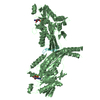
Entry Database : PDB / ID : 1jmoTitle Crystal Structure of the Heparin Cofactor II-S195A Thrombin Complex HEPARIN COFACTOR II Thrombin, heavy chain Thrombin, light chain Keywords / / / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Homo sapiens (human)Method / / / Resolution : 2.2 Å Authors Baglin, T.P. / Carrell, R.W. / Esmon, C.T. / Huntington, J.A. Journal : Proc.Natl.Acad.Sci.USA / Year : 2002Title : Crystal structures of native and thrombin-complexed heparin cofactor II reveal a multistep allosteric mechanism.Authors : Baglin, T.P. / Carrell, R.W. / Church, F.C. / Esmon, C.T. / Huntington, J.A. History Deposition Jul 19, 2001 Deposition site / Processing site Revision 1.0 Aug 30, 2002 Provider / Type Revision 1.1 Apr 27, 2008 Group Revision 1.2 Jul 13, 2011 Group / Version format complianceRevision 2.0 Jul 29, 2020 Group Advisory / Atomic model ... Advisory / Atomic model / Data collection / Database references / Derived calculations / Non-polymer description / Structure summary Category atom_site / chem_comp ... atom_site / chem_comp / database_PDB_caveat / entity / pdbx_branch_scheme / pdbx_chem_comp_identifier / pdbx_entity_branch / pdbx_entity_branch_descriptor / pdbx_entity_branch_link / pdbx_entity_branch_list / pdbx_entity_nonpoly / pdbx_nonpoly_scheme / pdbx_struct_assembly_gen / pdbx_struct_conn_angle / pdbx_unobs_or_zero_occ_atoms / pdbx_unobs_or_zero_occ_residues / pdbx_validate_chiral / struct_asym / struct_conn / struct_ref_seq_dif / struct_site / struct_site_gen Item _atom_site.B_iso_or_equiv / _atom_site.Cartn_x ... _atom_site.B_iso_or_equiv / _atom_site.Cartn_x / _atom_site.Cartn_y / _atom_site.Cartn_z / _atom_site.auth_asym_id / _atom_site.auth_atom_id / _atom_site.auth_comp_id / _atom_site.label_asym_id / _atom_site.label_atom_id / _atom_site.label_comp_id / _atom_site.label_entity_id / _atom_site.type_symbol / _chem_comp.formula / _chem_comp.formula_weight / _chem_comp.id / _chem_comp.mon_nstd_flag / _chem_comp.name / _chem_comp.type / _entity.formula_weight / _entity.pdbx_description / _entity.pdbx_number_of_molecules / _entity.src_method / _entity.type / _pdbx_struct_assembly_gen.asym_id_list / _pdbx_struct_conn_angle.ptnr2_label_asym_id / _pdbx_struct_conn_angle.ptnr3_label_asym_id / _struct_conn.pdbx_dist_value / _struct_conn.pdbx_leaving_atom_flag / _struct_conn.pdbx_role / _struct_conn.ptnr1_auth_asym_id / _struct_conn.ptnr1_auth_comp_id / _struct_conn.ptnr1_auth_seq_id / _struct_conn.ptnr1_label_asym_id / _struct_conn.ptnr1_label_atom_id / _struct_conn.ptnr1_label_comp_id / _struct_conn.ptnr1_label_seq_id / _struct_conn.ptnr2_auth_asym_id / _struct_conn.ptnr2_auth_comp_id / _struct_conn.ptnr2_auth_seq_id / _struct_conn.ptnr2_label_asym_id / _struct_conn.ptnr2_label_atom_id / _struct_conn.ptnr2_label_comp_id / _struct_conn.ptnr2_label_seq_id / _struct_ref_seq_dif.details Description / Provider / Type Revision 2.1 Oct 27, 2021 Group / Database references / Structure summaryCategory chem_comp / database_2 ... chem_comp / database_2 / pdbx_unobs_or_zero_occ_atoms / pdbx_unobs_or_zero_occ_residues / struct_ref_seq_dif Item _chem_comp.pdbx_synonyms / _database_2.pdbx_DOI ... _chem_comp.pdbx_synonyms / _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _struct_ref_seq_dif.details Revision 2.2 Apr 3, 2024 Group / Refinement descriptionCategory / chem_comp_bond / pdbx_initial_refinement_modelRevision 2.3 Oct 30, 2024 Group / Category / pdbx_modification_feature
Show all Show less
 Yorodumi
Yorodumi Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords Function and homology information
Function and homology information Homo sapiens (human)
Homo sapiens (human) X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.2 Å
MOLECULAR REPLACEMENT / Resolution: 2.2 Å  Authors
Authors Citation
Citation Journal: Proc.Natl.Acad.Sci.USA / Year: 2002
Journal: Proc.Natl.Acad.Sci.USA / Year: 2002 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 1jmo.cif.gz
1jmo.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb1jmo.ent.gz
pdb1jmo.ent.gz PDB format
PDB format 1jmo.json.gz
1jmo.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/jm/1jmo
https://data.pdbj.org/pub/pdb/validation_reports/jm/1jmo ftp://data.pdbj.org/pub/pdb/validation_reports/jm/1jmo
ftp://data.pdbj.org/pub/pdb/validation_reports/jm/1jmo Links
Links Assembly
Assembly
 Components
Components Homo sapiens (human) / Cell line (production host): BHK culture / Organ (production host): kidney / Production host:
Homo sapiens (human) / Cell line (production host): BHK culture / Organ (production host): kidney / Production host: 
 Homo sapiens (human) / Cell line (production host): BHK culture / Organ (production host): kidney / Production host:
Homo sapiens (human) / Cell line (production host): BHK culture / Organ (production host): kidney / Production host: 
 Homo sapiens (human) / Tissue fraction: blood plasma / References: UniProt: P05546
Homo sapiens (human) / Tissue fraction: blood plasma / References: UniProt: P05546





 X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  SRS
SRS  / Beamline: PX14.1 / Wavelength: 1.488 Å
/ Beamline: PX14.1 / Wavelength: 1.488 Å Processing
Processing MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller












 PDBj
PDBj












