[English] 日本語
 Yorodumi
Yorodumi- PDB-1uqs: The Crystal Structure of Human CD1b with a Bound Bacterial Glycolipid -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1uqs | ||||||
|---|---|---|---|---|---|---|---|
| Title | The Crystal Structure of Human CD1b with a Bound Bacterial Glycolipid | ||||||
 Components Components |
| ||||||
 Keywords Keywords | GLYCOPROTEIN / LIPID / GMM / CD1B / MHC / ANTIGEN PRESENTATION | ||||||
| Function / homology |  Function and homology information Function and homology information: / : / : / : / : / negative regulation of receptor binding / endogenous lipid antigen binding / exogenous lipid antigen binding / antigen processing and presentation, endogenous lipid antigen via MHC class Ib / lipopeptide binding ...: / : / : / : / : / negative regulation of receptor binding / endogenous lipid antigen binding / exogenous lipid antigen binding / antigen processing and presentation, endogenous lipid antigen via MHC class Ib / lipopeptide binding / antigen processing and presentation, exogenous lipid antigen via MHC class Ib / retina homeostasis / positive regulation of protein binding / regulation of membrane depolarization / early endosome lumen / Nef mediated downregulation of MHC class I complex cell surface expression / DAP12 interactions / Endosomal/Vacuolar pathway / T cell mediated cytotoxicity / Antigen Presentation: Folding, assembly and peptide loading of class I MHC / regulation of iron ion transport / cellular response to iron(III) ion / negative regulation of iron ion transport / negative regulation of forebrain neuron differentiation / antigen processing and presentation of exogenous protein antigen via MHC class Ib, TAP-dependent / iron ion transport / peptide antigen assembly with MHC class I protein complex / ER to Golgi transport vesicle membrane / regulation of erythrocyte differentiation / response to molecule of bacterial origin / HFE-transferrin receptor complex / MHC class I peptide loading complex / transferrin transport / cellular response to iron ion / negative regulation of receptor-mediated endocytosis / positive regulation of T cell cytokine production / antigen processing and presentation of endogenous peptide antigen via MHC class I / MHC class I protein complex / peptide antigen assembly with MHC class II protein complex / negative regulation of neurogenesis / cellular response to nicotine / MHC class II protein complex / positive regulation of receptor-mediated endocytosis / multicellular organismal-level iron ion homeostasis / positive regulation of T cell mediated cytotoxicity / specific granule lumen / antigen processing and presentation of exogenous peptide antigen via MHC class II / positive regulation of immune response / peptide antigen binding / phagocytic vesicle membrane / recycling endosome membrane / positive regulation of T cell activation / negative regulation of epithelial cell proliferation / Interferon gamma signaling / Immunoregulatory interactions between a Lymphoid and a non-Lymphoid cell / Modulation by Mtb of host immune system / sensory perception of smell / tertiary granule lumen / positive regulation of cellular senescence / MHC class II protein complex binding / T cell differentiation in thymus / DAP12 signaling / antimicrobial humoral immune response mediated by antimicrobial peptide / late endosome membrane / negative regulation of neuron projection development / antibacterial humoral response / protein refolding / ER-Phagosome pathway / cellular response to lipopolysaccharide / early endosome membrane / amyloid fibril formation / protein homotetramerization / defense response to Gram-negative bacterium / intracellular iron ion homeostasis / adaptive immune response / learning or memory / endosome membrane / defense response to Gram-positive bacterium / immune response / endoplasmic reticulum lumen / Amyloid fiber formation / Golgi membrane / external side of plasma membrane / innate immune response / lysosomal membrane / focal adhesion / Neutrophil degranulation / SARS-CoV-2 activates/modulates innate and adaptive immune responses / structural molecule activity / cell surface / endoplasmic reticulum / Golgi apparatus / protein homodimerization activity / : / extracellular exosome / extracellular region / membrane / identical protein binding / plasma membrane / cytosol Similarity search - Function | ||||||
