+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: EMDB / ID: EMD-23949 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | The insulin receptor ectodomain in complex with a venom hybrid insulin analog - "head" region | |||||||||
 マップデータ マップデータ | ||||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | insulin / receptor / venom / cone snail / HORMONE / TOXIN | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報regulation of female gonad development / positive regulation of meiotic cell cycle / insulin-like growth factor II binding / positive regulation of developmental growth / male sex determination / insulin receptor complex / insulin-like growth factor I binding / positive regulation of protein-containing complex disassembly / insulin receptor activity / exocrine pancreas development ...regulation of female gonad development / positive regulation of meiotic cell cycle / insulin-like growth factor II binding / positive regulation of developmental growth / male sex determination / insulin receptor complex / insulin-like growth factor I binding / positive regulation of protein-containing complex disassembly / insulin receptor activity / exocrine pancreas development / dendritic spine maintenance / cargo receptor activity / insulin binding / adrenal gland development / neuronal cell body membrane / negative regulation of glycogen catabolic process / PTB domain binding / positive regulation of nitric oxide mediated signal transduction / negative regulation of feeding behavior / negative regulation of fatty acid metabolic process / Signaling by Insulin receptor / IRS activation / Insulin processing / regulation of protein secretion / positive regulation of peptide hormone secretion / positive regulation of respiratory burst / amyloid-beta clearance / negative regulation of acute inflammatory response / Regulation of gene expression in beta cells / alpha-beta T cell activation / positive regulation of receptor internalization / regulation of embryonic development / insulin receptor substrate binding / protein kinase activator activity / positive regulation of dendritic spine maintenance / Synthesis, secretion, and deacylation of Ghrelin / negative regulation of respiratory burst involved in inflammatory response / epidermis development / negative regulation of protein secretion / activation of protein kinase B activity / positive regulation of insulin receptor signaling pathway / negative regulation of gluconeogenesis / positive regulation of glycogen biosynthetic process / fatty acid homeostasis / Signal attenuation / FOXO-mediated transcription of oxidative stress, metabolic and neuronal genes / negative regulation of lipid catabolic process / heart morphogenesis / positive regulation of lipid biosynthetic process / regulation of protein localization to plasma membrane / transport across blood-brain barrier / negative regulation of oxidative stress-induced intrinsic apoptotic signaling pathway / phosphatidylinositol 3-kinase binding / nitric oxide-cGMP-mediated signaling / transport vesicle / COPI-mediated anterograde transport / positive regulation of nitric-oxide synthase activity / Insulin receptor recycling / negative regulation of reactive oxygen species biosynthetic process / insulin-like growth factor receptor binding / positive regulation of brown fat cell differentiation / NPAS4 regulates expression of target genes / dendrite membrane / endoplasmic reticulum-Golgi intermediate compartment membrane / neuron projection maintenance / positive regulation of mitotic nuclear division / Insulin receptor signalling cascade / receptor-mediated endocytosis / positive regulation of glycolytic process / positive regulation of cytokine production / positive regulation of long-term synaptic potentiation / endosome lumen / acute-phase response / positive regulation of protein secretion / positive regulation of D-glucose import / learning / insulin receptor binding / positive regulation of cell differentiation / Regulation of insulin secretion / wound healing / receptor protein-tyrosine kinase / negative regulation of protein catabolic process / positive regulation of neuron projection development / hormone activity / regulation of synaptic plasticity / caveola / cellular response to growth factor stimulus / receptor internalization / Golgi lumen / positive regulation of protein localization to nucleus / memory / cognition / vasodilation / male gonad development / cellular response to insulin stimulus / glucose metabolic process / positive regulation of nitric oxide biosynthetic process / late endosome / insulin receptor signaling pathway / glucose homeostasis 類似検索 - 分子機能 | |||||||||
| 生物種 |  Homo sapiens (ヒト) Homo sapiens (ヒト) | |||||||||
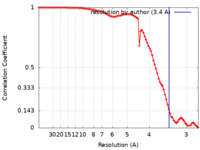
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 3.4 Å | |||||||||
 データ登録者 データ登録者 | Blakely AD / Xiong X | |||||||||
| 資金援助 | 1件
| |||||||||
