J.Biol.Chem. / Year: 2011Title: Recognition and specificity determinants of the human cbx chromodomains. Authors: Kaustov, L. / Ouyang, H. / Amaya, M. / Lemak, A. / Nady, N. / Duan, S. / Wasney, G.A. / Li, Z. / Vedadi, M. / Schapira, M. / Min, J. / Arrowsmith, C.H. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3gv6.cif.gz 3gv6.cif.gz | 27.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3gv6.ent.gz pdb3gv6.ent.gz | 16.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3gv6.json.gz 3gv6.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/gv/3gv6 https://data.pdbj.org/pub/pdb/validation_reports/gv/3gv6 ftp://data.pdbj.org/pub/pdb/validation_reports/gv/3gv6 ftp://data.pdbj.org/pub/pdb/validation_reports/gv/3gv6 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  2l11C  2l12C  2l1bC  3fdtC  3h91C  3i90C  3i91C  3dm1S S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 7137.208 Da / Num. of mol.: 1 / Fragment: UNP residues 8-65 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: CBX6 / Plasmid: P28A-LIC / Production host: Homo sapiens (human) / Gene: CBX6 / Plasmid: P28A-LIC / Production host:  |
|---|---|
| #2: Protein/peptide | Mass: 1607.877 Da / Num. of mol.: 1 / Fragment: UNP residues 2-16 / Source method: obtained synthetically / Details: This sequence occurs naturally in Xenopus laevis / Source: (synth.) |
| #3: Water | ChemComp-HOH / |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 1.73 Å3/Da / Density % sol: 29.03 % |
|---|---|
| Crystal grow | Temperature: 297 K / Method: vapor diffusion, sitting drop / pH: 7.5 Details: 14% PEG 3350, 0.2M MgCl2, 0.1M HEPES, pH7.5 , VAPOR DIFFUSION, SITTING DROP, temperature 297K |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU FR-E+ SUPERBRIGHT / Wavelength: 1.54 Å ROTATING ANODE / Type: RIGAKU FR-E+ SUPERBRIGHT / Wavelength: 1.54 Å |
| Detector | Type: RIGAKU RAXIS IV++ / Detector: IMAGE PLATE / Date: Mar 3, 2009 / Details: VeriMax HR |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.54 Å / Relative weight: 1 |
| Reflection | Resolution: 1.76→50 Å / Num. all: 6339 / Num. obs: 6339 / % possible obs: 98.8 % / Observed criterion σ(F): 0 / Observed criterion σ(I): 0 / Redundancy: 6 % / Biso Wilson estimate: 28.2 Å2 / Rmerge(I) obs: 0.08 / Rsym value: 0.08 / Net I/σ(I): 10.2 |
| Reflection shell | Resolution: 1.76→1.82 Å / Redundancy: 4.1 % / Rmerge(I) obs: 0.473 / Mean I/σ(I) obs: 1.66 / Num. unique all: 547 / Rsym value: 0.473 / % possible all: 89.2 |
- Processing
Processing
| Software | Name: REFMAC / Version: 5.2.0019 / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB entry 3DM1 Resolution: 1.76→24.85 Å / Cor.coef. Fo:Fc: 0.953 / Cor.coef. Fo:Fc free: 0.94 / SU B: 3.142 / SU ML: 0.1 / Cross valid method: THROUGHOUT / σ(F): 0 / σ(I): 0 / ESU R: 0.153 / ESU R Free: 0.151 / Stereochemistry target values: MAXIMUM LIKELIHOOD / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.4 Å / Solvent model: MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 26.219 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.76→24.85 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 1.76→1.807 Å / Total num. of bins used: 20
|
 Movie
Movie Controller
Controller








 PDBj
PDBj




