[English] 日本語
 Yorodumi
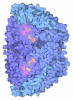
Yorodumi- PDB-7b2h: Crystal structure of the methyl-coenzyme M reductase from Methano... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7b2h | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Crystal structure of the methyl-coenzyme M reductase from Methanothermobacter Marburgensis derivatized with xenon | |||||||||
 Components Components | (Methyl-coenzyme M reductase I subunit ...) x 3 | |||||||||
 Keywords Keywords | TRANSFERASE / Ethyl-CoM reductase / Methyl-CoM reductase / xenon-derivatization / F430-cofactor / post-translational modification / coenzyme M / coenzyme B / thermophile / archaea. | |||||||||
| Function / homology |  Function and homology information Function and homology informationcoenzyme-B sulfoethylthiotransferase / coenzyme-B sulfoethylthiotransferase activity / methanogenesis / metal ion binding / cytoplasm Similarity search - Function | |||||||||
| Biological species |   Methanothermobacter marburgensis (archaea) Methanothermobacter marburgensis (archaea) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / MOLECULAR REPLACEMENT /  molecular replacement / Resolution: 2.12 Å molecular replacement / Resolution: 2.12 Å | |||||||||
 Authors Authors | Wagner, T. / Lemaire, O.N. / Engilberge, S. | |||||||||
| Funding support |  Germany, 2items Germany, 2items
| |||||||||
 Citation Citation |  Journal: Science / Year: 2021 Journal: Science / Year: 2021Title: Crystal structure of a key enzyme for anaerobic ethane activation. Authors: Hahn, C.J. / Lemaire, O.N. / Kahnt, J. / Engilberge, S. / Wegener, G. / Wagner, T. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7b2h.cif.gz 7b2h.cif.gz | 993 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7b2h.ent.gz pdb7b2h.ent.gz | 821.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7b2h.json.gz 7b2h.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/b2/7b2h https://data.pdbj.org/pub/pdb/validation_reports/b2/7b2h ftp://data.pdbj.org/pub/pdb/validation_reports/b2/7b2h ftp://data.pdbj.org/pub/pdb/validation_reports/b2/7b2h | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  7b1sC  7b2cC  5a0yS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Methyl-coenzyme M reductase I subunit ... , 3 types, 6 molecules ADBECF
| #1: Protein | Mass: 60637.648 Da / Num. of mol.: 2 / Mutation: wild-type / Source method: isolated from a natural source Details: The alpha subunit contains six modified amino acids near the active site region. Methylated His-257, Arg-271, Gln-400 and Cys-452. A thioglycine at position 445, forming a thiopeptide bond. ...Details: The alpha subunit contains six modified amino acids near the active site region. Methylated His-257, Arg-271, Gln-400 and Cys-452. A thioglycine at position 445, forming a thiopeptide bond. A didehydroaspartate residue at position 450. A methylated Arg-271 Source: (natural)   Methanothermobacter marburgensis (strain ATCC BAA-927 / DSM 2133 / JCM 14651 / NBRC 100331 / OCM 82 / Marburg) (archaea) Methanothermobacter marburgensis (strain ATCC BAA-927 / DSM 2133 / JCM 14651 / NBRC 100331 / OCM 82 / Marburg) (archaea)Cell line: / / Organ: / / Plasmid details: / / Variant: / Strain: ATCC BAA-927 / DSM 2133 / JCM 14651 / NBRC 100331 / OCM 82 / Marburg Tissue: / References: UniProt: P11558, coenzyme-B sulfoethylthiotransferase #2: Protein | Mass: 47279.676 Da / Num. of mol.: 2 / Mutation: wild-type / Source method: isolated from a natural source Source: (natural)   Methanothermobacter marburgensis (strain ATCC BAA-927 / DSM 2133 / JCM 14651 / NBRC 100331 / OCM 82 / Marburg) (archaea) Methanothermobacter marburgensis (strain ATCC BAA-927 / DSM 2133 / JCM 14651 / NBRC 100331 / OCM 82 / Marburg) (archaea)Cell line: / / Organ: / / Plasmid details: / / Variant: / Strain: ATCC BAA-927 / DSM 2133 / JCM 14651 / NBRC 100331 / OCM 82 / Marburg Tissue: / References: UniProt: P11560, coenzyme-B sulfoethylthiotransferase #3: Protein | Mass: 28797.234 Da / Num. of mol.: 2 / Mutation: wild-type / Source method: isolated from a natural source Source: (natural)   Methanothermobacter marburgensis (strain ATCC BAA-927 / DSM 2133 / JCM 14651 / NBRC 100331 / OCM 82 / Marburg) (archaea) Methanothermobacter marburgensis (strain ATCC BAA-927 / DSM 2133 / JCM 14651 / NBRC 100331 / OCM 82 / Marburg) (archaea)Cell line: / / Organ: / / Plasmid details: / / Variant: / Strain: ATCC BAA-927 / DSM 2133 / JCM 14651 / NBRC 100331 / OCM 82 / Marburg Tissue: / References: UniProt: P11562, coenzyme-B sulfoethylthiotransferase |
|---|
-Non-polymers , 12 types, 823 molecules 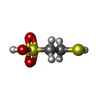
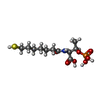




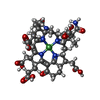
















| #4: Chemical | | #5: Chemical | #6: Chemical | ChemComp-K / #7: Chemical | ChemComp-PEG / #8: Chemical | ChemComp-GOL / | #9: Chemical | ChemComp-EDO / | #10: Chemical | #11: Chemical | ChemComp-MG / #12: Chemical | ChemComp-XE / #13: Chemical | ChemComp-SO4 / | #14: Chemical | #15: Water | ChemComp-HOH / | |
|---|
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.14 Å3/Da / Density % sol: 42.52 % / Description: Butterfly intense yellow color |
|---|---|
| Crystal grow | Temperature: 291.15 K / Method: vapor diffusion / pH: 7.5 Details: Crystal was obtained under aerobic condition using a crystallisation reservoir containing 27.5 % (v/v) polyethylene glycol 400, 100 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid ...Details: Crystal was obtained under aerobic condition using a crystallisation reservoir containing 27.5 % (v/v) polyethylene glycol 400, 100 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) pH 7.5, 250 mM MgCl2, 200 mM NaCl and 20 mM 2-oxoglutarate. The protein sample was in 25 mM Tris-HCl pH 7.6, 10% glycerol and 2 mM DTT at a protein concentration of 25 mg ml-1. PH range: 7 - 8 / Temp details: / |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SLS SLS  / Beamline: X06DA / Wavelength: 2.07 Å / Beamline: X06DA / Wavelength: 2.07 Å |
| Detector | Type: DECTRIS PILATUS 2M-F / Detector: PIXEL / Date: Nov 8, 2019 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 2.07 Å / Relative weight: 1 |
| Reflection | Resolution: 2.12→48.17 Å / Num. obs: 116190 / % possible obs: 89.36 % / Redundancy: 6.5 % / CC1/2: 0.998 / Rmerge(I) obs: 0.0902 / Rpim(I) all: 0.03796 / Rrim(I) all: 0.09807 / Net I/σ(I): 14.24 |
| Reflection shell | Resolution: 2.12→2.198 Å / Redundancy: 4.3 % / Rmerge(I) obs: 1.173 / Mean I/σ(I) obs: 1.01 / Num. unique obs: 7094 / CC1/2: 0.511 / Rpim(I) all: 0.6248 / Rrim(I) all: 1.339 / % possible all: 54.62 |
-Phasing
| Phasing | Method:  molecular replacement molecular replacement |
|---|
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 5A0Y Resolution: 2.12→48.17 Å / SU ML: 0.23 / Cross valid method: THROUGHOUT / σ(F): 0.89 / Phase error: 23.02 / Stereochemistry target values: ML Details: Riding hydrogens were added for the last refinement cycles but omitted in the deposited model
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 150.21 Å2 / Biso mean: 50.839 Å2 / Biso min: 22.16 Å2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 2.12→48.17 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Rfactor Rfree error: 0 / Total num. of bins used: 30
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Origin x: 21.9668 Å / Origin y: 39.188 Å / Origin z: -32.4923 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj