+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6qnp | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | CLATHRIN HEAVY CHAIN N-TERMINAL DOMAIN BOUND TO GTSE1 LIDL MOTIF | |||||||||
 Components Components |
| |||||||||
 Keywords Keywords | CELL CYCLE / BETA-PROPELLER / CLATHRIN-BOX / LIDL-MOTIF | |||||||||
| Function / homology |  Function and homology information Function and homology informationclathrin coat of trans-Golgi network vesicle / clathrin light chain binding / WNT5A-dependent internalization of FZD2, FZD5 and ROR2 / negative regulation of hyaluronan biosynthetic process / clathrin complex / clathrin coat / Entry of Influenza Virion into Host Cell via Endocytosis / WNT5A-dependent internalization of FZD4 / amyloid-beta clearance by transcytosis / clathrin coat of coated pit ...clathrin coat of trans-Golgi network vesicle / clathrin light chain binding / WNT5A-dependent internalization of FZD2, FZD5 and ROR2 / negative regulation of hyaluronan biosynthetic process / clathrin complex / clathrin coat / Entry of Influenza Virion into Host Cell via Endocytosis / WNT5A-dependent internalization of FZD4 / amyloid-beta clearance by transcytosis / clathrin coat of coated pit / clathrin coat disassembly / Retrograde neurotrophin signalling / clathrin-coated endocytic vesicle / clathrin coat assembly / mitotic spindle microtubule / LDL clearance / Formation of annular gap junctions / clathrin-dependent endocytosis / ALK mutants bind TKIs / Gap junction degradation / endolysosome membrane / retrograde transport, endosome to Golgi / clathrin-coated vesicle / RHOV GTPase cycle / low-density lipoprotein particle receptor binding / Lysosome Vesicle Biogenesis / Golgi Associated Vesicle Biogenesis / ubiquitin-specific protease binding / RHOU GTPase cycle / Recycling pathway of L1 / EPH-ephrin mediated repulsion of cells / negative regulation of protein localization to plasma membrane / microtubule-based process / cytoplasmic microtubule / MHC class II antigen presentation / receptor-mediated endocytosis / VLDLR internalisation and degradation / regulation of mitotic spindle organization / trans-Golgi network membrane / DNA damage response, signal transduction by p53 class mediator / clathrin-coated endocytic vesicle membrane / transferrin transport / G2/M Checkpoints / intracellular protein transport / receptor internalization / spindle / autophagy / centriolar satellite / disordered domain specific binding / osteoblast differentiation / The role of GTSE1 in G2/M progression after G2 checkpoint / mitotic spindle / Signaling by ALK fusions and activated point mutants / melanosome / Cargo recognition for clathrin-mediated endocytosis / mitotic cell cycle / double-stranded RNA binding / extracellular vesicle / Clathrin-mediated endocytosis / microtubule cytoskeleton / sperm midpiece / microtubule binding / lysosome / endosome / cell division / focal adhesion / protein kinase binding / structural molecule activity / protein-containing complex / RNA binding / extracellular exosome / nucleoplasm / membrane / plasma membrane / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / MOLECULAR REPLACEMENT /  molecular replacement / Resolution: 2.7 Å molecular replacement / Resolution: 2.7 Å | |||||||||
 Authors Authors | Porfetye, A.T. / Lin, Y. / Vetter, I.R. | |||||||||
 Citation Citation |  Journal: J.Cell Biol. / Year: 2020 Journal: J.Cell Biol. / Year: 2020Title: Clathrin's adaptor interaction sites are repurposed to stabilize microtubules during mitosis. Authors: Rondelet, A. / Lin, Y.C. / Singh, D. / Porfetye, A.T. / Thakur, H.C. / Hecker, A. / Brinkert, P. / Schmidt, N. / Bendre, S. / Muller, F. / Mazul, L. / Widlund, P.O. / Bange, T. / Hiller, M. ...Authors: Rondelet, A. / Lin, Y.C. / Singh, D. / Porfetye, A.T. / Thakur, H.C. / Hecker, A. / Brinkert, P. / Schmidt, N. / Bendre, S. / Muller, F. / Mazul, L. / Widlund, P.O. / Bange, T. / Hiller, M. / Vetter, I.R. / Bird, A.W. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6qnp.cif.gz 6qnp.cif.gz | 305.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6qnp.ent.gz pdb6qnp.ent.gz | 246.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6qnp.json.gz 6qnp.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/qn/6qnp https://data.pdbj.org/pub/pdb/validation_reports/qn/6qnp ftp://data.pdbj.org/pub/pdb/validation_reports/qn/6qnp ftp://data.pdbj.org/pub/pdb/validation_reports/qn/6qnp | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  6qnnC  5m5tS C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| 3 | 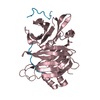
| ||||||||
| 4 | 
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Antibody | Mass: 40543.516 Da / Num. of mol.: 4 Fragment: LIDL motifs A-D, REFSEQ RESIDUES 653-719, N-TERMINAL 'GPLGS' ARE NON-NATURAL Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: CLTC, CLH17, CLTCL2, KIAA0034 / Production host: Homo sapiens (human) / Gene: CLTC, CLH17, CLTCL2, KIAA0034 / Production host:  #2: Protein | Mass: 7040.994 Da / Num. of mol.: 4 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GTSE1 / Production host: Homo sapiens (human) / Gene: GTSE1 / Production host:  #3: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 2 X-RAY DIFFRACTION / Number of used crystals: 2 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.39 Å3/Da / Density % sol: 40.69 % |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion / pH: 7 Details: 12% PEG 4000, 0.1M SODIUM ACETATE, 0.1M HEPES pH 7.0 |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SLS SLS  / Beamline: X10SA / Wavelength: 0.97793 Å / Beamline: X10SA / Wavelength: 0.97793 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Detector | Type: DECTRIS PILATUS3 6M / Detector: PIXEL / Date: Sep 8, 2017 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation wavelength | Wavelength: 0.97793 Å / Relative weight: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection | Resolution: 2.7→49.19 Å / Num. obs: 43602 / % possible obs: 99.2 % / Redundancy: 6.876 % / Biso Wilson estimate: 65.226 Å2 / CC1/2: 0.998 / Rmerge(I) obs: 0.141 / Rrim(I) all: 0.153 / Χ2: 1.002 / Net I/σ(I): 10.39 / Num. measured all: 299788 / Scaling rejects: 47 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection shell | Diffraction-ID: 1
|
-Phasing
| Phasing | Method:  molecular replacement molecular replacement |
|---|
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 5M5T Resolution: 2.7→49.19 Å / SU ML: 0.46 / Cross valid method: THROUGHOUT / σ(F): 1.35 / Phase error: 34.12
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 125.64 Å2 / Biso mean: 71.1235 Å2 / Biso min: 29.26 Å2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 2.7→49.19 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Rfactor Rfree error: 0 / Total num. of bins used: 16
|
 Movie
Movie Controller
Controller













 PDBj
PDBj



















