+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-10498 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
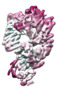
| Title | CryoEM structure of the ternary DOCK2-ELMO1-RAC1 complex | ||||||||||||
 Map data Map data | |||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | guanine nucleotide exchange factor / cytoskeleton / actin / cryoEM / SIGNALING PROTEIN | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationalpha-beta T cell proliferation / membrane raft polarization / myeloid dendritic cell activation involved in immune response / establishment of T cell polarity / embryonic olfactory bulb interneuron precursor migration / anatomical structure arrangement / regulation of ERK5 cascade / angiotensin-activated signaling pathway involved in heart process / cerebral cortex GABAergic interneuron development / regulation of respiratory burst ...alpha-beta T cell proliferation / membrane raft polarization / myeloid dendritic cell activation involved in immune response / establishment of T cell polarity / embryonic olfactory bulb interneuron precursor migration / anatomical structure arrangement / regulation of ERK5 cascade / angiotensin-activated signaling pathway involved in heart process / cerebral cortex GABAergic interneuron development / regulation of respiratory burst / positive regulation of ovarian follicle development / auditory receptor cell morphogenesis / cerebral cortex radially oriented cell migration / erythrocyte enucleation / regulation of neutrophil migration / negative regulation of interleukin-23 production / macropinocytosis / Activated NTRK2 signals through CDK5 / localization within membrane / immunological synapse formation / kinocilium / regulation of cell adhesion involved in heart morphogenesis / interneuron migration / ruffle assembly / engulfment of apoptotic cell / NTRK2 activates RAC1 / Inactivation of CDC42 and RAC1 / NADPH oxidase complex / cochlea morphogenesis / regulation of hydrogen peroxide metabolic process / regulation of neuron maturation / guanyl-nucleotide exchange factor complex / respiratory burst / WNT5:FZD7-mediated leishmania damping / SEMA3A-Plexin repulsion signaling by inhibiting Integrin adhesion / cortical cytoskeleton organization / negative thymic T cell selection / positive regulation of skeletal muscle acetylcholine-gated channel clustering / ruffle organization / midbrain dopaminergic neuron differentiation / epithelial cell morphogenesis / GTP-dependent protein binding / positive regulation of bicellular tight junction assembly / cell projection assembly / thioesterase binding / myoblast fusion / positive thymic T cell selection / regulation of lamellipodium assembly / negative regulation of fibroblast migration / regulation of neuron migration / regulation of stress fiber assembly / RHO GTPases activate CIT / cell-cell junction organization / motor neuron axon guidance / Nef and signal transduction / hepatocyte growth factor receptor signaling pathway / PCP/CE pathway / Activation of RAC1 / sphingosine-1-phosphate receptor signaling pathway / RHO GTPases activate KTN1 / regulation of nitric oxide biosynthetic process / DCC mediated attractive signaling / regulation of small GTPase mediated signal transduction / quinolinate biosynthetic process / MET activates RAP1 and RAC1 / Azathioprine ADME / Sema4D mediated inhibition of cell attachment and migration / hyperosmotic response / CD28 dependent Vav1 pathway / Ephrin signaling / positive regulation of neutrophil chemotaxis / Wnt signaling pathway, planar cell polarity pathway / superoxide anion generation / regulation of receptor signaling pathway via JAK-STAT / lamellipodium assembly / phagocytosis, engulfment / NRAGE signals death through JNK / positive regulation of ruffle assembly / dendrite morphogenesis / small GTPase-mediated signal transduction / Rho GDP-dissociation inhibitor binding / Activation of RAC1 downstream of NMDARs / positive regulation of Rho protein signal transduction / positive regulation of dendritic spine development / synaptic transmission, GABAergic / pericentriolar material / positive regulation of actin filament polymerization / establishment or maintenance of cell polarity / semaphorin-plexin signaling pathway / Rac protein signal transduction / RHO GTPases activate PAKs / RHOG GTPase cycle / Sema3A PAK dependent Axon repulsion / ficolin-1-rich granule membrane / EPH-ephrin mediated repulsion of cells / regulation of postsynapse assembly / RHOA GTPase cycle / RAC2 GTPase cycle / positive regulation of focal adhesion assembly / RHO GTPases Activate NADPH Oxidases Similarity search - Function | ||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 4.1 Å | ||||||||||||
 Authors Authors | Chang L / Yang J | ||||||||||||
| Funding support |  United Kingdom, European Union, 3 items United Kingdom, European Union, 3 items
| ||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2020 Journal: Nat Commun / Year: 2020Title: Structure of the DOCK2-ELMO1 complex provides insights into regulation of the auto-inhibited state. Authors: Leifu Chang / Jing Yang / Chang Hwa Jo / Andreas Boland / Ziguo Zhang / Stephen H McLaughlin / Afnan Abu-Thuraia / Ryan C Killoran / Matthew J Smith / Jean-Francois Côté / David Barford /     Abstract: DOCK (dedicator of cytokinesis) proteins are multidomain guanine nucleotide exchange factors (GEFs) for RHO GTPases that regulate intracellular actin dynamics. DOCK proteins share catalytic (DOCK) ...DOCK (dedicator of cytokinesis) proteins are multidomain guanine nucleotide exchange factors (GEFs) for RHO GTPases that regulate intracellular actin dynamics. DOCK proteins share catalytic (DOCK) and membrane-associated (DOCK) domains. The structurally-related DOCK1 and DOCK2 GEFs are specific for RAC, and require ELMO (engulfment and cell motility) proteins for function. The N-terminal RAS-binding domain (RBD) of ELMO (ELMO) interacts with RHOG to modulate DOCK1/2 activity. Here, we determine the cryo-EM structures of DOCK2-ELMO1 alone, and as a ternary complex with RAC1, together with the crystal structure of a RHOG-ELMO2 complex. The binary DOCK2-ELMO1 complex adopts a closed, auto-inhibited conformation. Relief of auto-inhibition to an active, open state, due to a conformational change of the ELMO1 subunit, exposes binding sites for RAC1 on DOCK2, and RHOG and BAI GPCRs on ELMO1. Our structure explains how up-stream effectors, including DOCK2 and ELMO1 phosphorylation, destabilise the auto-inhibited state to promote an active GEF. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_10498.map.gz emd_10498.map.gz | 49.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-10498-v30.xml emd-10498-v30.xml emd-10498.xml emd-10498.xml | 20.6 KB 20.6 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_10498.png emd_10498.png | 106.7 KB | ||