 引用 引用 |  ジャーナル: Nat Chem Biol / 年: 2022 ジャーナル: Nat Chem Biol / 年: 2022タイトル: Symmetric and asymmetric receptor conformation continuum induced by a new insulin. 著者: Xiaochun Xiong / Alan Blakely / Jin Hwan Kim / John G Menting / Ingmar B Schäfer / Heidi L Schubert / Rahul Agrawal / Theresia Gutmann / Carlie Delaine / Yi Wolf Zhang / Gizem Olay Artik / ...著者: Xiaochun Xiong / Alan Blakely / Jin Hwan Kim / John G Menting / Ingmar B Schäfer / Heidi L Schubert / Rahul Agrawal / Theresia Gutmann / Carlie Delaine / Yi Wolf Zhang / Gizem Olay Artik / Allanah Merriman / Debbie Eckert / Michael C Lawrence / Ünal Coskun / Simon J Fisher / Briony E Forbes / Helena Safavi-Hemami / Christopher P Hill / Danny Hung-Chieh Chou /     要旨: Cone snail venoms contain a wide variety of bioactive peptides, including insulin-like molecules with distinct structural features, binding modes and biochemical properties. Here, we report an active ...Cone snail venoms contain a wide variety of bioactive peptides, including insulin-like molecules with distinct structural features, binding modes and biochemical properties. Here, we report an active humanized cone snail venom insulin with an elongated A chain and a truncated B chain, and use cryo-electron microscopy (cryo-EM) and protein engineering to elucidate its interactions with the human insulin receptor (IR) ectodomain. We reveal how an extended A chain can compensate for deletion of B-chain residues, which are essential for activity of human insulin but also compromise therapeutic utility by delaying dissolution from the site of subcutaneous injection. This finding suggests approaches to developing improved therapeutic insulins. Curiously, the receptor displays a continuum of conformations from the symmetric state to a highly asymmetric low-abundance structure that displays coordination of a single humanized venom insulin using elements from both of the previously characterized site 1 and site 2 interactions. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | EMマップ:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| 添付画像 |
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_23949.map.gz emd_23949.map.gz | 59.6 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-23949-v30.xml emd-23949-v30.xml emd-23949.xml emd-23949.xml | 18.9 KB 18.9 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_23949_fsc.xml emd_23949_fsc.xml | 8.9 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_23949.png emd_23949.png | 106.7 KB | ||
| マスクデータ |  emd_23949_msk_1.map emd_23949_msk_1.map | 64 MB |  マスクマップ マスクマップ | |
| Filedesc metadata |  emd-23949.cif.gz emd-23949.cif.gz | 6.6 KB | ||
| その他 |  emd_23949_half_map_1.map.gz emd_23949_half_map_1.map.gz emd_23949_half_map_2.map.gz emd_23949_half_map_2.map.gz | 59.3 MB 59.3 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-23949 http://ftp.pdbj.org/pub/emdb/structures/EMD-23949 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-23949 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-23949 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_23949_validation.pdf.gz emd_23949_validation.pdf.gz | 809.1 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_23949_full_validation.pdf.gz emd_23949_full_validation.pdf.gz | 808.6 KB | 表示 | |
| XML形式データ |  emd_23949_validation.xml.gz emd_23949_validation.xml.gz | 16.3 KB | 表示 | |
| CIF形式データ |  emd_23949_validation.cif.gz emd_23949_validation.cif.gz | 20.9 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-23949 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-23949 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-23949 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-23949 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  7mqoMC  7mqrC  7mqsC M: このマップから作成された原子モデル C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | |
| 電子顕微鏡画像生データ |  EMPIAR-10736 (タイトル: Cryo-EM of the human insulin receptor ectodomain in complex with an insulin analog with truncated B chain and enlongated A chain EMPIAR-10736 (タイトル: Cryo-EM of the human insulin receptor ectodomain in complex with an insulin analog with truncated B chain and enlongated A chainData size: 11.7 TB Data #1: Unaligned multi-frame micrographs (40 e-/A2 dose) [micrographs - multiframe] Data #2: unaligned micrographs (60 e-/A2 dose) [micrographs - multiframe]) |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_23949.map.gz / 形式: CCP4 / 大きさ: 64 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_23949.map.gz / 形式: CCP4 / 大きさ: 64 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.365 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
CCP4マップ ヘッダ情報:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-添付データ
-マスク #1
| ファイル |  emd_23949_msk_1.map emd_23949_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: #1
| ファイル | emd_23949_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: #2
| ファイル | emd_23949_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : Insulin receptor ectodomain in complex with insulin analog Vh-Ins...
| 全体 | 名称: Insulin receptor ectodomain in complex with insulin analog Vh-Ins-HSLQ - focused refinement. |
|---|---|
| 要素 |
|
-超分子 #1: Insulin receptor ectodomain in complex with insulin analog Vh-Ins...
