+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-23936 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the human SSU processome, state pre-A1 | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Ribosomal assembly intermediate / RIBOSOME | |||||||||
| Function / homology |  Function and homology information Function and homology informationmRNA cytidine N-acetyltransferase activity / U6 snRNA 2'-O-ribose methyltransferase activity / nucleologenesis / oocyte growth / snoRNA localization / granular component / leucine zipper domain binding / tRNA wobble cytosine modification / U4atac snRNP / tRNA cytidine N4-acetyltransferase activity ...mRNA cytidine N-acetyltransferase activity / U6 snRNA 2'-O-ribose methyltransferase activity / nucleologenesis / oocyte growth / snoRNA localization / granular component / leucine zipper domain binding / tRNA wobble cytosine modification / U4atac snRNP / tRNA cytidine N4-acetyltransferase activity / rRNA acetylation involved in maturation of SSU-rRNA / 18S rRNA cytidine N-acetyltransferase activity / tRNA acetylation / regulation of stem cell population maintenance / U4atac snRNA binding / CURI complex / UTP-C complex / t-UTP complex / pre-snoRNP complex / Pwp2p-containing subcomplex of 90S preribosome / Mpp10 complex / rRNA (pseudouridine) methyltransferase activity / negative regulation of amyloid precursor protein biosynthetic process / box C/D sno(s)RNA binding / rRNA modification / histone H2AQ104 methyltransferase activity / dense fibrillar component / box C/D sno(s)RNA 3'-end processing / histone methyltransferase binding / rRNA methyltransferase activity / preribosome / endonucleolytic cleavage of tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / endonucleolytic cleavage in 5'-ETS of tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / regulation of transcription elongation by RNA polymerase II / regulation of centrosome duplication / endonucleolytic cleavage to generate mature 5'-end of SSU-rRNA from (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / N-acetyltransferase activity / box C/D methylation guide snoRNP complex / cilium disassembly / positive regulation of rRNA processing / tRNA export from nucleus / embryonic cleavage / rRNA primary transcript binding / RNA splicing, via transesterification reactions / sno(s)RNA-containing ribonucleoprotein complex / rRNA base methylation / SUMOylation of RNA binding proteins / blastocyst formation / U2-type precatalytic spliceosome / protein localization to nucleolus / telomerase holoenzyme complex / rRNA methylation / U4 snRNA binding / transcription elongation factor activity / box C/D snoRNP assembly / spindle assembly involved in female meiosis / epigenetic programming in the zygotic pronuclei / neural precursor cell proliferation / U3 snoRNA binding / negative regulation of RNA splicing / protein acetylation / neural crest cell differentiation / NRAGE signals death through JNK / snoRNA binding / preribosome, small subunit precursor / precatalytic spliceosome / rRNA modification in the nucleus and cytosol / negative regulation of bicellular tight junction assembly / erythrocyte homeostasis / cytoplasmic side of rough endoplasmic reticulum membrane / Formation of the ternary complex, and subsequently, the 43S complex / rRNA metabolic process / Cul4-RING E3 ubiquitin ligase complex / positive regulation of transcription by RNA polymerase I / Ribosomal scanning and start codon recognition / negative regulation of telomere maintenance via telomerase / Association of TriC/CCT with target proteins during biosynthesis / decidualization / Translation initiation complex formation / RNA polymerase II complex binding / TFIID-class transcription factor complex binding / Protein hydroxylation / negative regulation of apoptotic signaling pathway / SARS-CoV-1 modulates host translation machinery / TOR signaling / mTORC1-mediated signalling / Peptide chain elongation / cellular response to ethanol / chromosome, centromeric region / Selenocysteine synthesis / Formation of a pool of free 40S subunits / positive regulation of intrinsic apoptotic signaling pathway by p53 class mediator / Eukaryotic Translation Termination / blastocyst development / 90S preribosome / SRP-dependent cotranslational protein targeting to membrane / Response of EIF2AK4 (GCN2) to amino acid deficiency / single fertilization / ubiquitin ligase inhibitor activity / Viral mRNA Translation Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.6 Å | |||||||||
