[English] 日本語
 Yorodumi
Yorodumi- PDB-3j5m: Cryo-EM structure of the BG505 SOSIP.664 HIV-1 Env trimer with 3 ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 3j5m | ||||||
|---|---|---|---|---|---|---|---|
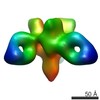
| Title | Cryo-EM structure of the BG505 SOSIP.664 HIV-1 Env trimer with 3 PGV04 Fabs | ||||||
 Components Components |
| ||||||
 Keywords Keywords | VIRAL PROTEIN/IMMUNE SYSTEM / HIV-1 trimeric spike / gp140 / SOSIP / broadly neutralizing antibody / PGV04 / Env / envelope glycoprotein / VIRAL PROTEIN-IMMUNE SYSTEM complex | ||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of plasma membrane raft polarization / positive regulation of receptor clustering / positive regulation of establishment of T cell polarity / host cell endosome membrane / clathrin-dependent endocytosis of virus by host cell / viral protein processing / fusion of virus membrane with host plasma membrane / virus-mediated perturbation of host defense response / fusion of virus membrane with host endosome membrane / viral envelope ...positive regulation of plasma membrane raft polarization / positive regulation of receptor clustering / positive regulation of establishment of T cell polarity / host cell endosome membrane / clathrin-dependent endocytosis of virus by host cell / viral protein processing / fusion of virus membrane with host plasma membrane / virus-mediated perturbation of host defense response / fusion of virus membrane with host endosome membrane / viral envelope / virion attachment to host cell / apoptotic process / host cell plasma membrane / structural molecule activity / virion membrane / identical protein binding / plasma membrane Similarity search - Function | ||||||
| Biological species |   Human immunodeficiency virus 1 Human immunodeficiency virus 1 Homo sapiens (human) Homo sapiens (human) | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 5.8 Å | ||||||
 Authors Authors | Lyumkis, D. / Julien, J.-P. / Wilson, I.A. / Ward, A.B. | ||||||
 Citation Citation |  Journal: Science / Year: 2013 Journal: Science / Year: 2013Title: Cryo-EM structure of a fully glycosylated soluble cleaved HIV-1 envelope trimer. Authors: Dmitry Lyumkis / Jean-Philippe Julien / Natalia de Val / Albert Cupo / Clinton S Potter / Per-Johan Klasse / Dennis R Burton / Rogier W Sanders / John P Moore / Bridget Carragher / Ian A ...Authors: Dmitry Lyumkis / Jean-Philippe Julien / Natalia de Val / Albert Cupo / Clinton S Potter / Per-Johan Klasse / Dennis R Burton / Rogier W Sanders / John P Moore / Bridget Carragher / Ian A Wilson / Andrew B Ward /  Abstract: The HIV-1 envelope glycoprotein (Env) trimer contains the receptor binding sites and membrane fusion machinery that introduce the viral genome into the host cell. As the only target for broadly ...The HIV-1 envelope glycoprotein (Env) trimer contains the receptor binding sites and membrane fusion machinery that introduce the viral genome into the host cell. As the only target for broadly neutralizing antibodies (bnAbs), Env is a focus for rational vaccine design. We present a cryo-electron microscopy reconstruction and structural model of a cleaved, soluble Env trimer (termed BG505 SOSIP.664 gp140) in complex with a CD4 binding site (CD4bs) bnAb, PGV04, at 5.8 angstrom resolution. The structure reveals the spatial arrangement of Env components, including the V1/V2, V3, HR1, and HR2 domains, as well as shielding glycans. The structure also provides insights into trimer assembly, gp120-gp41 interactions, and the CD4bs epitope cluster for bnAbs, which covers a more extensive area and defines a more complex site of vulnerability than previously described. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  3j5m.cif.gz 3j5m.cif.gz | 450.5 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb3j5m.ent.gz pdb3j5m.ent.gz | 360 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  3j5m.json.gz 3j5m.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  3j5m_validation.pdf.gz 3j5m_validation.pdf.gz | 419.9 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  3j5m_full_validation.pdf.gz 3j5m_full_validation.pdf.gz | 452.4 KB | Display | |
| Data in XML |  3j5m_validation.xml.gz 3j5m_validation.xml.gz | 59.2 KB | Display | |
| Data in CIF |  3j5m_validation.cif.gz 3j5m_validation.cif.gz | 94.2 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/j5/3j5m https://data.pdbj.org/pub/pdb/validation_reports/j5/3j5m ftp://data.pdbj.org/pub/pdb/validation_reports/j5/3j5m ftp://data.pdbj.org/pub/pdb/validation_reports/j5/3j5m | HTTPS FTP |
-Related structure data
