+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7egb | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
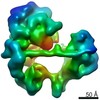
| Title | TFIID-based holo PIC on SCP promoter | |||||||||
 Components Components |
| |||||||||
 Keywords Keywords | TRANSCRIPTION / TFIID / preinitiation complex / core promoter / transcription initiation | |||||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of MHC class I biosynthetic process / microfibril binding / response to paraquat / spermine transport / SAGA complex assembly / lateral mesodermal cell differentiation / DNA-templated transcription open complex formation / allantois development / MMXD complex / pre-snoRNP complex ...negative regulation of MHC class I biosynthetic process / microfibril binding / response to paraquat / spermine transport / SAGA complex assembly / lateral mesodermal cell differentiation / DNA-templated transcription open complex formation / allantois development / MMXD complex / pre-snoRNP complex / positive regulation of androgen receptor signaling pathway / core TFIIH complex portion of holo TFIIH complex / TFIIH-class transcription factor complex binding / negative regulation of protein autoubiquitination / positive regulation of core promoter binding / Cytosolic iron-sulfur cluster assembly / negative regulation of MHC class II biosynthetic process / RNA polymerase II core complex assembly / transcription factor TFTC complex / central nervous system myelin formation / RNA polymerase II CTD heptapeptide repeat S5 kinase activity / regulation of cell cycle G1/S phase transition / RNA polymerase I general transcription initiation factor activity / positive regulation of mitotic recombination / transcription factor TFIIE complex / RNA polymerase transcription factor SL1 complex / hair cell differentiation / meiotic sister chromatid cohesion / nucleotide-excision repair factor 3 complex / phosphatase activator activity / nucleotide-excision repair, preincision complex assembly / hair follicle maturation / ventricular system development / SLIK (SAGA-like) complex / snRNA transcription by RNA polymerase II / RNA polymerase III general transcription initiation factor activity / TFIIF-class transcription factor complex binding / transcription factor TFIIK complex / transcriptional start site selection at RNA polymerase II promoter / RNA polymerase I core promoter sequence-specific DNA binding / CAK-ERCC2 complex / hepatocyte differentiation / transcription factor TFIIF complex / positive regulation of response to cytokine stimulus / female germ cell nucleus / RNA Polymerase III Transcription Initiation From Type 1 Promoter / RNA Polymerase III Transcription Initiation From Type 2 Promoter / RNA Polymerase III Transcription Initiation From Type 3 Promoter / embryonic cleavage / Formation of RNA Pol II elongation complex / Formation of the Early Elongation Complex / Transcriptional regulation by small RNAs / RNA Polymerase II Pre-transcription Events / TP53 Regulates Transcription of DNA Repair Genes / FGFR2 alternative splicing / RNA polymerase II transcribes snRNA genes / mRNA Capping / mRNA Splicing - Minor Pathway / Processing of Capped Intron-Containing Pre-mRNA / RNA Polymerase II Promoter Escape / RNA Polymerase II Transcription Pre-Initiation And Promoter Opening / RNA Polymerase II Transcription Initiation / RNA Polymerase II Transcription Elongation / RNA Polymerase II Transcription Initiation And Promoter Clearance / RNA Pol II CTD phosphorylation and interaction with CE / Estrogen-dependent gene expression / transcription factor TFIIA complex / Formation of TC-NER Pre-Incision Complex / Dual incision in TC-NER / Gap-filling DNA repair synthesis and ligation in TC-NER / mRNA Splicing - Major Pathway / C2H2 zinc finger domain binding / maintenance of protein location in nucleus / RNA Polymerase III Abortive And Retractive Initiation / UV protection / regulation of cyclin-dependent protein