[English] 日本語
 Yorodumi
Yorodumi- EMDB-0233: Cryo-EM structure of the Trypanosoma brucei mitochondrial ribosom... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-0233 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the Trypanosoma brucei mitochondrial ribosome - This entry contains the head of the small mitoribosomal subunit | |||||||||
 Map data Map data | map of the head of the T. brucei mitoribosome small subunit | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | mitoribosome / translation / Trypanosoma / small ribosomal subunit / 9S rRNA / ribosomal protein / RIBOSOME | |||||||||
| Function / homology |  Function and homology information Function and homology informationmitochondrial mRNA editing complex / mitochondrial RNA processing / kinetoplast / thiosulfate sulfurtransferase activity / nuclear lumen / ciliary plasm / mRNA stabilization / mitochondrial small ribosomal subunit / RNA processing / mitochondrion organization ...mitochondrial mRNA editing complex / mitochondrial RNA processing / kinetoplast / thiosulfate sulfurtransferase activity / nuclear lumen / ciliary plasm / mRNA stabilization / mitochondrial small ribosomal subunit / RNA processing / mitochondrion organization / structural constituent of ribosome / translation / mRNA binding / mitochondrion / RNA binding / nucleoplasm / nucleus / cytoplasm Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.08 Å | |||||||||
 Authors Authors | Ramrath DJF / Niemann M | |||||||||
| Funding support |  Switzerland, 1 items Switzerland, 1 items
| |||||||||
 Citation Citation |  Journal: Science / Year: 2018 Journal: Science / Year: 2018Title: Evolutionary shift toward protein-based architecture in trypanosomal mitochondrial ribosomes. Authors: David J F Ramrath / Moritz Niemann / Marc Leibundgut / Philipp Bieri / Céline Prange / Elke K Horn / Alexander Leitner / Daniel Boehringer / André Schneider / Nenad Ban /  Abstract: Ribosomal RNA (rRNA) plays key functional and architectural roles in ribosomes. Using electron microscopy, we determined the atomic structure of a highly divergent ribosome found in mitochondria of , ...Ribosomal RNA (rRNA) plays key functional and architectural roles in ribosomes. Using electron microscopy, we determined the atomic structure of a highly divergent ribosome found in mitochondria of , a unicellular parasite that causes sleeping sickness in humans. The trypanosomal mitoribosome features the smallest rRNAs and contains more proteins than all known ribosomes. The structure shows how the proteins have taken over the role of architectural scaffold from the rRNA: They form an autonomous outer shell that surrounds the entire particle and stabilizes and positions the functionally important regions of the rRNA. Our results also reveal the "minimal" set of conserved rRNA and protein components shared by all ribosomes that help us define the most essential functional elements. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_0233.map.gz emd_0233.map.gz | 12.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-0233-v30.xml emd-0233-v30.xml emd-0233.xml emd-0233.xml | 51.3 KB 51.3 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_0233.png emd_0233.png | 66.2 KB | ||
| Filedesc metadata |  emd-0233.cif.gz emd-0233.cif.gz | 15.4 KB | ||
| Others |  emd_0233_half_map_1.map.gz emd_0233_half_map_1.map.gz emd_0233_half_map_2.map.gz emd_0233_half_map_2.map.gz | 97.9 MB 97.9 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-0233 http://ftp.pdbj.org/pub/emdb/structures/EMD-0233 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-0233 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-0233 | HTTPS FTP |
-Validation report
| Summary document |  emd_0233_validation.pdf.gz emd_0233_validation.pdf.gz | 365.3 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_0233_full_validation.pdf.gz emd_0233_full_validation.pdf.gz | 364.4 KB | Display | |
| Data in XML |  emd_0233_validation.xml.gz emd_0233_validation.xml.gz | 12.4 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-0233 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-0233 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-0233 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-0233 | HTTPS FTP |
-Related structure data
| Related structure data |  6hizMC  0229C  0230C  0231C  0232C  6hivC  6hiwC  6hixC  6hiyC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_0233.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_0233.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | map of the head of the T. brucei mitoribosome small subunit | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.39 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
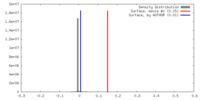
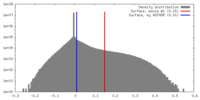
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Half map: half map (even) of the head of the...
| File | emd_0233_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map (even) of the head of the T. brucei mitoribosome small subunit | ||||||||||||
| Projections & Slices |
| ||||||||||||
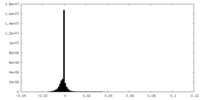
| Density Histograms |
-Half map: half map (odd) of the head of the...
| File | emd_0233_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | half map (odd) of the head of the T. brucei mitoribosome small subunit | ||||||||||||
| Projections & Slices |
| ||||||||||||
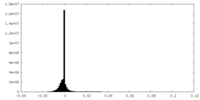
| Density Histograms |
- Sample components
Sample components
+Entire : head of the T. brucei mitoribosome small subunit
+Supramolecule #1: head of the T. brucei mitoribosome small subunit
+Macromolecule #1: mS48
+Macromolecule #2: mS59
+Macromolecule #3: mS49
+Macromolecule #4: mS50
+Macromolecule #5: mS52
+Macromolecule #6: mS53
+Macromolecule #7: mS54
+Macromolecule #8: mS55
+Macromolecule #9: mS57
+Macromolecule #10: mS58
+Macromolecule #11: mS67
+Macromolecule #12: mS69
+Macromolecule #13: mS70
+Macromolecule #14: mS71
+Macromolecule #15: mS72
+Macromolecule #16: uS3m
+Macromolecule #17: uS9m
+Macromolecule #18: uS10m
+Macromolecule #19: uS11m
+Macromolecule #20: uS14m
+Macromolecule #21: uS18m
+Macromolecule #22: uS19m
+Macromolecule #23: mS29
+Macromolecule #24: mS33
+Macromolecule #25: mS35
+Macromolecule #27: Unknown protein
+Macromolecule #28: Unknown protein
+Macromolecule #26: RNA (143-MER)
+Macromolecule #29: SPERMIDINE
+Macromolecule #30: URIDINE 5'-TRIPHOSPHATE
+Macromolecule #31: GUANOSINE-5'-TRIPHOSPHATE
+Macromolecule #32: MAGNESIUM ION
+Macromolecule #33: water
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE-PROPANE / Chamber humidity: 98 % / Chamber temperature: 278 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Detector mode: INTEGRATING / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: OTHER / Details: initial model from 2D class averages in EMAN |
|---|---|
| Final reconstruction | Algorithm: FOURIER SPACE / Resolution.type: BY AUTHOR / Resolution: 3.08 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: RELION / Number images used: 101308 |
| Initial angle assignment | Type: OTHER |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD |
-Atomic model buiding 1
| Details | We used Coot and O for initial model building and refined the structure using Phenix |
|---|---|
| Refinement | Protocol: AB INITIO MODEL / Overall B value: 45 |
| Output model |  PDB-6hiz: |
 Movie
Movie Controller
Controller















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)