[English] 日本語
 Yorodumi
Yorodumi- PDB-1u7m: Solution structure of a diiron protein model: Due Ferri(II) turn ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1u7m | ||||||
|---|---|---|---|---|---|---|---|
| Title | Solution structure of a diiron protein model: Due Ferri(II) turn mutant | ||||||
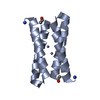
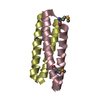
 Components Components | Four-helix bundle model | ||||||
 Keywords Keywords | DE NOVO PROTEIN / Diiron proteins / four-helix bundle / protein design / inter-helical loops | ||||||
| Function / homology | Immunoglobulin FC, subunit C / Single alpha-helices involved in coiled-coils or other helix-helix interfaces / Up-down Bundle / Mainly Alpha Function and homology information Function and homology information | ||||||
| Biological species |  | ||||||
| Method | SOLUTION NMR / Torsion Angle Dynamics, Simulated Annealing, Energy Restrained Minimization | ||||||
 Authors Authors | Maglio, O. / Nastri, F. / Calhoun, J.R. / Lahr, S. / Pavone, V. / DeGrado, W.F. / Lombardi, A. | ||||||
 Citation Citation |  Journal: J.Mol.Biol. / Year: 2005 Journal: J.Mol.Biol. / Year: 2005Title: Analysis and Design of Turns in alpha-Helical Hairpins Authors: Lahr, S.J. / Engel, D.E. / Stayrook, S.E. / Maglio, O. / North, B. / Geremia, S. / Lombardi, A. / Degrado, W.F. #1:  Journal: To be Published Journal: To be PublishedTitle: Solution characterization of diiron proteins containing two distinct types of inter-helical loops Authors: Maglio, O. / Nastri, F. / Calhoun, J.R. / Lahr, S. / Pavone, V. / DeGrado, W.F. / Lombardi, A. #2: Journal: Protein Sci. / Year: 2001 Title: Proton and metal ion-dependent assembly of a model diiron protein Authors: Pasternak, A. / Kaplan, J. / Lear, J.D. / DeGrado, W.F. #3:  Journal: Proc.Natl.Acad.Sci.USA / Year: 2000 Journal: Proc.Natl.Acad.Sci.USA / Year: 2000Title: Inaugural article: retrostructural analysis of metalloproteins: application to the design of a minimal model for diiron proteins Authors: Lombardi, A. / Summa, C.M. / Geremia, S. / Randaccio, L. / Pavone, V. / DeGrado, W.F. #4:  Journal: Proc.Natl.Acad.Sci.USA / Year: 2003 Journal: Proc.Natl.Acad.Sci.USA / Year: 2003Title: Preorganization of molecular binding sites in designed diiron proteins Authors: Maglio, O. / Nastri, F. / Pavone, V. / Lombardi, A. / DeGrado, W.F. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1u7m.cif.gz 1u7m.cif.gz | 714.3 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1u7m.ent.gz pdb1u7m.ent.gz | 598.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1u7m.json.gz 1u7m.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/u7/1u7m https://data.pdbj.org/pub/pdb/validation_reports/u7/1u7m ftp://data.pdbj.org/pub/pdb/validation_reports/u7/1u7m ftp://data.pdbj.org/pub/pdb/validation_reports/u7/1u7m | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| NMR ensembles |
|
- Components
Components
| #1: Protein | Mass: 6477.396 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   #2: Chemical | |
|---|
-Experimental details
-Experiment
| Experiment | Method: SOLUTION NMR | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| NMR experiment |
| ||||||||||||||||
| NMR details | Text: This structure was determined using standard 2D homonuclear technique |
- Sample preparation
Sample preparation
| Details | Contents: 0.5mM protein concentration; 50mM phosphate buffer; 90% H2O, 10% D2O Solvent system: 90% H2O/10% D2O |
|---|---|
| Sample conditions | Ionic strength: 50mm phosphate buffer / pH: 6.1 / Pressure: Ambient / Temperature: 298 K |
-NMR measurement
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M |
|---|---|
| Radiation wavelength | Relative weight: 1 |
| NMR spectrometer | Type: Bruker AVANCE / Manufacturer: Bruker / Model: AVANCE / Field strength: 800 MHz |
- Processing
Processing
| NMR software |
| ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method: Torsion Angle Dynamics, Simulated Annealing, Energy Restrained Minimization Software ordinal: 1 | ||||||||||||||||
| NMR representative | Selection criteria: lowest energy | ||||||||||||||||
| NMR ensemble | Conformer selection criteria: structures with the least restraint violations, structures with the lowest energy Conformers calculated total number: 40 / Conformers submitted total number: 20 |
 Movie
Movie Controller
Controller

















 PDBj
PDBj


 NMRPipe
NMRPipe