serine/threonine kinase activity / male pronucleus / female pronucleus / transcription factor TFIIH core complex / transcription factor TFIIH holo complex / cyclin-dependent protein serine/threonine kinase activator activity / RNA polymerase II general transcription initiation factor binding / germinal vesicle / DNA 5'-3' helicase / host-mediated activation of viral transcription / G protein-coupled receptor internalization / RNA polymerase binding / histone H3K4me3 reader activity / regulation of fat cell differentiation / nuclear vitamin D receptor binding / adult heart development / [RNA-polymerase]-subunit kinase / box C/D snoRNP assembly / Abortive elongation of HIV-1 transcript in the absence of Tat / SAGA complex / nuclear thyroid hormone receptor binding / FGFR2 alternative splicing / transcription preinitiation complex / RNA Polymerase I Transcription Termination / cyclin-dependent protein serine/threonine kinase regulator activity Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.3 Å | |||||||||
 Authors Authors | Chen, X. / Wu, Z. / Hou, H. / Qi, Y. / Wang, X. / Li, J. / Xu, Y. | |||||||||
| Funding support | 1items
| |||||||||
 Citation Citation |  Journal: Science / Year: 2021 Journal: Science / Year: 2021Title: Structural insights into preinitiation complex assembly on core promoters. Authors: Xizi Chen / Yilun Qi / Zihan Wu / Xinxin Wang / Jiabei Li / Dan Zhao / Haifeng Hou / Yan Li / Zishuo Yu / Weida Liu / Mo Wang / Yulei Ren / Ze Li / Huirong Yang / Yanhui Xu /  Abstract: Transcription factor IID (TFIID) recognizes core promoters and supports preinitiation complex (PIC) assembly for RNA polymerase II (Pol II)-mediated eukaryotic transcription. We determined the ...Transcription factor IID (TFIID) recognizes core promoters and supports preinitiation complex (PIC) assembly for RNA polymerase II (Pol II)-mediated eukaryotic transcription. We determined the structures of human TFIID-based PIC in three stepwise assembly states and revealed two-track PIC assembly: stepwise promoter deposition to Pol II and extensive modular reorganization on track I (on TATA-TFIID-binding element promoters) versus direct promoter deposition on track II (on TATA-only and TATA-less promoters). The two tracks converge at an ~50-subunit holo PIC in identical conformation, whereby TFIID stabilizes PIC organization and supports loading of cyclin-dependent kinase (CDK)-activating kinase (CAK) onto Pol II and CAK-mediated phosphorylation of the Pol II carboxyl-terminal domain. Unexpectedly, TBP of TFIID similarly bends TATA box and TATA-less promoters in PIC. Our study provides structural visualization of stepwise PIC assembly on highly diversified promoters. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7egb.cif.gz 7egb.cif.gz | 2.6 MB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7egb.ent.gz pdb7egb.ent.gz | Display |  PDB format PDB format | |
| PDBx/mmJSON format |  7egb.json.gz 7egb.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/eg/7egb https://data.pdbj.org/pub/pdb/validation_reports/eg/7egb ftp://data.pdbj.org/pub/pdb/validation_reports/eg/7egb ftp://data.pdbj.org/pub/pdb/validation_reports/eg/7egb | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  31111MC  7edxC  7eg7C  7eg8C  7eg9C  7egaC  7egcC  7egdC  7egeC  7egfC  7eggC  7eghC  7egiC  7egjC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Protein , 8 types, 8 molecules 089PUxyz
| #1: Protein | Mass: 35873.965 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: MNAT1, CAP35, MAT1, RNF66 / Production host: Homo sapiens (human) / Gene: MNAT1, CAP35, MAT1, RNF66 / Production host:  Homo sapiens (human) / References: UniProt: P51948 Homo sapiens (human) / References: UniProt: P51948 |
|---|---|
| #9: Protein | Mass: 39090.344 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: CDK7, CAK, CAK1, CDKN7, MO15, STK1 / Production host: Homo sapiens (human) / Gene: CDK7, CAK, CAK1, CDKN7, MO15, STK1 / Production host:  Homo sapiens (human) Homo sapiens (human)References: UniProt: P50613, cyclin-dependent kinase, [RNA-polymerase]-subunit kinase |