| Filedesc metadata |  emd-10498.cif.gz emd-10498.cif.gz | 7.4 KB | ||
| Others |  emd_10498_additional.map.gz emd_10498_additional.map.gz | 4.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-10498 http://ftp.pdbj.org/pub/emdb/structures/EMD-10498 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-10498 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-10498 | HTTPS FTP |
-Related structure data
| Related structure data |  6tgcMC  6tgbC  6ukaC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_10498.map.gz / Format: CCP4 / Size: 85.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_10498.map.gz / Format: CCP4 / Size: 85.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.43 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Additional map: #1
| File | emd_10498_additional.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Ternary complex of DOCK2-ELMO1-RAC1
| Entire | Name: Ternary complex of DOCK2-ELMO1-RAC1 |
|---|---|
| Components |
|
-Supramolecule #1: Ternary complex of DOCK2-ELMO1-RAC1
| Supramolecule | Name: Ternary complex of DOCK2-ELMO1-RAC1 / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 600 KDa |
-Macromolecule #1: Dedicator of cytokinesis protein 2
| Macromolecule | Name: Dedicator of cytokinesis protein 2 / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 195.902516 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MAPWRKADKE RHGVAIYNFQ GSGAPQLSLQ IGDVVRIQET CGDWYRGYLI KHKMLQGIFP KSFIHIKEVT VEKRRNTENI IPAEIPLAQ EVTTTLWEWG SIWKQLYVAS KKERFLQVQS MMYDLMEWRS QLLSGTLPKD ELKELKQKVT SKIDYGNKIL E LDLIVRDE ...String: MAPWRKADKE RHGVAIYNFQ GSGAPQLSLQ IGDVVRIQET CGDWYRGYLI KHKMLQGIFP KSFIHIKEVT VEKRRNTENI IPAEIPLAQ EVTTTLWEWG SIWKQLYVAS KKERFLQVQS MMYDLMEWRS QLLSGTLPKD ELKELKQKVT SKIDYGNKIL E LDLIVRDE DGNILDPDNT SVISLFHAHE EATDKITERI KEEMSKDQP(UNK) (UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)GFPE IIMPGDVRND IYITLLQGDF DKYNKTTQRN VEVIMCVCAE DGKTLP NAI CVGAGDKPMN EYRSVVYYQV KQPRWMETVK VAVPIEDMQR IHLRFMFRHR SSLESKDKGE KNFAMSYVKL MKEDGTT LH DGFHDLVVLK GDSKKMEDAS AYLTLPSYRH HVENKGATLS RSSSSVGGLS VSSRDVFSIS TLVCST(UNK)(UNK) (UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)IM MEH SQSDEY DILVFDALIY IIGLIADRKF Q(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)S SELVDFLMET FIMFKDLIGK NVYPGDWMAM SMVQNRVFLR AINKFAETMN QKFL EHTNF EFQLWNNYFH LAVAFITQDS LQLEQFSHAK YNKILNKYGD MRRLIGFSIR DMWYKLGQNK ICFIPGMVGP ILEMT LIPE AELRKATIPI FFDMMLCEYQ RSGDFKKFEN EIILKLDHEV EGGRGDEQYM QLLESILMEC AAEHPTIAKS VENFVN LVK GLLEKLLDYR GVMTDESKDN RMSCTVNLLN FYKDNNREEM YIRYLYKLRD LHLDCDNYTE AAYTLLLHTW LLKWSDE QC ASQVMQTGQQ HPQTHRQLKE TLYETIIGYF DKGKMWEEAI SLCKELAEQY EMEIFDYELL SQNLIQQAKF YESIMKIL R PKPDYFAVGY YGQGFPSFLR NKVFIYRGKE YERREDFQMQ LMTQFPNAEK MNTTSAPGDD VKNAPGQYIQ CFTVQPVLD EHPRFKNKPV PDQIINFYKS NYVQRFHYSR PVRRGTVDPE NEFASMWIER TSFVTAYKLP GILRWFEVVH MSQTTISPLE NAIETMSTA NEKILMMINQ YQSDETLPIN PLSMLLNGIV DPAVMGGFAK YEKAFFTEEY VRDHPEDQDK LTHLKDLIAW Q IPFLGAGI KIHEKRVSDN LRPFHDRMEE CFKNLKMKVE KEYGVREMPD FDDRRVGRPR SMLRSYRQMS IISLASMNSD CS TPSKPTS ESFDLELASP KTPRVEQEEP ISPGSTLPEV KLRRSKKRTK RSSVVFADEK AAAESDLKRL SRKHEFMSDT NLS EHAAIP LKASVLSQMS FASQSMPTIP ALALSVAGIP GLDEANTSPR LSQTFLQLSD GDKKTLTRKK VNQFFKTMLA SKSA EEGKQ IPDSLSTDL UniProtKB: Dedicator of cytokinesis protein 2 |
