[English] 日本語
 Yorodumi
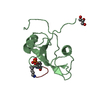
Yorodumi- PDB-6bub: Crystal structure of the PI3KC2alpha PX domain in space group P432 -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6bub | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of the PI3KC2alpha PX domain in space group P432 | ||||||
 Components Components | Phosphatidylinositol 4-phosphate 3-kinase C2 domain-containing subunit alpha | ||||||
 Keywords Keywords | TRANSFERASE / PX domain / lipid binding / phosphoinositide / PI3-kinase | ||||||
| Function / homology |  Function and homology information Function and homology informationvascular associated smooth muscle contraction / Synthesis of PIPs at the late endosome membrane / Synthesis of PIPs at the early endosome membrane / phosphatidylinositol-4-phosphate 3-kinase / Synthesis of PIPs at the Golgi membrane / clathrin coat assembly / phosphatidylinositol biosynthetic process / membrane organization / clathrin-coated vesicle / 1-phosphatidylinositol-4-phosphate 3-kinase activity ...vascular associated smooth muscle contraction / Synthesis of PIPs at the late endosome membrane / Synthesis of PIPs at the early endosome membrane / phosphatidylinositol-4-phosphate 3-kinase / Synthesis of PIPs at the Golgi membrane / clathrin coat assembly / phosphatidylinositol biosynthetic process / membrane organization / clathrin-coated vesicle / 1-phosphatidylinositol-4-phosphate 3-kinase activity / phosphatidylinositol-4,5-bisphosphate 3-kinase / 1-phosphatidylinositol-4,5-bisphosphate 3-kinase activity / phosphatidylinositol 3-kinase / positive regulation of cell migration involved in sprouting angiogenesis / phosphatidylinositol-3-phosphate biosynthetic process / 1-phosphatidylinositol-3-kinase activity / clathrin binding / Golgi Associated Vesicle Biogenesis / platelet-derived growth factor receptor signaling pathway / phosphatidylinositol-mediated signaling / exocytosis / Synthesis of PIPs at the plasma membrane / phosphatidylinositol binding / positive regulation of autophagy / trans-Golgi network / phosphatidylinositol 3-kinase/protein kinase B signal transduction / epidermal growth factor receptor signaling pathway / endocytosis / insulin receptor signaling pathway / cell migration / Clathrin-mediated endocytosis / vesicle / extracellular exosome / nucleoplasm / ATP binding / membrane / plasma membrane / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / MOLECULAR REPLACEMENT /  molecular replacement / Resolution: 2.604 Å molecular replacement / Resolution: 2.604 Å | ||||||
 Authors Authors | Chen, K.-E. / Collins, B.M. | ||||||
 Citation Citation |  Journal: Structure / Year: 2018 Journal: Structure / Year: 2018Title: Molecular Basis for Membrane Recruitment by the PX and C2 Domains of Class II Phosphoinositide 3-Kinase-C2α. Authors: Kai-En Chen / Vikas A Tillu / Mintu Chandra / Brett M Collins /  Abstract: Phosphorylation of phosphoinositides by the class II phosphatidylinositol 3-kinase (PI3K) PI3K-C2α is essential for many processes, including neuroexocytosis and formation of clathrin-coated ...Phosphorylation of phosphoinositides by the class II phosphatidylinositol 3-kinase (PI3K) PI3K-C2α is essential for many processes, including neuroexocytosis and formation of clathrin-coated vesicles. A defining feature of the class II PI3Ks is a C-terminal module composed of phox-homology (PX) and C2 membrane interacting domains; however, the mechanisms that control their specific cellular localization remain poorly understood. Here we report the crystal structure of the C2 domain of PI3K-C2α in complex with the phosphoinositide head-group mimic inositol hexaphosphate, revealing two distinct pockets for membrane binding. The C2 domain preferentially binds to phosphatidylinositol 4,5-bisphosphate and phosphatidylinositol (3,4,5)-trisphosphate, and low-resolution structures of the combined PX-C2 module by small-angle X-ray scattering reveal a compact conformation in which cooperative lipid binding by each domain binding can occur. Finally, we demonstrate an unexpected role for calcium in perturbing the membrane interactions of the PX-C2 module, which we speculate may be important for regulating the activity of PI3K-C2α. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6bub.cif.gz 6bub.cif.gz | 42.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6bub.ent.gz pdb6bub.ent.gz | 27.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6bub.json.gz 6bub.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/bu/6bub https://data.pdbj.org/pub/pdb/validation_reports/bu/6bub ftp://data.pdbj.org/pub/pdb/validation_reports/bu/6bub ftp://data.pdbj.org/pub/pdb/validation_reports/bu/6bub | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  6btyC  6btzC  6bu0C  2iwlS C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Components on special symmetry positions |
|
- Components
Components
| #1: Protein | Mass: 16738.223 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: PIK3C2A / Production host: Homo sapiens (human) / Gene: PIK3C2A / Production host:  References: UniProt: O00443, phosphatidylinositol-4-phosphate 3-kinase |
|---|---|
| #2: Chemical | ChemComp-SO4 / |
| #3: Chemical | ChemComp-GOL / |
| #4: Water | ChemComp-HOH / |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.92 Å3/Da / Density % sol: 68.6 % / Mosaicity: 0.11 ° |
|---|---|
| Crystal grow | Temperature: 295 K / Method: vapor diffusion, hanging drop / pH: 8 / Details: (NH4)2SO4 |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  Australian Synchrotron Australian Synchrotron  / Beamline: MX2 / Wavelength: 1 Å / Beamline: MX2 / Wavelength: 1 Å |
| Detector | Type: ADSC QUANTUM 315r / Detector: CCD / Date: Nov 5, 2016 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1 Å / Relative weight: 1 |
| Reflection | Resolution: 2.6→47.48 Å / Num. obs: 8738 / % possible obs: 99.8 % / Redundancy: 40.6 % / Biso Wilson estimate: 47.02 Å2 / CC1/2: 0.997 / Rmerge(I) obs: 0.132 / Rpim(I) all: 0.021 / Rrim(I) all: 0.133 / Net I/σ(I): 24.9 / Num. measured all: 354795 / Scaling rejects: 14 |
| Reflection shell | Resolution: 2.6→2.72 Å / Redundancy: 40.7 % / Rmerge(I) obs: 0.869 / Num. measured all: 41490 / Num. unique obs: 1020 / CC1/2: 0.934 / Rpim(I) all: 0.136 / Rrim(I) all: 0.88 / Net I/σ(I) obs: 4.8 / % possible all: 98.6 |
-Phasing
| Phasing | Method:  molecular replacement molecular replacement | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Phasing MR | Model details: Phaser MODE: MR_AUTO
|
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 2IWL Resolution: 2.604→47.483 Å / SU ML: 0.32 / Cross valid method: THROUGHOUT / σ(F): 1.38 / Phase error: 25.74 / Stereochemistry target values: MLHL
| ||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | ||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 132.91 Å2 / Biso mean: 47.7949 Å2 / Biso min: 20 Å2 | ||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 2.604→47.483 Å
| ||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Rfactor Rfree error: 0 / Total num. of bins used: 3
|
 Movie
Movie Controller
Controller













 PDBj
PDBj








