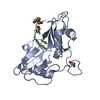
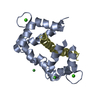
Entry Database : PDB / ID : 5o1wTitle Structure of Nrd1 RNA binding domain Protein NRD1 Keywords / / / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Saccharomyces cerevisiae (brewer's yeast)Method / / / Resolution : 2.3 Å Authors Franco-Echevarria, E. / Perez-Canadillas, J.M. / Gonzalez, B. Funding support Organization Grant number Country CSIC
Journal : Nucleic Acids Res. / Year : 2017Title : The structure of transcription termination factor Nrd1 reveals an original mode for GUAA recognition.Authors : Franco-Echevarria, E. / Gonzalez-Polo, N. / Zorrilla, S. / Martinez-Lumbreras, S. / Santiveri, C.M. / Campos-Olivas, R. / Sanchez, M. / Calvo, O. / Gonzalez, B. / Perez-Canadillas, J.M. History Deposition May 19, 2017 Deposition site / Processing site Revision 1.0 Aug 2, 2017 Provider / Type Revision 1.1 Sep 13, 2017 Group / Category / Item Revision 1.2 Oct 11, 2017 Group / Category Item _citation.journal_volume / _citation.page_first ... _citation.journal_volume / _citation.page_first / _citation.page_last / _citation.pdbx_database_id_DOI / _citation.pdbx_database_id_PubMed / _citation.title Revision 1.3 May 8, 2024 Group / Database references / Category / chem_comp_bond / database_2Item / _database_2.pdbx_database_accession
Show all Show less
 Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information
 X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  SAD / Resolution: 2.3 Å
SAD / Resolution: 2.3 Å  Authors
Authors Spain, 1items
Spain, 1items  Citation
Citation Journal: Nucleic Acids Res. / Year: 2017
Journal: Nucleic Acids Res. / Year: 2017 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 5o1w.cif.gz
5o1w.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb5o1w.ent.gz
pdb5o1w.ent.gz PDB format
PDB format 5o1w.json.gz
5o1w.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/o1/5o1w
https://data.pdbj.org/pub/pdb/validation_reports/o1/5o1w ftp://data.pdbj.org/pub/pdb/validation_reports/o1/5o1w
ftp://data.pdbj.org/pub/pdb/validation_reports/o1/5o1w Links
Links Assembly
Assembly
 Components
Components

 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  ESRF
ESRF  / Beamline: ID23-2 / Wavelength: 0.97625 Å
/ Beamline: ID23-2 / Wavelength: 0.97625 Å Processing
Processing SAD / Resolution: 2.3→56.9 Å / Cor.coef. Fo:Fc: 0.952 / Cor.coef. Fo:Fc free: 0.938 / SU B: 12.104 / SU ML: 0.135 / Cross valid method: THROUGHOUT / ESU R: 0.203 / ESU R Free: 0.175 / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
SAD / Resolution: 2.3→56.9 Å / Cor.coef. Fo:Fc: 0.952 / Cor.coef. Fo:Fc free: 0.938 / SU B: 12.104 / SU ML: 0.135 / Cross valid method: THROUGHOUT / ESU R: 0.203 / ESU R Free: 0.175 / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS Movie
Movie Controller
Controller
















 PDBj
PDBj


