+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-0452 | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
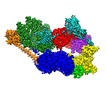
| Title | Cryo-EM structure of the human TFIIH core complex | |||||||||||||||
 Map data Map data | Sharpened and low-pass filtered cryo-EM map. | |||||||||||||||
 Sample Sample |
| |||||||||||||||
 Keywords Keywords | transcription / DNA repair / helicase / multiprotein complex | |||||||||||||||
| Function / homology |  Function and homology information Function and homology informationMMXD complex / core TFIIH complex portion of holo TFIIH complex / Cytosolic iron-sulfur cluster assembly / central nervous system myelin formation / positive regulation of mitotic recombination / hair cell differentiation / nucleotide-excision repair factor 3 complex / nucleotide-excision repair, preincision complex assembly / hair follicle maturation / ventricular system development ...MMXD complex / core TFIIH complex portion of holo TFIIH complex / Cytosolic iron-sulfur cluster assembly / central nervous system myelin formation / positive regulation of mitotic recombination / hair cell differentiation / nucleotide-excision repair factor 3 complex / nucleotide-excision repair, preincision complex assembly / hair follicle maturation / ventricular system development / transcription factor TFIIK complex / CAK-ERCC2 complex / embryonic cleavage / UV protection / regulation of cyclin-dependent protein serine/threonine kinase activity / transcription factor TFIIH core complex / transcription factor TFIIH holo complex / cyclin-dependent protein serine/threonine kinase activator activity / DNA 5'-3' helicase / G protein-coupled receptor internalization / adult heart development / nuclear thyroid hormone receptor binding / transcription preinitiation complex / RNA Polymerase I Transcription Termination / transcription factor TFIID complex / RNA polymerase II general transcription initiation factor activity / erythrocyte maturation / regulation of mitotic cell cycle phase transition / hematopoietic stem cell proliferation / RNA Pol II CTD phosphorylation and interaction with CE during HIV infection / RNA Pol II CTD phosphorylation and interaction with CE / Formation of the Early Elongation Complex / Formation of the HIV-1 Early Elongation Complex / mRNA Capping / spinal cord development / bone mineralization / HIV Transcription Initiation / RNA Polymerase II HIV Promoter Escape / Transcription of the HIV genome / RNA Polymerase II Promoter Escape / RNA Polymerase II Transcription Pre-Initiation And Promoter Opening / RNA Polymerase II Transcription Initiation / RNA Polymerase II Transcription Initiation And Promoter Clearance / ATPase activator activity / 3'-5' DNA helicase activity / DNA 3'-5' helicase / DNA topological change / RNA Polymerase I Transcription Initiation / intrinsic apoptotic signaling pathway by p53 class mediator / regulation of G1/S transition of mitotic cell cycle / hematopoietic stem cell differentiation / embryonic organ development / Tat-mediated elongation of the HIV-1 transcript / Cyclin E associated events during G1/S transition / Formation of HIV-1 elongation complex containing HIV-1 Tat / transcription elongation by RNA polymerase I / response to UV / Cyclin A:Cdk2-associated events at S phase entry / Formation of HIV elongation complex in the absence of HIV Tat / cyclin-dependent protein kinase holoenzyme complex / Cyclin A/B1/B2 associated events during G2/M transition / transcription by RNA polymerase I / RNA Polymerase II Transcription Elongation / Formation of RNA Pol II elongation complex / extracellular matrix organization / hormone-mediated signaling pathway / transcription-coupled nucleotide-excision repair / RNA Polymerase II Pre-transcription Events / insulin-like growth factor receptor signaling pathway / positive regulation of smooth muscle cell proliferation / DNA helicase activity / determination of adult lifespan / post-embryonic development / maturation of SSU-rRNA from tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / TP53 Regulates Transcription of DNA Repair Genes / nucleotide-excision repair / transcription initiation at RNA polymerase II promoter / promoter-specific chromatin binding / RNA Polymerase I Promoter Escape / transcription elongation by RNA polymerase II / chromosome segregation / G1/S transition of mitotic cell cycle / cellular response to gamma radiation / NoRC negatively regulates rRNA expression / response to calcium ion / spindle / multicellular organism growth / Dual Incision in GG-NER / Transcription-Coupled Nucleotide Excision Repair (TC-NER) / Formation of TC-NER Pre-Incision Complex / Formation of Incision Complex in GG-NER / Dual incision in TC-NER / Cyclin D associated events in G1 / Gap-filling DNA repair synthesis and ligation in TC-NER / intracellular protein localization / RUNX1 regulates transcription of genes involved in differentiation of HSCs / 4 iron, 4 sulfur cluster binding / response to oxidative stress / double-stranded DNA binding / transcription by RNA polymerase II Similarity search - Function | |||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.7 Å | |||||||||||||||
 Authors Authors | Greber BJ / Toso D | |||||||||||||||
| Funding support |  United States, United States,  Switzerland, 4 items Switzerland, 4 items
| |||||||||||||||
