[English] 日本語
 Yorodumi
Yorodumi- EMDB-4361: Cryo-EM density of heteromeric mLRRC8A/C volume-regulated anion c... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-4361 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
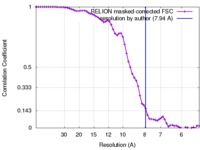
| Title | Cryo-EM density of heteromeric mLRRC8A/C volume-regulated anion channel at 7.94 A resolution | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 7.94 Å | |||||||||
 Authors Authors | Sawicka M / Deneka D / Lam AKM / Paulino C / Dutzler R | |||||||||
 Citation Citation |  Journal: Nature / Year: 2018 Journal: Nature / Year: 2018Title: Structure of a volume-regulated anion channel of the LRRC8 family. Authors: Dawid Deneka / Marta Sawicka / Andy K M Lam / Cristina Paulino / Raimund Dutzler /   Abstract: Volume-regulated anion channels are activated in response to hypotonic stress. These channels are composed of closely related paralogues of the leucine-rich repeat-containing protein 8 (LRRC8) family ...Volume-regulated anion channels are activated in response to hypotonic stress. These channels are composed of closely related paralogues of the leucine-rich repeat-containing protein 8 (LRRC8) family that co-assemble to form hexameric complexes. Here, using cryo-electron microscopy and X-ray crystallography, we determine the structure of a homomeric channel of the obligatory subunit LRRC8A. This protein conducts ions and has properties in common with endogenous heteromeric channels. Its modular structure consists of a transmembrane pore domain followed by a cytoplasmic leucine-rich repeat domain. The transmembrane domain, which is structurally related to connexin proteins, is wide towards the cytoplasm but constricted on the outside by a structural unit that acts as a selectivity filter. An excess of basic residues in the filter and throughout the pore attracts anions by electrostatic interaction. Our work reveals the previously unknown architecture of volume-regulated anion channels and their mechanism of selective anion conduction. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_4361.map.gz emd_4361.map.gz | 92.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-4361-v30.xml emd-4361-v30.xml emd-4361.xml emd-4361.xml | 10.9 KB 10.9 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_4361_fsc.xml emd_4361_fsc.xml | 6.2 KB | Display |  FSC data file FSC data file |
| Images |  emd_4361.png emd_4361.png | 11.4 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-4361 http://ftp.pdbj.org/pub/emdb/structures/EMD-4361 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4361 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4361 | HTTPS FTP |
-Related structure data
| Related structure data |  4362C  4366C  4367C  6fnwC  6g8zC  6g9lC  6g9oC C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_4361.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_4361.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.075 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Heterohexamer of mLRRC8A and mLRRC8C
| Entire | Name: Heterohexamer of mLRRC8A and mLRRC8C |
|---|---|
| Components |
|
-Supramolecule #1: Heterohexamer of mLRRC8A and mLRRC8C
| Supramolecule | Name: Heterohexamer of mLRRC8A and mLRRC8C / type: complex / ID: 1 / Parent: 0 |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  Homo sapiens (human) / Recombinant cell: HEK293S Homo sapiens (human) / Recombinant cell: HEK293S |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 3.5 mg/mL | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 8.5 Component:
| ||||||||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: GOLD / Mesh: 200 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Atmosphere: AIR | ||||||||||||
| Vitrification | Cryogen name: ETHANE-PROPANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV / Details: 2.5 ul sample volume, 3-7 s blotting time. |
- Electron microscopy
Electron microscopy
| Microscope | FEI POLARA 300 |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Digitization - Frames/image: 1-50 / Number real images: 1545 / Average exposure time: 12.5 sec. / Average electron dose: 1.2 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.0 mm / Nominal defocus max: 3.2 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 37313 |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Tecnai Polara / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller










 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)