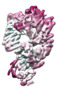
登録情報 データベース : EMDB / ID : EMD-10498タイトル CryoEM structure of the ternary DOCK2-ELMO1-RAC1 complex 複合体 : Ternary complex of DOCK2-ELMO1-RAC1タンパク質・ペプチド : Dedicator of cytokinesis protein 2タンパク質・ペプチド : Engulfment and cell motility protein 1タンパク質・ペプチド : Ras-related C3 botulinum toxin substrate 1 / / / / 機能・相同性 分子機能 ドメイン・相同性 構成要素
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 Homo sapiens (ヒト)手法 / / 解像度 : 4.1 Å Chang L / Yang J 資金援助 Organization Grant number 国 Medical Research Council (United Kingdom) MC_UP_1201/6 Cancer Research UK C576/A14109 Marie Sklodowska-Curie Actions, FragNET ITN 657725 European Union
ジャーナル : Nat Commun / 年 : 2020タイトル : Structure of the DOCK2-ELMO1 complex provides insights into regulation of the auto-inhibited state.著者 : Leifu Chang / Jing Yang / Chang Hwa Jo / Andreas Boland / Ziguo Zhang / Stephen H McLaughlin / Afnan Abu-Thuraia / Ryan C Killoran / Matthew J Smith / Jean-Francois Côté / David Barford / 要旨 : DOCK (dedicator of cytokinesis) proteins are multidomain guanine nucleotide exchange factors (GEFs) for RHO GTPases that regulate intracellular actin dynamics. DOCK proteins share catalytic (DOCK) ... DOCK (dedicator of cytokinesis) proteins are multidomain guanine nucleotide exchange factors (GEFs) for RHO GTPases that regulate intracellular actin dynamics. DOCK proteins share catalytic (DOCK) and membrane-associated (DOCK) domains. The structurally-related DOCK1 and DOCK2 GEFs are specific for RAC, and require ELMO (engulfment and cell motility) proteins for function. The N-terminal RAS-binding domain (RBD) of ELMO (ELMO) interacts with RHOG to modulate DOCK1/2 activity. Here, we determine the cryo-EM structures of DOCK2-ELMO1 alone, and as a ternary complex with RAC1, together with the crystal structure of a RHOG-ELMO2 complex. The binary DOCK2-ELMO1 complex adopts a closed, auto-inhibited conformation. Relief of auto-inhibition to an active, open state, due to a conformational change of the ELMO1 subunit, exposes binding sites for RAC1 on DOCK2, and RHOG and BAI GPCRs on ELMO1. Our structure explains how up-stream effectors, including DOCK2 and ELMO1 phosphorylation, destabilise the auto-inhibited state to promote an active GEF. 履歴 登録 2019年11月15日 - ヘッダ(付随情報) 公開 2020年7月29日 - マップ公開 2020年7月29日 - 更新 2024年7月10日 - 現状 2024年7月10日 処理サイト : PDBe / 状態 : 公開
すべて表示 表示を減らす
 データを開く
データを開く 基本情報
基本情報 マップデータ
マップデータ 試料
試料 キーワード
キーワード 機能・相同性情報
機能・相同性情報 Homo sapiens (ヒト)
Homo sapiens (ヒト) データ登録者
データ登録者 英国, European Union, 3件
英国, European Union, 3件  引用
引用 ジャーナル: Nat Commun / 年: 2020
ジャーナル: Nat Commun / 年: 2020



 構造の表示
構造の表示 ムービービューア
ムービービューア SurfView
SurfView Molmil
Molmil Jmol/JSmol
Jmol/JSmol ダウンロードとリンク
ダウンロードとリンク emd_10498.map.gz
emd_10498.map.gz EMDBマップデータ形式
EMDBマップデータ形式 emd-10498-v30.xml
emd-10498-v30.xml emd-10498.xml
emd-10498.xml EMDBヘッダ
EMDBヘッダ emd_10498.png
emd_10498.png emd-10498.cif.gz
emd-10498.cif.gz emd_10498_additional.map.gz
emd_10498_additional.map.gz http://ftp.pdbj.org/pub/emdb/structures/EMD-10498
http://ftp.pdbj.org/pub/emdb/structures/EMD-10498 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-10498
ftp://ftp.pdbj.org/pub/emdb/structures/EMD-10498 emd_10498_validation.pdf.gz
emd_10498_validation.pdf.gz EMDB検証レポート
EMDB検証レポート emd_10498_full_validation.pdf.gz
emd_10498_full_validation.pdf.gz emd_10498_validation.xml.gz
emd_10498_validation.xml.gz emd_10498_validation.cif.gz
emd_10498_validation.cif.gz https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-10498
https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-10498 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-10498
ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-10498 リンク
リンク EMDB (EBI/PDBe) /
EMDB (EBI/PDBe) /  EMDataResource
EMDataResource マップ
マップ ダウンロード / ファイル: emd_10498.map.gz / 形式: CCP4 / 大きさ: 85.5 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES)
ダウンロード / ファイル: emd_10498.map.gz / 形式: CCP4 / 大きさ: 85.5 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) 試料の構成要素
試料の構成要素 Homo sapiens (ヒト)
Homo sapiens (ヒト) Homo sapiens (ヒト)
Homo sapiens (ヒト) Trichoplusia ni (イラクサキンウワバ)
Trichoplusia ni (イラクサキンウワバ) Homo sapiens (ヒト)
Homo sapiens (ヒト) Trichoplusia ni (イラクサキンウワバ)
Trichoplusia ni (イラクサキンウワバ) Homo sapiens (ヒト)
Homo sapiens (ヒト) Trichoplusia ni (イラクサキンウワバ)
Trichoplusia ni (イラクサキンウワバ) 解析
解析 試料調製
試料調製 電子顕微鏡法
電子顕微鏡法 FIELD EMISSION GUN
FIELD EMISSION GUN
 画像解析
画像解析 ムービー
ムービー コントローラー
コントローラー

































 Z
Z Y
Y X
X