[English] 日本語
 Yorodumi
Yorodumi- EMDB-4366: Cryo-EM density of homomeric mLRRC8A volume-regulated anion chann... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-4366 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
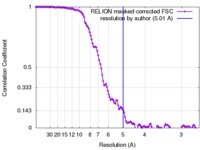
| Title | Cryo-EM density of homomeric mLRRC8A volume-regulated anion channel at 5.01 A resolution | |||||||||
 Map data Map data | None | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Chloride channel / Swelling-activated / VSOAC / Leucine-rich repeat / MEMBRANE PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationMiscellaneous transport and binding events / pre-B cell differentiation / volume-sensitive anion channel activity / aspartate transmembrane transport / cyclic-GMP-AMP transmembrane transporter activity / cyclic-GMP-AMP transmembrane import across plasma membrane / taurine transmembrane transport / monoatomic anion transmembrane transport / cell volume homeostasis / monoatomic anion transport ...Miscellaneous transport and binding events / pre-B cell differentiation / volume-sensitive anion channel activity / aspartate transmembrane transport / cyclic-GMP-AMP transmembrane transporter activity / cyclic-GMP-AMP transmembrane import across plasma membrane / taurine transmembrane transport / monoatomic anion transmembrane transport / cell volume homeostasis / monoatomic anion transport / protein hexamerization / response to osmotic stress / monoatomic ion channel complex / positive regulation of myoblast differentiation / intracellular glucose homeostasis / chloride transmembrane transport / positive regulation of insulin secretion / spermatogenesis / lysosomal membrane / cell surface / membrane / identical protein binding / plasma membrane / cytoplasm Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 5.01 Å | |||||||||
 Authors Authors | Sawicka M / Deneka D | |||||||||
| Funding support |  Switzerland, 1 items Switzerland, 1 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2018 Journal: Nature / Year: 2018Title: Structure of a volume-regulated anion channel of the LRRC8 family. Authors: Dawid Deneka / Marta Sawicka / Andy K M Lam / Cristina Paulino / Raimund Dutzler /   Abstract: Volume-regulated anion channels are activated in response to hypotonic stress. These channels are composed of closely related paralogues of the leucine-rich repeat-containing protein 8 (LRRC8) family ...Volume-regulated anion channels are activated in response to hypotonic stress. These channels are composed of closely related paralogues of the leucine-rich repeat-containing protein 8 (LRRC8) family that co-assemble to form hexameric complexes. Here, using cryo-electron microscopy and X-ray crystallography, we determine the structure of a homomeric channel of the obligatory subunit LRRC8A. This protein conducts ions and has properties in common with endogenous heteromeric channels. Its modular structure consists of a transmembrane pore domain followed by a cytoplasmic leucine-rich repeat domain. The transmembrane domain, which is structurally related to connexin proteins, is wide towards the cytoplasm but constricted on the outside by a structural unit that acts as a selectivity filter. An excess of basic residues in the filter and throughout the pore attracts anions by electrostatic interaction. Our work reveals the previously unknown architecture of volume-regulated anion channels and their mechanism of selective anion conduction. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_4366.map.gz emd_4366.map.gz | 95.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-4366-v30.xml emd-4366-v30.xml emd-4366.xml emd-4366.xml | 13.5 KB 13.5 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_4366_fsc.xml emd_4366_fsc.xml | 12.3 KB | Display |  FSC data file FSC data file |
| Images |  emd_4366.png emd_4366.png | 33.3 KB | ||
| Filedesc metadata |  emd-4366.cif.gz emd-4366.cif.gz | 5.9 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-4366 http://ftp.pdbj.org/pub/emdb/structures/EMD-4366 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4366 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4366 | HTTPS FTP |
-Related structure data
| Related structure data |  6g9lMC  4361C  4362C  4367C  6fnwC  6g8zC  6g9oC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_4366.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_4366.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | None | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.075 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Homohexameric mLRRC8A
| Entire | Name: Homohexameric mLRRC8A |
|---|---|
| Components |
|
-Supramolecule #1: Homohexameric mLRRC8A
| Supramolecule | Name: Homohexameric mLRRC8A / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Volume-regulated anion channel subunit LRRC8A
| Macromolecule | Name: Volume-regulated anion channel subunit LRRC8A / type: protein_or_peptide / ID: 1 / Number of copies: 6 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 94.239383 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MIPVTELRYF ADTQPAYRIL KPWWDVFTDY ISIVMLMIAV FGGTLQVTQD KMICLPCKWV TKDSCNDSFR GWAASSPEPT YPNSTVLPT PDTGPTGIKY DLDRHQYNYV DAVCYENRLH WFAKYFPYLV LLHTLIFLAC SNFWFKFPRT SSKLEHFVSI L LKCFDSPW ...String: MIPVTELRYF ADTQPAYRIL KPWWDVFTDY ISIVMLMIAV FGGTLQVTQD KMICLPCKWV TKDSCNDSFR GWAASSPEPT YPNSTVLPT PDTGPTGIKY DLDRHQYNYV DAVCYENRLH WFAKYFPYLV LLHTLIFLAC SNFWFKFPRT SSKLEHFVSI L LKCFDSPW TTRALSETVV EESDPKPAFS KMNGSMDKKS STVSEDVEAT VPMLQRTKSR IEQGIVDRSE TGVLDKKEGE QA KALFEKV KKFRTHVEEG DIVYRLYMRQ TIIKVIKFAL IICYTVYYVH NIKFDVDCTV DIESLTGYRT YRCAHPLATL FKI LASFYI SLVIFYGLIC MYTLWWMLRR SLKKYSFESI REESSYSDIP DVKNDFAFML HLIDQYDPLY SKRFAVFLSE VSEN KLRQL NLNNEWTLDK LRQRLTKNAQ DKLELHLFML SGIPDTVFDL VELEVLKLEL IPDVTIPPSI AQLTGLKELW LYHTA AKIE APALAFLREN LRALHIKFTD IKEIPLWIYS LKTLEELHLT GNLSAENNRY IVIDGLRELK RLKVLRLKSN LSKLPQ VVT DVGVHLQKLS INNEGTKLIV LNSLKKMVNL TELELIRCDL ERIPHSIFSL HNLQEIDLKD NNLKTIEEII SFQHLHR LT CLKLWYNHIA YIPIQIGNLT NLERLYLNRN KIEKIPTQLF YCRKLRYLDL SHNNLTFLPA DIGLLQNLQN LAVTANRI E ALPPELFQCR KLRALHLGNN VLQSLPSRVG ELTNLTQIEL RGNRLECLPV ELGECPLLKR SGLVVEEDLF STLPPEVKE RLWRADKEQA UniProtKB: Volume-regulated anion channel subunit LRRC8A |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 3.9 mg/mL | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 8.5 Component:
| ||||||||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: GOLD / Mesh: 200 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 30 sec. / Pretreatment - Atmosphere: AIR | ||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI POLARA 300 |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Digitization - Frames/image: 1-50 / Number real images: 9209 / Average exposure time: 12.5 sec. / Average electron dose: 1.2 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 37313 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.0 mm / Nominal defocus max: 3.2 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 37313 |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Tecnai Polara / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Protocol: RIGID BODY FIT |
|---|---|
| Output model |  PDB-6g9l: |
 Movie
Movie Controller
Controller















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)