-Macromolecule #2: Engulfment and cell motility protein 1
| Macromolecule | Name: Engulfment and cell motility protein 1 / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 83.891328 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MPPPADIVKV AIEWPGAYPK LMEIDQKKPL SAIIKEVCDG WSLANHEYFA LQHADSSNFY ITEKNRNEIK NGTILRLTTS PAQNAQQLH ERIQSSSMDA KLEALKDLAS LSRDVTFAQE FINLDGISLL TQMVESGTEL YQKLQKIMKP CFGDMLSFTL T AFVELMDH ...String: MPPPADIVKV AIEWPGAYPK LMEIDQKKPL SAIIKEVCDG WSLANHEYFA LQHADSSNFY ITEKNRNEIK NGTILRLTTS PAQNAQQLH ERIQSSSMDA KLEALKDLAS LSRDVTFAQE FINLDGISLL TQMVESGTEL YQKLQKIMKP CFGDMLSFTL T AFVELMDH GIVSWDTFSV AFIKKIASFV NKSAIDISIL QRSLAILESM VLNSHDLYQK VAQEITIGQL IPHLQGSDQE IQ TYTIAVI NALFLKAPDE RRQEMANILA QKQLRSIILT HVIRAQRAIN NEMAHQLYVL QVLTFNLLED RMMTKMDPQD QAQ RDIIFE LRRIAFDAES EPNNSSGSME KRKSMYTRDY KKLGFINHVN PAMDFTQTPP GMLALDNMLY FAKHHQDAYI RIVL ENSSR EDKHECPFGR SSIELTKMLC EILKVGELPS ETCNDFHPMF FTHDRSFEEF FCICIQLLNK TWKEMRATSE DFNKV MQVV KEQVMRALTT KPSSLDQFKS KLQNLSYTEI LKIRQSERMN QEDFQSRPIL ELKEKIQPEI LELIKQQRLN RLVEGT CFR KLNARRRQDK FWYCRLSPNH KVLHYGDLEE SPQGEVPHDS LQDKLPVADI KAVVTGKDCP HMKEKGALKQ NKEVLEL AF SILYDSNCQL NFIAPDKHEY CIWTDGLNAL LGKDMMSDLT RNDLDTLLSM EIKLRLLDLE NIQIPDAPPP IPKEPSNY D FVYDCN UniProtKB: Engulfment and cell motility protein 1 |
-Macromolecule #3: Ras-related C3 botulinum toxin substrate 1
| Macromolecule | Name: Ras-related C3 botulinum toxin substrate 1 / type: protein_or_peptide / ID: 3 / Number of copies: 2 / Enantiomer: LEVO / EC number: small monomeric GTPase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 21.478113 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MQAIKCVVVG DGAVGKTCLL ISYTTNAFPG EYIPTVFDNY SANVMVDGKP VNLGLWDTAG QEDYDRLRPL SYPQTDVFLI CFSLVSPAS FENVRAKWYP EVRHHCPNTP IILVGTKLDL RDDKDTIEKL KEKKLTPITY PQGLAMAKEI GAVKYLECSA L TQRGLKTV ...String: MQAIKCVVVG DGAVGKTCLL ISYTTNAFPG EYIPTVFDNY SANVMVDGKP VNLGLWDTAG QEDYDRLRPL SYPQTDVFLI CFSLVSPAS FENVRAKWYP EVRHHCPNTP IILVGTKLDL RDDKDTIEKL KEKKLTPITY PQGLAMAKEI GAVKYLECSA L TQRGLKTV FDEAIRAVLC PPPVKKRKRK CLLL UniProtKB: Ras-related C3 botulinum toxin substrate 1 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.2 mg/mL | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 8 Component:
| |||||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 300 / Pretreatment - Type: GLOW DISCHARGE | |||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK III |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Quantum ER / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: INSILICO MODEL Details: e2initialmodel.py from the EMAN2 package was used for generation of the initial model |
|---|---|
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Resolution.type: BY AUTHOR / Resolution: 4.1 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: RELION (ver. 1.4) / Number images used: 245763 |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
 Movie
Movie Controller
Controller






























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)