 Authors Authors | Vanden Broeck A / Singh S / Klinge S | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Science / Year: 2021 Journal: Science / Year: 2021Title: Nucleolar maturation of the human small subunit processome. Authors: Sameer Singh / Arnaud Vanden Broeck / Linamarie Miller / Malik Chaker-Margot / Sebastian Klinge /  Abstract: The human small subunit processome mediates early maturation of the small ribosomal subunit by coupling RNA folding to subsequent RNA cleavage and processing steps. We report the high-resolution ...The human small subunit processome mediates early maturation of the small ribosomal subunit by coupling RNA folding to subsequent RNA cleavage and processing steps. We report the high-resolution cryo–electron microscopy structures of maturing human small subunit (SSU) processomes at resolutions of 2.7 to 3.9 angstroms. These structures reveal the molecular mechanisms that enable crucial progressions during SSU processome maturation. RNA folding states within these particles are communicated to and coordinated with key enzymes that drive irreversible steps such as targeted exosome-mediated RNA degradation, protein-guided site-specific endonucleolytic RNA cleavage, and tightly controlled RNA unwinding. These conserved mechanisms highlight the SSU processome’s impressive structural plasticity, which endows this 4.5-megadalton nucleolar assembly with the distinctive ability to mature the small ribosomal subunit from within. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_23936.map.gz emd_23936.map.gz | 615.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-23936-v30.xml emd-23936-v30.xml emd-23936.xml emd-23936.xml | 125.6 KB 125.6 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_23936_fsc.xml emd_23936_fsc.xml | 19.8 KB | Display |  FSC data file FSC data file |
| Images |  emd_23936.png emd_23936.png | 161 KB | ||
| Masks |  emd_23936_msk_1.map emd_23936_msk_1.map | 669.9 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-23936.cif.gz emd-23936.cif.gz | 31.4 KB | ||
| Others |  emd_23936_additional_1.map.gz emd_23936_additional_1.map.gz emd_23936_half_map_1.map.gz emd_23936_half_map_1.map.gz emd_23936_half_map_2.map.gz emd_23936_half_map_2.map.gz | 614.3 MB 535 MB 534.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-23936 http://ftp.pdbj.org/pub/emdb/structures/EMD-23936 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-23936 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-23936 | HTTPS FTP |
-Related structure data
| Related structure data |  7mq8MC  7mq9C  7mqaC  7mqjC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | |
| EM raw data |  EMPIAR-10781 (Title: Nucleolar maturation of the human small subunit processome EMPIAR-10781 (Title: Nucleolar maturation of the human small subunit processomeData size: 74.6 TB Data #1: Unaligned multi-frame micrograph movies of human SSU processomes - Dataset 1 [micrographs - multiframe] Data #2: Unaligned multi-frame micrograph movies of human SSU processomes - Dataset 2 [micrographs - multiframe] Data #3: Unaligned multi-frame micrograph movies of human SSU processomes - Dataset 3 [micrographs - multiframe] Data #4: Unaligned multi-frame micrograph movies of human SSU processomes - Dataset 4 [micrographs - multiframe] Data #5: Unaligned multi-frame micrograph movies of human SSU processomes - Dataset 5 [micrographs - multiframe] Data #6: Unaligned multi-frame micrograph movies of human SSU processomes - Dataset 6 [micrographs - multiframe] Data #7: Aligned and averaged micrographs of human SSU processomes - Dataset 1 [micrographs - single frame] Data #8: Aligned and averaged micrographs of human SSU processomes - Dataset 2 [micrographs - single frame] Data #9: Aligned and averaged micrographs of human SSU processomes - Dataset 3 [micrographs - single frame] Data #10: Aligned and averaged micrographs of human SSU processomes - Dataset 4 [micrographs - single frame] Data #11: Aligned and averaged micrographs of human SSU processomes - Dataset 5 [micrographs - single frame] Data #12: Aligned and averaged micrographs of human SSU processomes - Dataset 6 [micrographs - single frame]) |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_23936.map.gz / Format: CCP4 / Size: 669.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_23936.map.gz / Format: CCP4 / Size: 669.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.08 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Mask #1
| File |  emd_23936_msk_1.map emd_23936_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
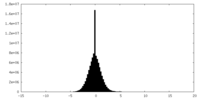
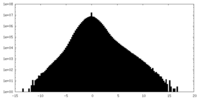
| Density Histograms |
-Additional map: #1
| File | emd_23936_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_23936_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_23936_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : Human SSU processome
+Supramolecule #1: Human SSU processome
+Macromolecule #1: 5'ETS rRNA
+Macromolecule #2: 18S rRNA
+Macromolecule #3: U3 snoRNA
