[English] 日本語
 Yorodumi
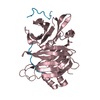
Yorodumi- PDB-5m5v: Clathrin heavy chain N-terminal domain bound to a clathrin-box mo... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5m5v | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Clathrin heavy chain N-terminal domain bound to a clathrin-box motif from hepatitis D virus large antigen (clade 2) | ||||||||||||
 Components Components |
| ||||||||||||
 Keywords Keywords | ENDOCYTOSIS / hepatitis delta virus / HDAg-L | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationRetrograde neurotrophin signalling / Recycling pathway of L1 / WNT5A-dependent internalization of FZD4 / WNT5A-dependent internalization of FZD2, FZD5 and ROR2 / LDL clearance / Gap junction degradation / Formation of annular gap junctions / RHOU GTPase cycle / RHOV GTPase cycle / clathrin coat of trans-Golgi network vesicle ...Retrograde neurotrophin signalling / Recycling pathway of L1 / WNT5A-dependent internalization of FZD4 / WNT5A-dependent internalization of FZD2, FZD5 and ROR2 / LDL clearance / Gap junction degradation / Formation of annular gap junctions / RHOU GTPase cycle / RHOV GTPase cycle / clathrin coat of trans-Golgi network vesicle / Golgi Associated Vesicle Biogenesis / clathrin light chain binding / Lysosome Vesicle Biogenesis / negative regulation of hyaluronan biosynthetic process / clathrin complex / MHC class II antigen presentation / VLDLR internalisation and degradation / clathrin coat of coated pit / clathrin coat disassembly / Cargo recognition for clathrin-mediated endocytosis / clathrin-coated endocytic vesicle / membrane coat / clathrin coat assembly / Clathrin-mediated endocytosis / arrestin family protein binding / receptor-mediated endocytosis / intracellular protein transport / virion component / viral penetration into host nucleus / spindle / autophagy / disordered domain specific binding / melanosome / mitotic cell cycle / host cell / protein domain specific binding / cell division / symbiont entry into host cell / host cell nucleus / structural molecule activity / mitochondrion / RNA binding / identical protein binding Similarity search - Function | ||||||||||||
| Biological species |   Hepatitis delta virus Hepatitis delta virus | ||||||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.96 Å MOLECULAR REPLACEMENT / Resolution: 1.96 Å | ||||||||||||
 Authors Authors | Muenzner, J. / Graham, S.C. | ||||||||||||
| Funding support |  United Kingdom, United Kingdom,  United States, 3items United States, 3items
| ||||||||||||
 Citation Citation |  Journal: Traffic / Year: 2017 Journal: Traffic / Year: 2017Title: Cellular and viral peptides bind multiple sites on the N-terminal domain of clathrin. Authors: Muenzner, J. / Traub, L.M. / Kelly, B.T. / Graham, S.C. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5m5v.cif.gz 5m5v.cif.gz | 309.4 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5m5v.ent.gz pdb5m5v.ent.gz | 251.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5m5v.json.gz 5m5v.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/m5/5m5v https://data.pdbj.org/pub/pdb/validation_reports/m5/5m5v ftp://data.pdbj.org/pub/pdb/validation_reports/m5/5m5v ftp://data.pdbj.org/pub/pdb/validation_reports/m5/5m5v | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  5m5rC  5m5sC  5m5tC  5m5uC  5m61C  1c9iS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||||||||||||
| 2 | 
| ||||||||||||||||||
| Unit cell |
| ||||||||||||||||||
| Components on special symmetry positions |
| ||||||||||||||||||
| Noncrystallographic symmetry (NCS) | NCS domain:
NCS domain segments: Component-ID: _ / Ens-ID: 1 / Beg auth comp-ID: ILE / Beg label comp-ID: ILE / End auth comp-ID: LEU / End label comp-ID: LEU / Refine code: _ / Auth seq-ID: 4 - 363 / Label seq-ID: 6 - 365
|
- Components
Components
| #1: Antibody | Mass: 40540.473 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Details: N-terminal residues 'GS' are residual following cleavage of the purification tag Source: (gene. exp.)   #2: Protein/peptide | Mass: 1012.181 Da / Num. of mol.: 6 / Fragment: Clathrin-box like motif, UNP Residues 203-209 / Source method: obtained synthetically Details: N- and C-terminal serine residues are non-natural and were added to enhance solubility Source: (synth.)  Hepatitis delta virus / References: UniProt: A4ZNG7 Hepatitis delta virus / References: UniProt: A4ZNG7#3: Chemical | ChemComp-GOL / | #4: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.64 Å3/Da / Density % sol: 66.19 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, sitting drop / pH: 7 Details: 200 nL protein:peptide (20 mg/mL NTD and 3.6 mM peptide) plus 400 nL reservoir equilibrated against a 80 uL reservoir of 1.75 M sodium malonate pH 7.0 |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  Diamond Diamond  / Beamline: I02 / Wavelength: 0.97949 Å / Beamline: I02 / Wavelength: 0.97949 Å |
| Detector | Type: DECTRIS PILATUS 6M-F / Detector: PIXEL / Date: Oct 7, 2013 / Details: KB mirrors |
| Radiation | Monochromator: Si(111) crystal / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.97949 Å / Relative weight: 1 |
| Reflection | Resolution: 1.96→39.2 Å / Num. obs: 79976 / % possible obs: 94.6 % / Redundancy: 2.5 % / CC1/2: 0.996 / Rmerge(I) obs: 0.081 / Net I/σ(I): 7.2 |
| Reflection shell | Resolution: 1.96→2 Å / Redundancy: 2.3 % / Rmerge(I) obs: 0.581 / Mean I/σ(I) obs: 1.5 / CC1/2: 0.563 / % possible all: 95.9 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 1C9I Resolution: 1.96→39.2 Å / Cor.coef. Fo:Fc: 0.967 / Cor.coef. Fo:Fc free: 0.958 / SU B: 5.697 / SU ML: 0.082 / Cross valid method: THROUGHOUT / ESU R: 0.119 / ESU R Free: 0.112 / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 33.338 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: 1 / Resolution: 1.96→39.2 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller











 PDBj
PDBj