| #10: Protein | Mass: 37695.473 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: CCNH / Production host: Homo sapiens (human) / Gene: CCNH / Production host:  Homo sapiens (human) / References: UniProt: P51946 Homo sapiens (human) / References: UniProt: P51946 |
| #22: Protein | Mass: 37729.938 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: TBP, GTF2D1, TF2D, TFIID / Production host: Homo sapiens (human) / Gene: TBP, GTF2D1, TF2D, TFIID / Production host:  Homo sapiens (human) / References: UniProt: P20226 Homo sapiens (human) / References: UniProt: P20226 |
| #27: Protein | Mass: 49516.094 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GTF2E1, TF2E1 / Production host: Homo sapiens (human) / Gene: GTF2E1, TF2E1 / Production host:  Homo sapiens (human) / References: UniProt: P29083 Homo sapiens (human) / References: UniProt: P29083 |
| #43: Protein | Mass: 7655.123 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #44: Protein | Mass: 13310.284 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #45: Protein | Mass: 7018.244 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
-General transcription factor IIH subunit ... , 5 types, 5 molecules 12345
| #2: Protein | Mass: 62116.492 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GTF2H1, BTF2 / Production host: Homo sapiens (human) / Gene: GTF2H1, BTF2 / Production host:  Homo sapiens (human) / References: UniProt: P32780 Homo sapiens (human) / References: UniProt: P32780 |
|---|---|
| #3: Protein | Mass: 44481.996 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GTF2H2, BTF2P44 / Production host: Homo sapiens (human) / Gene: GTF2H2, BTF2P44 / Production host:  Homo sapiens (human) / References: UniProt: Q13888 Homo sapiens (human) / References: UniProt: Q13888 |
| #4: Protein | Mass: 34416.008 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GTF2H3 / Production host: Homo sapiens (human) / Gene: GTF2H3 / Production host:  Homo sapiens (human) / References: UniProt: Q13889 Homo sapiens (human) / References: UniProt: Q13889 |
| #5: Protein | Mass: 52245.156 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GTF2H4 / Production host: Homo sapiens (human) / Gene: GTF2H4 / Production host:  Homo sapiens (human) / References: UniProt: Q92759 Homo sapiens (human) / References: UniProt: Q92759 |
| #6: Protein | Mass: 8060.362 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GTF2H5, C6orf175, TTDA / Production host: Homo sapiens (human) / Gene: GTF2H5, C6orf175, TTDA / Production host:  Homo sapiens (human) / References: UniProt: Q6ZYL4 Homo sapiens (human) / References: UniProt: Q6ZYL4 |
-General transcription and DNA repair factor IIH helicase subunit ... , 2 types, 2 molecules 67
| #7: Protein | Mass: 89404.734 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: ERCC3, XPB, XPBC / Production host: Homo sapiens (human) / Gene: ERCC3, XPB, XPBC / Production host:  Homo sapiens (human) / References: UniProt: P19447, DNA helicase Homo sapiens (human) / References: UniProt: P19447, DNA helicase |
|---|---|
| #8: Protein | Mass: 87021.078 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: ERCC2, XPD, XPDC / Production host: Homo sapiens (human) / Gene: ERCC2, XPD, XPDC / Production host:  Homo sapiens (human) / References: UniProt: P18074, DNA helicase Homo sapiens (human) / References: UniProt: P18074, DNA helicase |
-Transcription initiation factor ... , 17 types, 23 molecules ABDdEeFfGHIiJjLlOQRVckm