+Macromolecule #31: 5'ETS rRNA
+Macromolecule #4: 40S ribosomal protein S18
+Macromolecule #5: 40S ribosomal protein S4, X isoform
+Macromolecule #6: 40S ribosomal protein S5
+Macromolecule #7: 40S ribosomal protein S6
+Macromolecule #8: 40S ribosomal protein S7
+Macromolecule #9: 40S ribosomal protein S8
+Macromolecule #10: 40S ribosomal protein S9
+Macromolecule #11: 40S ribosomal protein S12
+Macromolecule #12: 40S ribosomal protein S16
+Macromolecule #13: 40S ribosomal protein S11
+Macromolecule #14: 40S ribosomal protein S24
+Macromolecule #15: 40S ribosomal protein S28
+Macromolecule #16: WD repeat-containing protein 75
+Macromolecule #17: Nucleolar protein 11
+Macromolecule #18: U3 small nucleolar RNA-associated protein 15 homolog
+Macromolecule #19: WD repeat-containing protein 43
+Macromolecule #20: HEAT repeat-containing protein 1
+Macromolecule #21: U3 small nucleolar RNA-associated protein 4 homolog
+Macromolecule #22: Periodic tryptophan protein 2 homolog
+Macromolecule #23: U3 small nucleolar RNA-associated protein 6 homolog
+Macromolecule #24: WD repeat-containing protein 3
+Macromolecule #25: Transducin beta-like protein 3
+Macromolecule #26: U3 small nucleolar RNA-associated protein 18 homolog
+Macromolecule #27: WD repeat-containing protein 36
+Macromolecule #28: DDB1- and CUL4-associated factor 13
+Macromolecule #29: WD repeat-containing protein 46
+Macromolecule #30: U3 small nucleolar ribonucleoprotein protein IMP3
+Macromolecule #32: U3 small nucleolar ribonucleoprotein protein MPP10
+Macromolecule #33: Something about silencing protein 10
+Macromolecule #34: Neuroguidin
+Macromolecule #35: Nucleolar protein 7
+Macromolecule #36: Uncharacterized protein C1orf131
+Macromolecule #37: 40S ribosomal protein S13
+Macromolecule #38: 40S ribosomal protein S14
+Macromolecule #39: Nucleolar protein 6
+Macromolecule #40: Ribosomal RNA-processing protein 7 homolog A
+Macromolecule #41: RNA cytidine acetyltransferase
+Macromolecule #42: 40S ribosomal protein S3a
+Macromolecule #43: Protein AATF
+Macromolecule #44: 40S ribosomal protein S15a
+Macromolecule #45: 40S ribosomal protein S27
+Macromolecule #46: RRP12-like protein
+Macromolecule #47: Ubiquitin-40S ribosomal protein S27a
+Macromolecule #48: 40S ribosomal protein S17
+Macromolecule #49: Nucleolar protein 10
+Macromolecule #50: KRR1 small subunit processome component homolog
+Macromolecule #51: Nucleolar protein 56
+Macromolecule #52: Nucleolar protein 58
+Macromolecule #53: rRNA 2'-O-methyltransferase fibrillarin
+Macromolecule #54: NHP2-like protein 1
+Macromolecule #55: U3 small nucleolar RNA-interacting protein 2
+Macromolecule #56: RNA 3'-terminal phosphate cyclase-like protein
+Macromolecule #57: Ribosome biogenesis protein BMS1 homolog
+Macromolecule #58: Ribosomal RNA small subunit methyltransferase NEP1
+Macromolecule #59: rRNA-processing protein FCF1 homolog
+Macromolecule #60: U3 small nucleolar ribonucleoprotein protein IMP4
+Macromolecule #61: Small subunit processome component 20 homolog
+Macromolecule #62: Deoxynucleotidyltransferase terminal-interacting protein 2
+Macromolecule #63: 40S ribosomal protein S23
+Macromolecule #64: U3 small nucleolar RNA-associated protein 14 homolog A
+Macromolecule #65: Nucleolar protein 14
+Macromolecule #66: Nucleolar complex protein 4 homolog
+Macromolecule #67: RNA-binding protein PNO1
+Macromolecule #68: Unassigned peptide
+Macromolecule #69: Unassigned peptides
+Macromolecule #70: Probable U3 small nucleolar RNA-associated protein 11
+Macromolecule #71: Bystin
+Macromolecule #72: MAGNESIUM ION
+Macromolecule #73: ADENOSINE-5'-TRIPHOSPHATE
+Macromolecule #74: ZINC ION
+Macromolecule #75: GUANOSINE-5'-TRIPHOSPHATE
+Macromolecule #76: S-ADENOSYL-L-HOMOCYSTEINE
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.6 |
|---|---|
| Grid | Model: Quantifoil R2/2 / Material: GOLD / Mesh: 400 / Support film - Material: CARBON / Support film - topology: HOLEY / Support film - Film thickness: 3 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 30 sec. |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 90 % / Chamber temperature: 283 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Number real images: 84904 / Average electron dose: 58.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 0.01 mm / Nominal defocus max: 2.7 µm / Nominal defocus min: 0.7000000000000001 µm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model |
| ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Space: REAL | ||||||||||||||||||||
| Output model |  PDB-7mq8: |
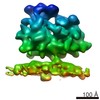
 Movie
Movie Controller
Controller







































































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)