| 超分子 | 名称: Insulin receptor ectodomain in complex with insulin analog Vh-Ins-HSLQ - focused refinement. タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: #1-#3 |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 209 KDa |
-分子 #1: Insulin A chain
| 分子 | 名称: Insulin A chain / タイプ: protein_or_peptide / ID: 1 / コピー数: 2 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 2.736106 KDa |
| 配列 | 文字列: GIVEQCCTSI CSLYQLENYC HSLQ UniProtKB: Insulin |
-分子 #2: Insulin B chain
| 分子 | 名称: Insulin B chain / タイプ: protein_or_peptide / ID: 2 / コピー数: 2 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 2.537951 KDa |
| 配列 | 文字列: FVNQHLCGSE LVEALYLVCL ER UniProtKB: Insulin |
-分子 #3: Isoform Short of Insulin receptor
| 分子 | 名称: Isoform Short of Insulin receptor / タイプ: protein_or_peptide / ID: 3 / コピー数: 2 / 光学異性体: LEVO / EC番号: receptor protein-tyrosine kinase |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 104.761875 KDa |
| 組換発現 | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 配列 | 文字列: HLYPGEVCPG MDIRNNLTRL HELENCSVIE GHLQILLMFK TRPEDFRDLS FPKLIMITDY LLLFRVYGLE SLKDLFPNLT VIRGSRLFF NYALVIFEMV HLKELGLYNL MNITRGSVRI EKNNELCYLA TIDWSRILDS VEDNYIVLNK DDNEECGDIC P GTAKGKTN ...文字列: HLYPGEVCPG MDIRNNLTRL HELENCSVIE GHLQILLMFK TRPEDFRDLS FPKLIMITDY LLLFRVYGLE SLKDLFPNLT VIRGSRLFF NYALVIFEMV HLKELGLYNL MNITRGSVRI EKNNELCYLA TIDWSRILDS VEDNYIVLNK DDNEECGDIC P GTAKGKTN CPATVINGQF VERCWTHSHC QKVCPTICKS HGCTAEGLCC HSECLGNCSQ PDDPTKCVAC RNFYLDGRCV ET CPPPYYH FQDWRCVNFS FCQDLHHKCK NSRRQGCHQY VIHNNKCIPE CPSGYTMNSS NLLCTPCLGP CPKVCHLLEG EKT IDSVTS AQELRGCTVI NGSLIINIRG GNNLAAELEA NLGLIEEISG YLKIRRSYAL VSLSFFRKLR LIRGETLEIG NYSF YALDN QNLRQLWDWS KHNLTITQGK LFFHYNPKLC LSEIHKMEEV SGTKGRQERN DIALKTNGDQ ASCENELLKF SYIRT SFDK ILLRWEPYWP PDFRDLLGFM LFYKEAPYQN VTEFDGQDAC GSNSWTVVDI DPPLRSNDPK SQNHPGWLMR GLKPWT QYA IFVKTLVTFS DERRTYGAKS DIIYVQTDAT NPSVPLDPIS VSNSSSQIIL KWKPPSDPNG NITHYLVFWE RQAEDSE LF ELDYCLKGLK LPSRTWSPPF ESEDSQKHNQ SEYEDSAGEC CSCPKTDSQI LKELEESSFR KTFEDYLHNV VFVPRPSR K RRSLGDVGNV TVAVPTVAAF PNTSSTSVPT SPEEHRPFEK VVNKESLVIS GLRHFTGYRI ELQACNQDTP EERCSVAAY VSARTMPEAK ADDIVGPVTH EIFENNVVHL MWQEPKEPNG LIVLYEVSYR RYGDEELHLC VSRKHFALER GCRLRGLSPG NYSVRIRAT SLAGNGSWTE PTYFYVTDYL DVPSNIAK UniProtKB: Insulin receptor |
-分子 #5: 2-acetamido-2-deoxy-beta-D-glucopyranose
| 分子 | 名称: 2-acetamido-2-deoxy-beta-D-glucopyranose / タイプ: ligand / ID: 5 / コピー数: 12 / 式: NAG |
|---|---|
| 分子量 | 理論値: 221.208 Da |
| Chemical component information |  ChemComp-NAG: |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 緩衝液 | pH: 8 / 構成要素: (名称: HEPES, Tris) 詳細: Equal parts HBS( 50 mM HEPES pH 7.5, 150 mM NaCl ) and TBS (25 mM Tris pH 8.5, 150 mM NaCl) |
|---|---|
| グリッド | モデル: Quantifoil R1.2/1.3 / 支持フィルム - 材質: CARBON / 支持フィルム - トポロジー: HOLEY |
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 80 % / チャンバー内温度: 277 K / 装置: FEI VITROBOT MARK II |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K2 SUMMIT (4k x 4k) 検出モード: COUNTING / 平均電子線量: 40.0 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
 ムービー
ムービー コントローラー
コントローラー




































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)