| #11: Protein | Mass: 212956.172 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: TAF1, BA2R, CCG1, CCGS, TAF2A / Production host: Homo sapiens (human) / Gene: TAF1, BA2R, CCG1, CCGS, TAF2A / Production host:  Homo sapiens (human) Homo sapiens (human)References: UniProt: P21675, histone acetyltransferase, non-specific serine/threonine protein kinase | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| #12: Protein | Mass: 137159.984 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: TAF2, CIF150, TAF2B / Production host: Homo sapiens (human) / Gene: TAF2, CIF150, TAF2B / Production host:  Homo sapiens (human) / References: UniProt: Q6P1X5 Homo sapiens (human) / References: UniProt: Q6P1X5 | ||||||||||||||||||||||||||||
| #13: Protein | Mass: 110221.883 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: TAF4, TAF2C, TAF2C1, TAF4A, TAFII130, TAFII135 / Production host: Homo sapiens (human) / Gene: TAF4, TAF2C, TAF2C1, TAF4A, TAFII130, TAFII135 / Production host:  Homo sapiens (human) / References: UniProt: O00268 Homo sapiens (human) / References: UniProt: O00268#14: Protein | Mass: 86932.109 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: TAF5, TAF2D / Production host: Homo sapiens (human) / Gene: TAF5, TAF2D / Production host:  Homo sapiens (human) / References: UniProt: Q15542 Homo sapiens (human) / References: UniProt: Q15542#15: Protein | Mass: 72749.297 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: TAF6, TAF2E, TAFII70 / Production host: Homo sapiens (human) / Gene: TAF6, TAF2E, TAFII70 / Production host:  Homo sapiens (human) / References: UniProt: P49848 Homo sapiens (human) / References: UniProt: P49848#16: Protein | | Mass: 40325.117 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: TAF7, TAF2F, TAFII55 / Production host: Homo sapiens (human) / Gene: TAF7, TAF2F, TAFII55 / Production host:  Homo sapiens (human) / References: UniProt: Q15545 Homo sapiens (human) / References: UniProt: Q15545#17: Protein | | Mass: 34304.359 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: TAF8, TAFII43, TBN / Production host: Homo sapiens (human) / Gene: TAF8, TAFII43, TBN / Production host:  Homo sapiens (human) / References: UniProt: Q7Z7C8 Homo sapiens (human) / References: UniProt: Q7Z7C8#18: Protein | Mass: 29006.838 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: TAF9, TAF2G, TAFII31 / Production host: Homo sapiens (human) / Gene: TAF9, TAF2G, TAFII31 / Production host:  Homo sapiens (human) / References: UniProt: Q16594 Homo sapiens (human) / References: UniProt: Q16594#19: Protein | Mass: 21731.248 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: TAF10, TAF2A, TAF2H, TAFII30 / Production host: Homo sapiens (human) / Gene: TAF10, TAF2A, TAF2H, TAFII30 / Production host:  Homo sapiens (human) / References: UniProt: Q12962 Homo sapiens (human) / References: UniProt: Q12962#20: Protein | Mass: 17948.467 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: TAF12, TAF15, TAF2J, TAFII20 / Production host: Homo sapiens (human) / Gene: TAF12, TAF15, TAF2J, TAFII20 / Production host:  Homo sapiens (human) / References: UniProt: Q16514 Homo sapiens (human) / References: UniProt: Q16514#21: Protein | | Mass: 12469.091 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GTF2A2, TF2A2 / Production host: Homo sapiens (human) / Gene: GTF2A2, TF2A2 / Production host:  Homo sapiens (human) / References: UniProt: P52657 Homo sapiens (human) / References: UniProt: P52657#23: Protein | | Mass: 41544.551 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GTF2A1, TF2A1 / Production host: Homo sapiens (human) / Gene: GTF2A1, TF2A1 / Production host:  Homo sapiens (human) / References: UniProt: P52655 Homo sapiens (human) / References: UniProt: P52655#24: Protein | | Mass: 34877.949 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GTF2B, TF2B, TFIIB / Production host: Homo sapiens (human) / Gene: GTF2B, TF2B, TFIIB / Production host:  Homo sapiens (human) / References: UniProt: Q00403, histone acetyltransferase Homo sapiens (human) / References: UniProt: Q00403, histone acetyltransferase#28: Protein | | Mass: 33106.824 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GTF2E2, TF2E2 / Production host: Homo sapiens (human) / Gene: GTF2E2, TF2E2 / Production host:  Homo sapiens (human) / References: UniProt: