+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1bzs | ||||||
|---|---|---|---|---|---|---|---|
| Title | CRYSTAL STRUCTURE OF MMP8 COMPLEXED WITH HMR2909 | ||||||
 Components Components | NEUTROPHIL COLLAGENASE | ||||||
 Keywords Keywords | HYDROLASE / METALLO PROTEINASE / HYDROXAMATE / MATRIX DEGRADATION | ||||||
| Function / homology |  Function and homology information Function and homology informationneutrophil collagenase / tumor necrosis factor binding / positive regulation of microglial cell activation / positive regulation of neuroinflammatory response / positive regulation of tumor necrosis factor-mediated signaling pathway / Activation of Matrix Metalloproteinases / endodermal cell differentiation / Collagen degradation / collagen catabolic process / extracellular matrix disassembly ...neutrophil collagenase / tumor necrosis factor binding / positive regulation of microglial cell activation / positive regulation of neuroinflammatory response / positive regulation of tumor necrosis factor-mediated signaling pathway / Activation of Matrix Metalloproteinases / endodermal cell differentiation / Collagen degradation / collagen catabolic process / extracellular matrix disassembly / Degradation of the extracellular matrix / extracellular matrix organization / metalloendopeptidase activity / specific granule lumen / positive regulation of tumor necrosis factor production / tertiary granule lumen / peptidase activity / : / cellular response to lipopolysaccharide / endopeptidase activity / serine-type endopeptidase activity / Neutrophil degranulation / proteolysis / extracellular space / extracellular region / zinc ion binding Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 1.7 Å MOLECULAR REPLACEMENT / Resolution: 1.7 Å | ||||||
 Authors Authors | Schreuder, H. / Brachvogel, V. / Loenze, P. | ||||||
 Citation Citation |  Journal: J.Med.Chem. / Year: 1999 Journal: J.Med.Chem. / Year: 1999Title: Quantitative structure-activity relationship of human neutrophil collagenase (MMP-8) inhibitors using comparative molecular field analysis and X-ray structure analysis. Authors: Matter, H. / Schwab, W. / Barbier, D. / Billen, G. / Haase, B. / Neises, B. / Schudok, M. / Thorwart, W. / Schreuder, H. / Brachvogel, V. / Lonze, P. / Weithmann, K.U. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1bzs.cif.gz 1bzs.cif.gz | 55 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1bzs.ent.gz pdb1bzs.ent.gz | 36.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1bzs.json.gz 1bzs.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  1bzs_validation.pdf.gz 1bzs_validation.pdf.gz | 710.2 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  1bzs_full_validation.pdf.gz 1bzs_full_validation.pdf.gz | 711.1 KB | Display | |
| Data in XML |  1bzs_validation.xml.gz 1bzs_validation.xml.gz | 11.3 KB | Display | |
| Data in CIF |  1bzs_validation.cif.gz 1bzs_validation.cif.gz | 16.3 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/bz/1bzs https://data.pdbj.org/pub/pdb/validation_reports/bz/1bzs ftp://data.pdbj.org/pub/pdb/validation_reports/bz/1bzs ftp://data.pdbj.org/pub/pdb/validation_reports/bz/1bzs | HTTPS FTP |
-Related structure data
| Related structure data |  1janS S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 1 molecules A
| #1: Protein | Mass: 18374.008 Da / Num. of mol.: 1 / Fragment: CATALYTIC DOMAIN Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Cell: NEUTROPHIL / Plasmid: PET / Species (production host): Escherichia coli / Production host: Homo sapiens (human) / Cell: NEUTROPHIL / Plasmid: PET / Species (production host): Escherichia coli / Production host:  |
|---|
-Non-polymers , 5 types, 214 molecules 

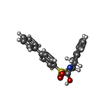






| #2: Chemical | | #3: Chemical | #4: Chemical | ChemComp-BSI / | #5: Chemical | ChemComp-EPE / | #6: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.16 Å3/Da / Density % sol: 43 % | ||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | Temperature: 298 K / Method: vapor diffusion, hanging drop / pH: 6 Details: PROTEIN SOLUTION: 10 MG/ML MMP8 IN 25 MM MES, 100 MM NACL, 20 MM CACL2, 0.1 MM ZNCL2, PH 6.0, WITH THREEFOLD EXCESS INHIBITOR IN DMF ADDED. RESERVOIR SOLUTION: 10-25% (W/W) PEG6000. CRYSTALS ...Details: PROTEIN SOLUTION: 10 MG/ML MMP8 IN 25 MM MES, 100 MM NACL, 20 MM CACL2, 0.1 MM ZNCL2, PH 6.0, WITH THREEFOLD EXCESS INHIBITOR IN DMF ADDED. RESERVOIR SOLUTION: 10-25% (W/W) PEG6000. CRYSTALS APPEAR IN ABOUT 1 WEEK., VAPOR DIFFUSION, HANGING DROP, temperature 298K | ||||||||||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS | ||||||||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: ENRAF-NONIUS FR571 / Wavelength: 1.5418 ROTATING ANODE / Type: ENRAF-NONIUS FR571 / Wavelength: 1.5418 |
| Detector | Type: MARRESEARCH / Detector: IMAGE PLATE / Date: Aug 29, 1997 / Details: COLLIMATOR |
| Radiation | Monochromator: GRAPHITE / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.5418 Å / Relative weight: 1 |
| Reflection | Resolution: 1.7→20 Å / Num. obs: 18344 / % possible obs: 99.8 % / Redundancy: 7 % / Rsym value: 4.8 / Net I/σ(I): 26.9 |
| Reflection shell | Resolution: 1.7→2 Å / Redundancy: 7.3 % / Mean I/σ(I) obs: 17.2 / Rsym value: 9.6 / % possible all: 99.7 |
| Reflection | *PLUS Num. measured all: 129143 / Rmerge(I) obs: 0.048 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 1JAN Resolution: 1.7→8 Å / Data cutoff high absF: 10000000 / Data cutoff low absF: 0.1 / Isotropic thermal model: RESTRAINED / σ(F): 0
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 13.3 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.7→8 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 1.7→1.78 Å / Total num. of bins used: 8 /
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Xplor file |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Software | *PLUS Name:  X-PLOR / Version: 3.1 / Classification: refinement X-PLOR / Version: 3.1 / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS Highest resolution: 1.7 Å / Lowest resolution: 8 Å / σ(F): 0 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS Biso mean: 13.3 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | *PLUS Highest resolution: 1.7 Å / Rfactor Rwork: 0.273 |
 Movie
Movie Controller
Controller













 PDBj
PDBj