| Biological species |  HOMO SAPIENS (human) HOMO SAPIENS (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 3.1 Å MOLECULAR REPLACEMENT / Resolution: 3.1 Å | ||||||
 Authors Authors | Batuwangala, T. / Shepherd, D. / Gadola, S.D. / Gibson, K.J.C. / Zaccai, N.R. / Besra, G.S. / Cerundolo, V. / Jones, E.Y. | ||||||
 Citation Citation |  Journal: J Immunol. / Year: 2004 Journal: J Immunol. / Year: 2004Title: The crystal structure of human CD1b with a bound bacterial glycolipid. Authors: Batuwangala, T. / Shepherd, D. / Gadola, S.D. / Gibson, K.J. / Zaccai, N.R. / Fersht, A.R. / Besra, G.S. / Cerundolo, V. / Jones, E.Y. | ||||||
| History |
| ||||||
| Remark 700 | SHEET THE SHEET STRUCTURE OF THIS MOLECULE IS BIFURCATED. IN ORDER TO REPRESENT THIS FEATURE IN ... SHEET THE SHEET STRUCTURE OF THIS MOLECULE IS BIFURCATED. IN ORDER TO REPRESENT THIS FEATURE IN THE SHEET RECORDS BELOW, TWO SHEETS ARE DEFINED. |
- Structure visualization
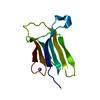
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1uqs.cif.gz 1uqs.cif.gz | 90.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1uqs.ent.gz pdb1uqs.ent.gz | 68.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1uqs.json.gz 1uqs.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/uq/1uqs https://data.pdbj.org/pub/pdb/validation_reports/uq/1uqs ftp://data.pdbj.org/pub/pdb/validation_reports/uq/1uqs ftp://data.pdbj.org/pub/pdb/validation_reports/uq/1uqs | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  1gzqS S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 33088.215 Da / Num. of mol.: 1 / Fragment: FRAGMENT: RESIDUES 18-295 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  HOMO SAPIENS (human) / Plasmid: PET23D / Production host: HOMO SAPIENS (human) / Plasmid: PET23D / Production host:  |
|---|---|
| #2: Protein | Mass: 11879.356 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  HOMO SAPIENS (human) / Plasmid: PET23D / Production host: HOMO SAPIENS (human) / Plasmid: PET23D / Production host:  |
| #3: Chemical | ChemComp-GMM / |
| #4: Water | ChemComp-HOH / |
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 2 X-RAY DIFFRACTION / Number of used crystals: 2 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.5 Å3/Da / Density % sol: 67 % / Description: WEISSENBERG METHOD | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | pH: 5.6 Details: 0.1M SODIUM CITRATE PH 5.6, 0.5M AMMONIUM SULFATE, 0.5M LITHIUM SULFATE | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Temperature: 20 ℃ / pH: 6.5 / Method: vapor diffusion, sitting drop | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: ID14-4 / Wavelength: 1.01 / Beamline: ID14-4 / Wavelength: 1.01 |
| Detector | Type: ADSC CCD / Detector: CCD / Date: Nov 15, 2002 / Details: MIRRORS |
| Radiation | Monochromator: SILICON CRYSTAL / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1.01 Å / Relative weight: 1 |
| Reflection | Resolution: 3.1→20 Å / Num. obs: 11697 / % possible obs: 99.9 % / Observed criterion σ(I): 1 / Redundancy: 12.8 % / Rmerge(I) obs: 0.077 / Net I/σ(I): 34.9 |
| Reflection shell | Resolution: 3.1→3.21 Å / Rmerge(I) obs: 0.604 / Mean I/σ(I) obs: 5.7 / % possible all: 100 |
| Reflection | *PLUS Highest resolution: 3.1 Å / Lowest resolution: 20 Å / Num. measured all: 150075 / Rmerge(I) obs: 0.077 |
| Reflection shell | *PLUS % possible obs: 100 % / Rmerge(I) obs: 0.604 / Mean I/σ(I) obs: 5.7 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 1GZQ Resolution: 3.1→19.9 Å / Rfactor Rfree error: 0.01 / Data cutoff high absF: 1353771.84 / Isotropic thermal model: GROUP / Cross valid method: THROUGHOUT / σ(F): 0
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Solvent model: FLAT MODEL / Bsol: 28.6 Å2 / ksol: 0.194067 e/Å3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 87 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine analyze |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 3.1→19.9 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 3.1→3.29 Å / Rfactor Rfree error: 0.041 / Total num. of bins used: 6
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Xplor file |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS Rfactor Rfree: 0.291 / Rfactor Rwork: 0.233 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS
|
 Movie
Movie Controller
Controller





































































 PDBj
PDBj