| Related structure data |  5779MC  5780C  5781C  5782C M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 53121.105 Da / Num. of mol.: 3 / Fragment: UNP residues 30-504 / Mutation: T332N,A501C Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Human immunodeficiency virus 1 / Gene: env / Cell line (production host): HEK / Production host: Human immunodeficiency virus 1 / Gene: env / Cell line (production host): HEK / Production host:  Homo sapiens (human) / References: UniProt: Q2N0S6 Homo sapiens (human) / References: UniProt: Q2N0S6#2: Protein | Mass: 5464.728 Da / Num. of mol.: 3 / Fragment: SEE REMARK 999 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Human immunodeficiency virus 1 / Gene: env / Cell line (production host): HEK / Production host: Human immunodeficiency virus 1 / Gene: env / Cell line (production host): HEK / Production host:  Homo sapiens (human) Homo sapiens (human)#3: Antibody | Mass: 23073.822 Da / Num. of mol.: 3 / Fragment: Fab Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Cell line (production host): HEK / Production host: Homo sapiens (human) / Cell line (production host): HEK / Production host:  Homo sapiens (human) Homo sapiens (human)#4: Antibody | Mass: 24644.771 Da / Num. of mol.: 3 / Fragment: Fab Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Cell line (production host): HEK / Production host: Homo sapiens (human) / Cell line (production host): HEK / Production host:  Homo sapiens (human) Homo sapiens (human)Sequence details | THE SEQUENCE OF CHAINS B, F, AND J IS ...THE SEQUENCE OF CHAINS B, F, AND J IS AVGIGAVFLG | |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component |
| ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight | Value: 0.6 MDa / Experimental value: NO | ||||||||||||||||||||
| Buffer solution | Name: TBS / pH: 7.5 / Details: TBS | ||||||||||||||||||||
| Specimen | Conc.: 1 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES | ||||||||||||||||||||
| Specimen support | Details: 400 mesh C-Flat CF-22-4C, plasma treated for 5 seconds | ||||||||||||||||||||
| Vitrification | Instrument: HOMEMADE PLUNGER / Cryogen name: ETHANE Details: 3 uL sample applied to grid, adsorbed for 30 seconds, blotted, and plunge-frozen in liquid ethane Method: Specimen was prepared for cryo-EM by applying 3 microliters of sample to a freshly plasma cleaned holey carbon C-flat grid (Protochips, Inc.), allowing the sample to adsorb to the grid for 30 ...Method: Specimen was prepared for cryo-EM by applying 3 microliters of sample to a freshly plasma cleaned holey carbon C-flat grid (Protochips, Inc.), allowing the sample to adsorb to the grid for 30 seconds, followed by blotting with a small piece of filter paper and plunge-freezing into liquid ethane using a manual cryo-plunger in an ambient environment (4 C). |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Tecnai F20 / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TECNAI F20 / Date: Feb 21, 2013 / Details: electron counting mode |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 200 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 200 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal magnification: 29000 X / Calibrated magnification: 29000 X / Nominal defocus max: 5000 nm / Nominal defocus min: 1000 nm / Cs: 2 mm Astigmatism: objective lens astigmatism was corrected by observing Thon rings with the Leginon software Camera length: 0 mm |
| Specimen holder | Specimen holder model: SIDE ENTRY, EUCENTRIC |
| Image recording | Electron dose: 32 e/Å2 / Film or detector model: GATAN K2 SUMMIT (4k x 4k) |
| Image scans | Num. digital images: 6355 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Relative weight: 1 |
- Processing
Processing
| EM software |
| |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF correction | Details: Frealign | |||||||||||||||
| Symmetry | Point symmetry: C3 (3 fold cyclic) | |||||||||||||||
| 3D reconstruction | Method: projection matching using a resolution-limited scheme Resolution: 5.8 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 49572 / Nominal pixel size: 1.21 Å / Actual pixel size: 1.21 Å Details: Final maps were calculated after sorting for the presence of sub-stoichiometrically labeled trimers (Single particle details: reconstructed using a resolution-limited refinement procedure ...Details: Final maps were calculated after sorting for the presence of sub-stoichiometrically labeled trimers (Single particle details: reconstructed using a resolution-limited refinement procedure implemented in Xmipp and Frealign) (Single particle--Applied symmetry: C3) Symmetry type: POINT | |||||||||||||||
| Atomic model building | Space: REAL | |||||||||||||||
| Atomic model building |
| |||||||||||||||
| Refinement step | Cycle: LAST
|
 Movie
Movie Controller
Controller







 PDBj
PDBj








