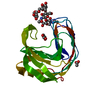
登録情報 データベース : PDB / ID : 1pz8タイトル Modulation of agrin function by alternative splicing and Ca2+ binding Agrin キーワード / 機能・相同性 分子機能 ドメイン・相同性 構成要素
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 Gallus gallus (ニワトリ)手法 / / / 解像度 : 2.35 Å データ登録者 Stetefeld, J. / Alexandrescu, A.T. / Maciejewski, M.W. / Jenny, M. / Rathgeb-Szabo, K. / Schulthess, T. / Landwehr, R. / Frank, S. / Ruegg, M.A. / Kammerer, R.A. ジャーナル : STRUCTURE / 年 : 2004タイトル : Modulation of agrin function by alternative splicing and Ca2+ binding.著者 : Stetefeld, J. / Alexandrescu, A.T. / Maciejewski, M.W. / Jenny, M. / Rathgeb-Szabo, K. / Schulthess, T. / Landwehr, R. / Frank, S. / Ruegg, M.A. / Kammerer, R.A. 履歴 登録 2003年7月10日 登録サイト / 処理サイト 改定 1.0 2004年4月13日 Provider / タイプ 改定 1.1 2008年4月29日 Group 改定 1.2 2011年7月13日 Group 改定 1.3 2024年11月6日 Group Data collection / Database references ... Data collection / Database references / Derived calculations / Structure summary カテゴリ chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / diffrn_source / pdbx_entry_details / pdbx_modification_feature / pdbx_struct_conn_angle / struct_conn / struct_site Item _database_2.pdbx_DOI / _database_2.pdbx_database_accession ... _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _diffrn_source.pdbx_synchrotron_site / _pdbx_struct_conn_angle.ptnr1_auth_comp_id / _pdbx_struct_conn_angle.ptnr1_auth_seq_id / _pdbx_struct_conn_angle.ptnr1_label_asym_id / _pdbx_struct_conn_angle.ptnr1_label_atom_id / _pdbx_struct_conn_angle.ptnr1_label_comp_id / _pdbx_struct_conn_angle.ptnr1_label_seq_id / _pdbx_struct_conn_angle.ptnr3_auth_comp_id / _pdbx_struct_conn_angle.ptnr3_auth_seq_id / _pdbx_struct_conn_angle.ptnr3_label_asym_id / _pdbx_struct_conn_angle.ptnr3_label_atom_id / _pdbx_struct_conn_angle.ptnr3_label_comp_id / _pdbx_struct_conn_angle.ptnr3_label_seq_id / _pdbx_struct_conn_angle.value / _struct_conn.pdbx_dist_value / _struct_conn.ptnr1_auth_comp_id / _struct_conn.ptnr1_auth_seq_id / _struct_conn.ptnr1_label_asym_id / _struct_conn.ptnr1_label_atom_id / _struct_conn.ptnr1_label_comp_id / _struct_conn.ptnr1_label_seq_id / _struct_conn.ptnr2_auth_comp_id / _struct_conn.ptnr2_auth_seq_id / _struct_conn.ptnr2_label_asym_id / _struct_conn.ptnr2_label_atom_id / _struct_conn.ptnr2_label_comp_id / _struct_conn.ptnr2_label_seq_id / _struct_site.pdbx_auth_asym_id / _struct_site.pdbx_auth_comp_id / _struct_site.pdbx_auth_seq_id
すべて表示 表示を減らす Remark 999 SEQUENCE the c-terminal agrin domain has alternative splice variants, we solved the structure of ... SEQUENCE the c-terminal agrin domain has alternative splice variants, we solved the structure of B11 (1pz7) and B8 with (1pz8) and without (1pz9) calcium, therefore the sequence-file conatins either 11 or 8 residues more than the so called B0-insert, the insert starts at TKS- and ends at EKA, B11:PDALDYPAEPS B8:HLSNEIPA however, not all the splice-residues are detectable in the electron density map
 データを開く
データを開く 基本情報
基本情報 要素
要素 キーワード
キーワード 機能・相同性情報
機能・相同性情報
 X線回折 /
X線回折 /  シンクロトロン /
シンクロトロン /  分子置換 / 解像度: 2.35 Å
分子置換 / 解像度: 2.35 Å  データ登録者
データ登録者 引用
引用 ジャーナル: STRUCTURE / 年: 2004
ジャーナル: STRUCTURE / 年: 2004 構造の表示
構造の表示 Molmil
Molmil Jmol/JSmol
Jmol/JSmol ダウンロードとリンク
ダウンロードとリンク ダウンロード
ダウンロード 1pz8.cif.gz
1pz8.cif.gz PDBx/mmCIF形式
PDBx/mmCIF形式 pdb1pz8.ent.gz
pdb1pz8.ent.gz PDB形式
PDB形式 1pz8.json.gz
1pz8.json.gz PDBx/mmJSON形式
PDBx/mmJSON形式 その他のダウンロード
その他のダウンロード 1pz8_validation.pdf.gz
1pz8_validation.pdf.gz wwPDB検証レポート
wwPDB検証レポート 1pz8_full_validation.pdf.gz
1pz8_full_validation.pdf.gz 1pz8_validation.xml.gz
1pz8_validation.xml.gz 1pz8_validation.cif.gz
1pz8_validation.cif.gz https://data.pdbj.org/pub/pdb/validation_reports/pz/1pz8
https://data.pdbj.org/pub/pdb/validation_reports/pz/1pz8 ftp://data.pdbj.org/pub/pdb/validation_reports/pz/1pz8
ftp://data.pdbj.org/pub/pdb/validation_reports/pz/1pz8 リンク
リンク 集合体
集合体




 要素
要素

 X線回折
X線回折 試料調製
試料調製 シンクロトロン / サイト:
シンクロトロン / サイト:  EMBL/DESY, HAMBURG
EMBL/DESY, HAMBURG  / ビームライン: BW7B / 波長: 1 Å
/ ビームライン: BW7B / 波長: 1 Å 解析
解析 分子置換 / 解像度: 2.35→20 Å / Cor.coef. Fo:Fc: 0.926 / Cor.coef. Fo:Fc free: 0.894 / SU B: 12.738 / SU ML: 0.309 / 交差検証法: THROUGHOUT / σ(F): 0 / ESU R: 0.689 / ESU R Free: 0.291 / 立体化学のターゲット値: MAXIMUM LIKELIHOOD / 詳細: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
分子置換 / 解像度: 2.35→20 Å / Cor.coef. Fo:Fc: 0.926 / Cor.coef. Fo:Fc free: 0.894 / SU B: 12.738 / SU ML: 0.309 / 交差検証法: THROUGHOUT / σ(F): 0 / ESU R: 0.689 / ESU R Free: 0.291 / 立体化学のターゲット値: MAXIMUM LIKELIHOOD / 詳細: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS ムービー
ムービー コントローラー
コントローラー














 PDBj
PDBj







