+ Open data
Open data
- Basic information
Basic information
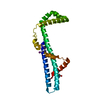
| Entry | Database: PDB / ID: 2ak5 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | beta PIX-SH3 complexed with a Cbl-b peptide | |||||||||
 Components Components |
| |||||||||
 Keywords Keywords | ENDOCYTOSIS/EXOCYTOSIS / adaptor proteins / Cin85 / PIX/COOL / Cbl / protein-protein interaction / endocytosis / ENDOCYTOSIS-EXOCYTOSIS COMPLEX | |||||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of microtubule nucleation / Ephrin signaling / presynaptic actin cytoskeleton organization / EGFR downregulation / RHOU GTPase cycle / RHOV GTPase cycle / NRAGE signals death through JNK / G alpha (12/13) signalling events / RHOQ GTPase cycle / RAC1 GTPase cycle ...negative regulation of microtubule nucleation / Ephrin signaling / presynaptic actin cytoskeleton organization / EGFR downregulation / RHOU GTPase cycle / RHOV GTPase cycle / NRAGE signals death through JNK / G alpha (12/13) signalling events / RHOQ GTPase cycle / RAC1 GTPase cycle / postsynaptic actin cytoskeleton organization / regulation of platelet-derived growth factor receptor-alpha signaling pathway / RHOA GTPase cycle / storage vacuole / astrocyte cell migration / T cell anergy / positive regulation of growth hormone secretion / regulation protein catabolic process at postsynapse / positive regulation of T cell anergy / CD4-positive, alpha-beta T cell proliferation / negative regulation of CD4-positive, alpha-beta T cell proliferation / gamma-tubulin binding / ATP-dependent protein binding / lamellipodium assembly / NLS-bearing protein import into nucleus / small GTPase-mediated signal transduction / regulation of postsynaptic neurotransmitter receptor internalization / mitotic spindle pole / negative regulation of T cell activation / Golgi organization / negative regulation of epidermal growth factor receptor signaling pathway / negative regulation of T cell receptor signaling pathway / protein K63-linked ubiquitination / hematopoietic progenitor cell differentiation / Rho protein signal transduction / ruffle / phosphotyrosine residue binding / guanyl-nucleotide exchange factor activity / positive regulation of protein ubiquitination / protein catabolic process / RING-type E3 ubiquitin transferase / receptor tyrosine kinase binding / GABA-ergic synapse / positive regulation of protein catabolic process / ubiquitin protein ligase activity / Antigen processing: Ubiquitination & Proteasome degradation / T cell receptor signaling pathway / lamellipodium / growth cone / cell cortex / neuron projection / intracellular signal transduction / postsynapse / protein stabilization / immune response / positive regulation of apoptotic process / membrane raft / focal adhesion / neuronal cell body / calcium ion binding / centrosome / protein kinase binding / glutamatergic synapse / signal transduction / protein-containing complex / zinc ion binding / nucleoplasm / plasma membrane / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.85 Å MOLECULAR REPLACEMENT / Resolution: 1.85 Å | |||||||||
 Authors Authors | Jozic, D. / Cardenes, N. / Deribe, Y.L. / Moncalian, G. / Hoeller, D. / Groemping, Y. / Dikic, I. / Rittinger, K. / Bravo, J. | |||||||||
 Citation Citation |  Journal: Nat.Struct.Mol.Biol. / Year: 2005 Journal: Nat.Struct.Mol.Biol. / Year: 2005Title: Cbl promotes clustering of endocytic adaptor proteins. Authors: Jozic, D. / Cardenes, N. / Deribe, Y.L. / Moncalian, G. / Hoeller, D. / Groemping, Y. / Dikic, I. / Rittinger, K. / Bravo, J. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2ak5.cif.gz 2ak5.cif.gz | 41.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2ak5.ent.gz pdb2ak5.ent.gz | 29 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2ak5.json.gz 2ak5.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ak/2ak5 https://data.pdbj.org/pub/pdb/validation_reports/ak/2ak5 ftp://data.pdbj.org/pub/pdb/validation_reports/ak/2ak5 ftp://data.pdbj.org/pub/pdb/validation_reports/ak/2ak5 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  2bz8C  1semS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 7290.925 Da / Num. of mol.: 2 / Fragment: beta-pix SH3A Source method: isolated from a genetically manipulated source Source: (gene. exp.)   #2: Protein/peptide | | Mass: 1007.236 Da / Num. of mol.: 1 / Fragment: residues 904-911 / Source method: obtained synthetically Details: The peptide was chemically synthesized. The sequence of the peptide can be naturally found in Homo sapiens (Human)Cbl-b References: UniProt: Q13191 #3: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 2 X-RAY DIFFRACTION / Number of used crystals: 2 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.59 Å3/Da / Density % sol: 52.43 % |
|---|---|
| Crystal grow | Temperature: 291 K / pH: 6.5 Details: PEG 3000, SODIUM CITRATE, pH 6.5, VAPOR DIFFUSION, SITTING DROP, temperature 291K, pH 6.50 |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SRS SRS  / Beamline: PX14.2 / Wavelength: 1.5418 / Beamline: PX14.2 / Wavelength: 1.5418 |
| Detector | Type: ADSC QUANTUM 4 / Detector: CCD / Details: MIRRORS |
| Radiation | Monochromator: SI 111 / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 1.85→26.17 Å / Num. obs: 14106 / % possible obs: 99.5 % / Redundancy: 2.94 % / Rsym value: 0.086 / Net I/σ(I): 31.8 |
| Reflection shell | Resolution: 1.85→1.93 Å / % possible all: 98.1 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: pdb entry 1SEM Resolution: 1.85→26.17 Å / Cor.coef. Fo:Fc: 0.948 / Cor.coef. Fo:Fc free: 0.9263 / SU B: 9.0032 / SU ML: 0.1122 / Cross valid method: THROUGHOUT / ESU R: 0.3046 / ESU R Free: 0.1528
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.4 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.85→26.17 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj




