+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 4wnv | ||||||
|---|---|---|---|---|---|---|---|
| Title | Human Cytochrome P450 2D6 Quinine Complex | ||||||
 Components Components | Cytochrome P450 2D6 | ||||||
 Keywords Keywords | OXIDOREDUCTASE/OXIDOREDUCTASE INHIBITOR / CYP2D6 / P450 2D6 / P450 / MONOOXYGENASE / OXIDOREDUCTASE-OXIDOREDUCTASE INHIBITOR complex | ||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of binding / negative regulation of organofluorine metabolic process / Miscellaneous substrates / isoquinoline alkaloid metabolic process / Fatty acids / coumarin metabolic process / Oxidoreductases; Acting on paired donors, with incorporation or reduction of molecular oxygen; With reduced flavin or flavoprotein as one donor, and incorporation of one atom of oxygen into the other donor / CYP2E1 reactions / arachidonate metabolic process / anandamide 8,9 epoxidase activity ...negative regulation of binding / negative regulation of organofluorine metabolic process / Miscellaneous substrates / isoquinoline alkaloid metabolic process / Fatty acids / coumarin metabolic process / Oxidoreductases; Acting on paired donors, with incorporation or reduction of molecular oxygen; With reduced flavin or flavoprotein as one donor, and incorporation of one atom of oxygen into the other donor / CYP2E1 reactions / arachidonate metabolic process / anandamide 8,9 epoxidase activity / anandamide 11,12 epoxidase activity / anandamide 14,15 epoxidase activity / alkaloid catabolic process / Biosynthesis of maresin-like SPMs / monoterpenoid metabolic process / alkaloid metabolic process / oxidative demethylation / Xenobiotics / oxidoreductase activity, acting on paired donors, with incorporation or reduction of molecular oxygen, reduced flavin or flavoprotein as one donor, and incorporation of one atom of oxygen / long-chain fatty acid biosynthetic process / estrogen metabolic process / retinol metabolic process / Aspirin ADME / steroid metabolic process / xenobiotic catabolic process / xenobiotic metabolic process / cholesterol metabolic process / monooxygenase activity / oxidoreductase activity / iron ion binding / intracellular membrane-bounded organelle / heme binding / endoplasmic reticulum membrane / endoplasmic reticulum / mitochondrion / cytoplasm Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.35 Å MOLECULAR REPLACEMENT / Resolution: 2.35 Å | ||||||
 Authors Authors | Wang, A. / Stout, C.D. / Johnson, E.F. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: J.Biol.Chem. / Year: 2015 Journal: J.Biol.Chem. / Year: 2015Title: Contributions of Ionic Interactions and Protein Dynamics to Cytochrome P450 2D6 (CYP2D6) Substrate and Inhibitor Binding. Authors: Wang, A. / Stout, C.D. / Zhang, Q. / Johnson, E.F. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  4wnv.cif.gz 4wnv.cif.gz | 742.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb4wnv.ent.gz pdb4wnv.ent.gz | 615.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  4wnv.json.gz 4wnv.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  4wnv_validation.pdf.gz 4wnv_validation.pdf.gz | 1.8 MB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  4wnv_full_validation.pdf.gz 4wnv_full_validation.pdf.gz | 1.8 MB | Display | |
| Data in XML |  4wnv_validation.xml.gz 4wnv_validation.xml.gz | 67.2 KB | Display | |
| Data in CIF |  4wnv_validation.cif.gz 4wnv_validation.cif.gz | 90.7 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/wn/4wnv https://data.pdbj.org/pub/pdb/validation_reports/wn/4wnv ftp://data.pdbj.org/pub/pdb/validation_reports/wn/4wnv ftp://data.pdbj.org/pub/pdb/validation_reports/wn/4wnv | HTTPS FTP |
-Related structure data
| Related structure data |  3tbgSC  3tdaC  4wntC  4wnuC  4wnwC S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| 3 | 
| ||||||||
| 4 | 
| ||||||||
| Unit cell |
| ||||||||
| Details | monomeric |
- Components
Components
-Protein , 1 types, 4 molecules ABCD
| #1: Protein | Mass: 53730.566 Da / Num. of mol.: 4 / Fragment: UNP RESIDUES 34-497 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: CYP2D6, CYP2DL1 / Plasmid: PCWORI / Production host: Homo sapiens (human) / Gene: CYP2D6, CYP2DL1 / Plasmid: PCWORI / Production host:  |
|---|
-Non-polymers , 6 types, 320 molecules 
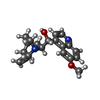









| #2: Chemical | ChemComp-HEM / #3: Chemical | ChemComp-QI9 / #4: Chemical | ChemComp-GOL / #5: Chemical | ChemComp-ZN / #6: Chemical | #7: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.22 Å3/Da / Density % sol: 61.81 % |
|---|---|
| Crystal grow | Temperature: 298 K / Method: vapor diffusion, hanging drop Details: PEG3350, SODIUM ACETATE, SODIUM CACODYLATE, POTASSIUM PHOSPHATE, SODIUM CHLORIDE, ZINC CHLORIDE, GLYCEROL, BETA-MERCAPTOETHANOL, THIORIDAZINE, HEGA-10, FACIAL AMPHIPHILE 231_CHOL. Quinine ...Details: PEG3350, SODIUM ACETATE, SODIUM CACODYLATE, POTASSIUM PHOSPHATE, SODIUM CHLORIDE, ZINC CHLORIDE, GLYCEROL, BETA-MERCAPTOETHANOL, THIORIDAZINE, HEGA-10, FACIAL AMPHIPHILE 231_CHOL. Quinine was added to the mother liquor after crystallization of the P450 2D6 thiordidazine complex. |
-Data collection
| Diffraction | Mean temperature: 100 K | |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SSRL SSRL  / Beamline: BL7-1 / Wavelength: 1.097173 Å / Beamline: BL7-1 / Wavelength: 1.097173 Å | |||||||||||||||||||||||||||
| Detector | Type: ADSC QUANTUM 315r / Detector: CCD / Date: Jul 25, 2011 / Details: Rh coated flat mirror | |||||||||||||||||||||||||||
| Radiation | Monochromator: Side scattering bent cube-root I-beam single crystal Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | |||||||||||||||||||||||||||
| Radiation wavelength | Wavelength: 1.097173 Å / Relative weight: 1 | |||||||||||||||||||||||||||
| Reflection | Resolution: 2.35→38.74 Å / Num. obs: 114948 / % possible obs: 98.4 % / Redundancy: 3.5 % / Biso Wilson estimate: 25.96 Å2 / CC1/2: 0.995 / Rmerge(I) obs: 0.06 / Rpim(I) all: 0.037 / Net I/σ(I): 16.8 / Num. measured all: 401591 | |||||||||||||||||||||||||||
| Reflection shell | Diffraction-ID: 1 / Rejects: _
|
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 3TBG Resolution: 2.35→35.426 Å / SU ML: 0.25 / Cross valid method: THROUGHOUT / σ(F): 0.01 / Phase error: 25 / Stereochemistry target values: ML
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 103.61 Å2 / Biso mean: 35.4643 Å2 / Biso min: 8.34 Å2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 2.35→35.426 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Total num. of bins used: 30
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller
























 PDBj
PDBj