P29084 Homo sapiens (human) / References: UniProt: P29084#31: Protein | | Mass: 103769.320 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: TAF3 / Production host: Homo sapiens (human) / Gene: TAF3 / Production host:  Homo sapiens (human) / References: UniProt: Q5VWG9 Homo sapiens (human) / References: UniProt: Q5VWG9#32: Protein | | Mass: 23340.094 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: TAF11, TAF2I, PRO2134 / Production host: Homo sapiens (human) / Gene: TAF11, TAF2I, PRO2134 / Production host:  Homo sapiens (human) / References: UniProt: Q15544 Homo sapiens (human) / References: UniProt: Q15544#33: Protein | | Mass: 14307.068 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: TAF13, TAF2K, TAFII18 / Production host: Homo sapiens (human) / Gene: TAF13, TAF2K, TAFII18 / Production host:  Homo sapiens (human) / References: UniProt: Q15543 Homo sapiens (human) / References: UniProt: Q15543 |
-General transcription factor IIF subunit ... , 2 types, 2 molecules ST
| #25: Protein | Mass: 58343.578 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GTF2F1, RAP74 / Production host: Homo sapiens (human) / Gene: GTF2F1, RAP74 / Production host:  Homo sapiens (human) / References: UniProt: P35269 Homo sapiens (human) / References: UniProt: P35269 |
|---|---|
| #26: Protein | Mass: 28427.309 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GTF2F2, RAP30 / Production host: Homo sapiens (human) / Gene: GTF2F2, RAP30 / Production host:  Homo sapiens (human) / References: UniProt: P13984, DNA helicase Homo sapiens (human) / References: UniProt: P13984, DNA helicase |
-DNA chain , 2 types, 2 molecules XY
| #29: DNA chain | Mass: 29188.615 Da / Num. of mol.: 1 / Source method: obtained synthetically / Source: (synth.)  Homo sapiens (human) Homo sapiens (human) |
|---|---|
| #30: DNA chain | Mass: 21094.428 Da / Num. of mol.: 1 / Source method: obtained synthetically / Source: (synth.)  Homo sapiens (human) Homo sapiens (human) |
-DNA-directed RNA ... , 9 types, 9 molecules opqrstuvw
| #34: Protein | Mass: 217420.047 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) Homo sapiens (human)References: UniProt: P24928, DNA-directed RNA polymerase, RNA-directed RNA polymerase |
|---|---|
| #35: Protein | Mass: 134041.422 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  References: UniProt: A0A4X1TVZ5, DNA-directed RNA polymerase |
| #36: Protein | Mass: 31439.074 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #37: Protein | Mass: 16331.255 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #38: Protein | Mass: 24644.318 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #39: Protein | Mass: 14477.001 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #40: Protein | Mass: 19314.283 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #41: Protein | Mass: 17162.273 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #42: Protein | Mass: 14541.221 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
-Non-polymers , 3 types, 19 molecules 




| #46: Chemical | ChemComp-ZN / #47: Chemical | ChemComp-SF4 / | #48: Chemical | ChemComp-MG / | |
|---|
-Details
| Has ligand of interest | N |
|---|---|
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Source (natural) |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Source (recombinant) |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Buffer solution | pH: 7.9 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal defocus max: 2500 nm / Nominal defocus min: 1500 nm |
| Image recording | Electron dose: 50 e/Å2 / Film or detector model: GATAN K2 SUMMIT (4k x 4k) |
- Processing
Processing
| CTF correction | Type: NONE |
|---|---|
| 3D reconstruction | Resolution: 3.3 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 92806 / Symmetry type: POINT |
 Movie
Movie Controller
Controller





















































 PDBj
PDBj
























