 Citation Citation |  Journal: Elife / Year: 2019 Journal: Elife / Year: 2019Title: The complete structure of the human TFIIH core complex. Authors: Basil J Greber / Daniel B Toso / Jie Fang / Eva Nogales /  Abstract: Transcription factor IIH (TFIIH) is a heterodecameric protein complex critical for transcription initiation by RNA polymerase II and nucleotide excision DNA repair. The TFIIH core complex is ...Transcription factor IIH (TFIIH) is a heterodecameric protein complex critical for transcription initiation by RNA polymerase II and nucleotide excision DNA repair. The TFIIH core complex is sufficient for its repair functions and harbors the XPB and XPD DNA-dependent ATPase/helicase subunits, which are affected by human disease mutations. Transcription initiation additionally requires the CdK activating kinase subcomplex. Previous structural work has provided only partial insight into the architecture of TFIIH and its interactions within transcription pre-initiation complexes. Here, we present the complete structure of the human TFIIH core complex, determined by phase-plate cryo-electron microscopy at 3.7 Å resolution. The structure uncovers the molecular basis of TFIIH assembly, revealing how the recruitment of XPB by p52 depends on a pseudo-symmetric dimer of homologous domains in these two proteins. The structure also suggests a function for p62 in the regulation of XPD, and allows the mapping of previously unresolved human disease mutations. | |||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_0452.map.gz emd_0452.map.gz | 59.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-0452-v30.xml emd-0452-v30.xml emd-0452.xml emd-0452.xml | 32.1 KB 32.1 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_0452.png emd_0452.png | 166 KB | ||
| Filedesc metadata |  emd-0452.cif.gz emd-0452.cif.gz | 8.9 KB | ||
| Others |  emd_0452_half_map_1.map.gz emd_0452_half_map_1.map.gz emd_0452_half_map_2.map.gz emd_0452_half_map_2.map.gz | 49.7 MB 49.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-0452 http://ftp.pdbj.org/pub/emdb/structures/EMD-0452 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-0452 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-0452 | HTTPS FTP |
-Related structure data
| Related structure data |  6nmiMC  0587C  0588C  0589C  0602C  0603C  0604C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | |
| EM raw data |  EMPIAR-10430 (Title: Phase-plate cryo-EM of human TFIIH / Data size: 9.2 TB EMPIAR-10430 (Title: Phase-plate cryo-EM of human TFIIH / Data size: 9.2 TBData #1: Unaligned movies of human TFIIH, using the Volta phase plate, dataset 1 [micrographs - multiframe] Data #2: Unaligned movies of human TFIIH, using the Volta phase plate, dataset 2 [micrographs - multiframe] Data #3: Unaligned movies of human TFIIH, using the Volta phase plate, dataset 3 [micrographs - multiframe] Data #4: Unaligned movies of human TFIIH, using the Volta phase plate, dataset 4 [micrographs - multiframe] Data #5: Unaligned movies of human TFIIH, using the Volta phase plate, dataset 5 [micrographs - multiframe] Data #6: Unaligned movies of human TFIIH, using the Volta phase plate, dataset 6 [micrographs - multiframe]) |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_0452.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_0452.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Sharpened and low-pass filtered cryo-EM map. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.15 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
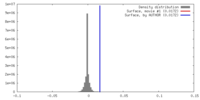
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Half map: Unfiltered half-map.
| File | emd_0452_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Unfiltered half-map. | ||||||||||||
| Projections & Slices |
| ||||||||||||
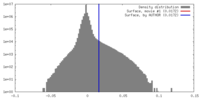
| Density Histograms |
-Half map: Unfiltered half-map.
| File | emd_0452_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Unfiltered half-map. | ||||||||||||
| Projections & Slices |
| ||||||||||||
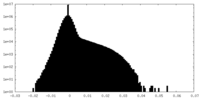
| Density Histograms |
- Sample components
Sample components
+Entire : Transcription factor IIH (TFIIH)
+Supramolecule #1: Transcription factor IIH (TFIIH)
+Macromolecule #1: General transcription and DNA repair factor IIH helicase subunit XPB
+Macromolecule #2: General transcription and DNA repair factor IIH helicase subunit XPD
+Macromolecule #3: General transcription factor IIH subunit 1, p62
+Macromolecule #4: General transcription factor IIH subunit 4, p52
+Macromolecule #5: General transcription factor IIH subunit 2, p44
+Macromolecule #6: General transcription factor IIH subunit 3, p34
+Macromolecule #7: General transcription factor IIH subunit 5, p8
+Macromolecule #8: CDK-activating kinase assembly factor MAT1
+Macromolecule #9: IRON/SULFUR CLUSTER
+Macromolecule #10: ZINC ION
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.0049 mg/mL | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.9 Component:
| ||||||||||||||
| Grid | Model: C-flat / Material: COPPER / Mesh: 400 / Support film - #0 - Film type ID: 1 / Support film - #0 - Material: CARBON / Support film - #0 - topology: HOLEY / Support film - #1 - Film type ID: 2 / Support film - #1 - Material: CARBON / Support film - #1 - topology: CONTINUOUS / Pretreatment - Type: PLASMA CLEANING / Details: Gatan Solarus | ||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV Details: A thin film of continuous carbon was floated onto Protochips C-flat CF-4/2 holey carbon grids and glow discharged or plasma cleaned before application of 4 uL of sample solution.. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Phase plate: VOLTA PHASE PLATE / Energy filter - Name: GIF Quantum LS / Energy filter - Slit width: 20 eV |
| Details | Residual beam tilt corrected in RELION 3. |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Number grids imaged: 7 / Number real images: 21437 / Average exposure time: 8.25 sec. / Average electron dose: 50.0 e/Å2 Details: Images were collected as dose-fractionated movie frames (33 or 50 frames per exposure). |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 43478 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 0.7000000000000001 µm / Nominal defocus min: 0.5 µm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model |
| ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Details | Reference structures and homology models were docked and subsequently completely rebuilt according to the density. Parts of the structure were traced de novo. | ||||||||||||||||
| Refinement | Space: REAL / Protocol: RIGID BODY FIT | ||||||||||||||||
| Output model |  PDB-6nmi: |
 Movie
Movie Controller
Controller





























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)