+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-9137 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of NLRC4-CARD filament | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | NLRC4 / Helical assembly / Inflammasome / PROTEIN FIBRIL | |||||||||
| Function / homology |  Function and homology information Function and homology informationIPAF inflammasome complex / The IPAF inflammasome / icosanoid biosynthetic process / canonical inflammasome complex / caspase binding / positive regulation of protein processing / pattern recognition receptor signaling pathway / TP53 Regulates Transcription of Caspase Activators and Caspases / pyroptotic inflammatory response / detection of bacterium ...IPAF inflammasome complex / The IPAF inflammasome / icosanoid biosynthetic process / canonical inflammasome complex / caspase binding / positive regulation of protein processing / pattern recognition receptor signaling pathway / TP53 Regulates Transcription of Caspase Activators and Caspases / pyroptotic inflammatory response / detection of bacterium / endopeptidase activator activity / activation of innate immune response / bioluminescence / : / positive regulation of interleukin-1 beta production / generation of precursor metabolites and energy / protein homooligomerization / positive regulation of inflammatory response / defense response to bacterium / positive regulation of apoptotic process / inflammatory response / innate immune response / apoptotic process / magnesium ion binding / protein homodimerization activity / ATP binding / identical protein binding / plasma membrane / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) / Homo sapiens (human) /  Vaccinia virus Vaccinia virus | |||||||||
| Method | helical reconstruction / cryo EM / Resolution: 3.4 Å | |||||||||
 Authors Authors | Zheng W / Matyszewski M | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: J Biol Chem / Year: 2018 Journal: J Biol Chem / Year: 2018Title: Cryo-EM structure of the NLRC4 filament provides insights into how symmetric and asymmetric supramolecular structures drive inflammasome assembly. Authors: Mariusz Matyszewski / Weili Zheng / Jacob Lueck / Brendan Antiochos / Edward H Egelman / Jungsan Sohn /  Abstract: Inflammasomes are supramolecular signaling platforms integral to innate immune defense against invading pathogens. The NOD-like receptor (NLR) family apoptosis inhibitory protein (NAIP)·NLR family ...Inflammasomes are supramolecular signaling platforms integral to innate immune defense against invading pathogens. The NOD-like receptor (NLR) family apoptosis inhibitory protein (NAIP)·NLR family caspase-recruiting domain (CARD) domain-containing 4 (NLRC4) inflammasome recognizes intracellular bacteria and induces the polymerization of the caspase-1 protease, which in turn executes maturation of interleukin-1β (IL-1β) and pyroptosis. Several high-resolution structures of the fully assembled NAIP·NLRC4 complex are available, but these structures do not resolve the architecture of the CARD filament in atomic detail. Here, we present the cryo-EM structure of the filament assembled by the CARD of human NLRC4 (NLRC4) at 3.4 Å resolution. The structure revealed that the helical architecture of the NLRC4 filament is essentially identical to that of the downstream filament assembled by the CARD of caspase-1 (casp1), but deviates from the split washer-like assembly of the NAIP·NLRC4 oligomer. Our results suggest that architectural complementarity is a major driver for the recognition between upstream and downstream CARD assemblies in inflammasomes. Furthermore, a Monte Carlo simulation of the NLRC4 filament assembly rationalized why an (un)decameric NLRC4 oligomer is optimal for assembling the helical base of the NLRC4 filament. Together, our results explain how symmetric and asymmetric supramolecular assemblies enable high-fidelity signaling in inflammasomes. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_9137.map.gz emd_9137.map.gz | 5.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-9137-v30.xml emd-9137-v30.xml emd-9137.xml emd-9137.xml | 11.5 KB 11.5 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_9137.png emd_9137.png | 185.2 KB | ||
| Filedesc metadata |  emd-9137.cif.gz emd-9137.cif.gz | 5.9 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-9137 http://ftp.pdbj.org/pub/emdb/structures/EMD-9137 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9137 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9137 | HTTPS FTP |
-Related structure data
| Related structure data |  6mksMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_9137.map.gz / Format: CCP4 / Size: 8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_9137.map.gz / Format: CCP4 / Size: 8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.32 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
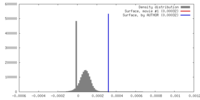
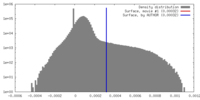
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : NLRC4-CARD filament
| Entire | Name: NLRC4-CARD filament |
|---|---|
| Components |
|
-Supramolecule #1: NLRC4-CARD filament
| Supramolecule | Name: NLRC4-CARD filament / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Chimera protein of NLR family CARD domain-containing protein 4 an...
| Macromolecule | Name: Chimera protein of NLR family CARD domain-containing protein 4 and EGFP type: protein_or_peptide / ID: 1 / Number of copies: 31 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Vaccinia virus Vaccinia virus |
| Molecular weight | Theoretical: 38.668805 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GTGMNFIKDN SRALIQRMGM TVIKQITDDL FVWNVLNREE VNIICCEKVE QDAARGIIHM ILKKGSESCN LFLKSLKEWN YPLFQDLNG QSLFHQTSEG KLMVSKGEEL FTGVVPILVE LDGDVNGHKF SVSGEGEGDA TYGKLTLKFI CTTGKLPVPW P TLVTTLTY ...String: GTGMNFIKDN SRALIQRMGM TVIKQITDDL FVWNVLNREE VNIICCEKVE QDAARGIIHM ILKKGSESCN LFLKSLKEWN YPLFQDLNG QSLFHQTSEG KLMVSKGEEL FTGVVPILVE LDGDVNGHKF SVSGEGEGDA TYGKLTLKFI CTTGKLPVPW P TLVTTLTY GVQCFSRYPD HMKQHDFFKS AMPEGYVQER TIFFKDDGNY KTRAEVKFEG DTLVNRIELK GIDFKEDGNI LG HKLEYNY NSHNVYIMAD KQKNGIKANF KIRHNIEDGS VQLADHYQQN TPIGDGPVLL PDNHYLSTQS ALSKDPNEKR DHM VLLEFV TAAGITLEHH HHHH UniProtKB: NLR family CARD domain-containing protein 4, EGFP |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | helical reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Buffer | pH: 7 Details: 20mM HEPES at pH 7.4, 400mM NaCl, 10% glycerol, 1mM EDTA and 1mM DTT |
|---|---|
| Grid | Material: COPPER / Pretreatment - Type: GLOW DISCHARGE |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Number real images: 1690 / Average exposure time: 12.0 sec. / Average electron dose: 42.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm |
| Sample stage | Cooling holder cryogen: NITROGEN |
- Image processing
Image processing
| Final reconstruction | Applied symmetry - Helical parameters - Δz: 5.0 Å Applied symmetry - Helical parameters - Δ&Phi: 100.6 ° Applied symmetry - Helical parameters - Axial symmetry: C1 (asymmetric) Algorithm: BACK PROJECTION / Resolution.type: BY AUTHOR / Resolution: 3.4 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: SPIDER / Number images used: 299537 |
|---|---|
| Segment selection | Number selected: 402078 / Software - Name: EMAN2 |
| Startup model | Type of model: OTHER / Details: Featureless cylinder |
| Final angle assignment | Type: NOT APPLICABLE |
 Movie
Movie Controller
Controller























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)