+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2y3v | ||||||
|---|---|---|---|---|---|---|---|
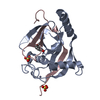
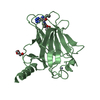
| Title | N-terminal head domain of Danio rerio SAS-6 | ||||||
 Components Components | SPINDLE ASSEMBLY ABNORMAL PROTEIN 6 HOMOLOG | ||||||
 Keywords Keywords | STRUCTURAL PROTEIN / CYTOSKELETON / BASAL BODY / CENTRIOLE / CARTWHEEL / CARTWHEEL HUB | ||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of mitotic spindle organization / deuterosome / procentriole replication complex / positive regulation of centriole replication / nuclear division / centriole replication / positive regulation of spindle assembly / centrosome duplication / embryonic cleavage / positive regulation of G1/S transition of mitotic cell cycle ...positive regulation of mitotic spindle organization / deuterosome / procentriole replication complex / positive regulation of centriole replication / nuclear division / centriole replication / positive regulation of spindle assembly / centrosome duplication / embryonic cleavage / positive regulation of G1/S transition of mitotic cell cycle / centriole / mitotic spindle organization / spermatogenesis / centrosome / cytoplasm Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.92 Å MOLECULAR REPLACEMENT / Resolution: 1.92 Å | ||||||
 Authors Authors | van Breugel, M. | ||||||
 Citation Citation |  Journal: Science / Year: 2011 Journal: Science / Year: 2011Title: Structures of SAS-6 suggest its organization in centrioles. Authors: van Breugel, M. / Hirono, M. / Andreeva, A. / Yanagisawa, H.A. / Yamaguchi, S. / Nakazawa, Y. / Morgner, N. / Petrovich, M. / Ebong, I.O. / Robinson, C.V. / Johnson, C.M. / Veprintsev, D. / Zuber, B. | ||||||
| History |
| ||||||
| Remark 700 | SHEET DETERMINATION METHOD: DSSP THE SHEETS PRESENTED AS "AA" IN EACH CHAIN ON SHEET RECORDS BELOW ... SHEET DETERMINATION METHOD: DSSP THE SHEETS PRESENTED AS "AA" IN EACH CHAIN ON SHEET RECORDS BELOW IS ACTUALLY AN 7-STRANDED BARREL THIS IS REPRESENTED BY A 8-STRANDED SHEET IN WHICH THE FIRST AND LAST STRANDS ARE IDENTICAL. THE SHEETS PRESENTED AS "BA" IN EACH CHAIN ON SHEET RECORDS BELOW IS ACTUALLY AN 7-STRANDED BARREL THIS IS REPRESENTED BY A 8-STRANDED SHEET IN WHICH THE FIRST AND LAST STRANDS ARE IDENTICAL. THE SHEETS PRESENTED AS "CA" IN EACH CHAIN ON SHEET RECORDS BELOW IS ACTUALLY AN 7-STRANDED BARREL THIS IS REPRESENTED BY A 8-STRANDED SHEET IN WHICH THE FIRST AND LAST STRANDS ARE IDENTICAL. THE SHEETS PRESENTED AS "DA" IN EACH CHAIN ON SHEET RECORDS BELOW IS ACTUALLY AN 7-STRANDED BARREL THIS IS REPRESENTED BY A 8-STRANDED SHEET IN WHICH THE FIRST AND LAST STRANDS ARE IDENTICAL. |
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2y3v.cif.gz 2y3v.cif.gz | 141.9 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2y3v.ent.gz pdb2y3v.ent.gz | 113.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2y3v.json.gz 2y3v.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  2y3v_validation.pdf.gz 2y3v_validation.pdf.gz | 451.7 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  2y3v_full_validation.pdf.gz 2y3v_full_validation.pdf.gz | 459.1 KB | Display | |
| Data in XML |  2y3v_validation.xml.gz 2y3v_validation.xml.gz | 27.6 KB | Display | |
| Data in CIF |  2y3v_validation.cif.gz 2y3v_validation.cif.gz | 39.4 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/y3/2y3v https://data.pdbj.org/pub/pdb/validation_reports/y3/2y3v ftp://data.pdbj.org/pub/pdb/validation_reports/y3/2y3v ftp://data.pdbj.org/pub/pdb/validation_reports/y3/2y3v | HTTPS FTP |
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||||||||||
| 2 | 
| ||||||||||||||||
| 3 | 
| ||||||||||||||||
| 4 | 
| ||||||||||||||||
| Unit cell |
| ||||||||||||||||
| Noncrystallographic symmetry (NCS) | NCS oper:
|
- Components
Components
| #1: Protein | Mass: 18273.865 Da / Num. of mol.: 4 / Fragment: HEAD DOMAIN, RESIDUES 1-156 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   #2: Water | ChemComp-HOH / | Sequence details | THE N-TERMINAL THREE RESIDUES (GPH) STEM FROM THE EXPRESSION VECTOR. THE S102N MUTATION PROBABLY ...THE N-TERMINAL THREE RESIDUES (GPH) STEM FROM THE EXPRESSION | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.9 Å3/Da / Density % sol: 57.66 % Description: THE SELENOMETHIONINE DERIVATIVE STRUCTURE WAS SOLVED BY MAD USING A DERIVATIVE OF A L57M,L90M MUTANT. AN INITIAL MODEL WAS BUILT USING THE EXPERIMENTAL ELECTRON DENSITY MAP AND THEN USED ...Description: THE SELENOMETHIONINE DERIVATIVE STRUCTURE WAS SOLVED BY MAD USING A DERIVATIVE OF A L57M,L90M MUTANT. AN INITIAL MODEL WAS BUILT USING THE EXPERIMENTAL ELECTRON DENSITY MAP AND THEN USED FOR MOLECULAR REPLACEMENT AGAINST THE NATIVE DATASET. |
|---|---|
| Crystal grow | pH: 8.5 / Details: 0.1 M TRIS PH 8.5, 25% PEG 3350. |
-Data collection
| Diffraction | Mean temperature: 77 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: ID29 / Wavelength: 0.979 / Beamline: ID29 / Wavelength: 0.979 |
| Detector | Type: ADSC QUANTUM 315r / Detector: CCD / Date: Dec 12, 2009 |
| Radiation | Monochromator: LIQUID NITROGEN COOLED CHANNEL-CUT SILICON MONOCHROMATOR Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.979 Å / Relative weight: 1 |
| Reflection | Resolution: 1.92→54.8 Å / Num. obs: 63745 / % possible obs: 99.4 % / Observed criterion σ(I): 0 / Redundancy: 12.3 % / Rmerge(I) obs: 0.13 / Net I/σ(I): 12.4 |
| Reflection shell | Resolution: 1.92→2.02 Å / Redundancy: 11.9 % / Rmerge(I) obs: 1.5 / Mean I/σ(I) obs: 1.8 / % possible all: 98.6 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: SE-MET DERIVATIVE Resolution: 1.92→54.799 Å / SU ML: 0.28 / σ(F): 0 / Phase error: 26.81 / Stereochemistry target values: ML Details: RESIDUES A-2, C-2, AND C145 TO C156, D-2 TO D0, AND D155-D156, ARE DISORDERED. LOOP REGION C112 TO C120 SHOWED POOR ELECTRON DENSITY.
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.83 Å / VDW probe radii: 1.1 Å / Solvent model: FLAT BULK SOLVENT MODEL / Bsol: 40.852 Å2 / ksol: 0.35 e/Å3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 39.18 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.92→54.799 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj
