[English] 日本語
 Yorodumi
Yorodumi- PDB-6t78: Structure of human Sox11 transcription factor in complex with a s... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6t78 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of human Sox11 transcription factor in complex with a short DNA fragment | ||||||||||||
 Components Components |
| ||||||||||||
 Keywords Keywords | NUCLEAR PROTEIN / Sox11 / transcription factor / pioneer factor / DNA | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationclosure of optic fissure / positive regulation of lens epithelial cell proliferation / soft palate development / negative regulation of transcription regulatory region DNA binding / cornea development in camera-type eye / noradrenergic neuron differentiation / negative regulation of lymphocyte proliferation / regulation of transforming growth factor beta receptor signaling pathway / lens morphogenesis in camera-type eye / embryonic skeletal system morphogenesis ...closure of optic fissure / positive regulation of lens epithelial cell proliferation / soft palate development / negative regulation of transcription regulatory region DNA binding / cornea development in camera-type eye / noradrenergic neuron differentiation / negative regulation of lymphocyte proliferation / regulation of transforming growth factor beta receptor signaling pathway / lens morphogenesis in camera-type eye / embryonic skeletal system morphogenesis / positive regulation of hippo signaling / neuroepithelial cell differentiation / embryonic digestive tract morphogenesis / sympathetic nervous system development / camera-type eye morphogenesis / hard palate development / positive regulation of hormone secretion / positive regulation of BMP signaling pathway / oligodendrocyte development / positive regulation of ossification / negative regulation of glial cell proliferation / positive regulation of neurogenesis / positive regulation of stem cell proliferation / ventricular septum morphogenesis / spinal cord development / eyelid development in camera-type eye / outflow tract morphogenesis / lung morphogenesis / skeletal muscle cell differentiation / positive regulation of osteoblast differentiation / glial cell proliferation / positive regulation of neuron differentiation / kidney development / brain development / neuron differentiation / nervous system development / DNA-binding transcription activator activity, RNA polymerase II-specific / DNA-binding transcription factor activity, RNA polymerase II-specific / transcription cis-regulatory region binding / RNA polymerase II cis-regulatory region sequence-specific DNA binding / DNA-binding transcription factor activity / negative regulation of gene expression / positive regulation of gene expression / chromatin / negative regulation of transcription by RNA polymerase II / positive regulation of transcription by RNA polymerase II / nucleoplasm / nucleus / plasma membrane Similarity search - Function | ||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human)synthetic construct (others) | ||||||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / MOLECULAR REPLACEMENT /  molecular replacement / Resolution: 2.504 Å molecular replacement / Resolution: 2.504 Å | ||||||||||||
 Authors Authors | Dodonova, S.O. / Zhu, F. / Dienemann, C. / Taipale, J. / Cramer, P. | ||||||||||||
| Funding support |  Germany, 3items Germany, 3items
| ||||||||||||
 Citation Citation |  Journal: Nature / Year: 2020 Journal: Nature / Year: 2020Title: Nucleosome-bound SOX2 and SOX11 structures elucidate pioneer factor function. Authors: Svetlana O Dodonova / Fangjie Zhu / Christian Dienemann / Jussi Taipale / Patrick Cramer /   Abstract: 'Pioneer' transcription factors are required for stem-cell pluripotency, cell differentiation and cell reprogramming. Pioneer factors can bind nucleosomal DNA to enable gene expression from regions ...'Pioneer' transcription factors are required for stem-cell pluripotency, cell differentiation and cell reprogramming. Pioneer factors can bind nucleosomal DNA to enable gene expression from regions of the genome with closed chromatin. SOX2 is a prominent pioneer factor that is essential for pluripotency and self-renewal of embryonic stem cells. Here we report cryo-electron microscopy structures of the DNA-binding domains of SOX2 and its close homologue SOX11 bound to nucleosomes. The structures show that SOX factors can bind and locally distort DNA at superhelical location 2. The factors also facilitate detachment of terminal nucleosomal DNA from the histone octamer, which increases DNA accessibility. SOX-factor binding to the nucleosome can also lead to a repositioning of the N-terminal tail of histone H4 that includes residue lysine 16. We speculate that this repositioning is incompatible with higher-order nucleosome stacking, which involves contacts of the H4 tail with a neighbouring nucleosome. Our results indicate that pioneer transcription factors can use binding energy to initiate chromatin opening, and thereby facilitate nucleosome remodelling and subsequent transcription. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6t78.cif.gz 6t78.cif.gz | 137.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6t78.ent.gz pdb6t78.ent.gz | 103.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6t78.json.gz 6t78.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/t7/6t78 https://data.pdbj.org/pub/pdb/validation_reports/t7/6t78 ftp://data.pdbj.org/pub/pdb/validation_reports/t7/6t78 ftp://data.pdbj.org/pub/pdb/validation_reports/t7/6t78 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  6t79C  6t7aC  6t7bC  6t7cC  6t7dC  1gt0S S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 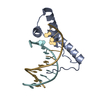
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2 | 
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unit cell |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Noncrystallographic symmetry (NCS) | NCS domain:
NCS domain segments:
NCS ensembles :
|
- Components
Components
| #1: Protein | Mass: 12856.941 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: SOX11 / Plasmid: LIC-1B (MacroLabs) / Production host: Homo sapiens (human) / Gene: SOX11 / Plasmid: LIC-1B (MacroLabs) / Production host:  #2: DNA chain | Mass: 4890.181 Da / Num. of mol.: 2 / Source method: obtained synthetically / Source: (synth.) synthetic construct (others) #3: DNA chain | Mass: 4900.273 Da / Num. of mol.: 2 / Source method: obtained synthetically / Source: (synth.) synthetic construct (others) #4: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.76 Å3/Da / Density % sol: 55.43 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, hanging drop / pH: 4.5 Details: Sodium Acetate 100 mM pH 4.5, Calcium Acetate Hydrate 200 mM, 15-18 % PEG 400 |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SLS SLS  / Beamline: X10SA / Wavelength: 0.999 Å / Beamline: X10SA / Wavelength: 0.999 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Detector | Type: DECTRIS PILATUS3 6M / Detector: PIXEL / Date: Apr 12, 2017 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation wavelength | Wavelength: 0.999 Å / Relative weight: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection | Resolution: 2.5→45.956 Å / Num. obs: 17066 / % possible obs: 99.9 % / Redundancy: 20.55 % / Biso Wilson estimate: 75.835 Å2 / CC1/2: 1 / Rmerge(I) obs: 0.126 / Rrim(I) all: 0.129 / Χ2: 1.052 / Net I/σ(I): 16.54 / Num. measured all: 350700 / Scaling rejects: 96 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection shell | Diffraction-ID: 1
|
-Phasing
| Phasing | Method:  molecular replacement molecular replacement | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Phasing MR |
|
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 1GT0 Resolution: 2.504→45.956 Å / SU ML: 0.44 / Cross valid method: THROUGHOUT / σ(F): 1.35 / Phase error: 32.88
| |||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å | |||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 215.77 Å2 / Biso mean: 96.3747 Å2 / Biso min: 48.08 Å2 | |||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 2.504→45.956 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints NCS |
| |||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Rfactor Rfree error: 0
| |||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Origin x: 29.4328 Å / Origin y: 55.5287 Å / Origin z: 23.2924 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller















 PDBj
PDBj







































