+検索条件
-Structure paper
| タイトル | High-resolution in situ structures of mammalian respiratory supercomplexes. |
|---|---|
| ジャーナル・号・ページ | Nature, Vol. 631, Issue 8019, Page 232-239, Year 2024 |
| 掲載日 | 2024年5月29日 |
 著者 著者 | Wan Zheng / Pengxin Chai / Jiapeng Zhu / Kai Zhang /   |
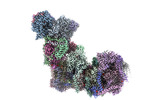
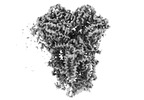
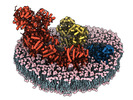
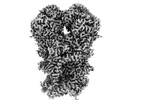
| PubMed 要旨 | Mitochondria play a pivotal part in ATP energy production through oxidative phosphorylation, which occurs within the inner membrane through a series of respiratory complexes. Despite extensive in ...Mitochondria play a pivotal part in ATP energy production through oxidative phosphorylation, which occurs within the inner membrane through a series of respiratory complexes. Despite extensive in vitro structural studies, determining the atomic details of their molecular mechanisms in physiological states remains a major challenge, primarily because of loss of the native environment during purification. Here we directly image porcine mitochondria using an in situ cryo-electron microscopy approach. This enables us to determine the structures of various high-order assemblies of respiratory supercomplexes in their native states. We identify four main supercomplex organizations: IIIIIV, IIIIIV, IIIIIV and IIIIIV, which potentially expand into higher-order arrays on the inner membranes. These diverse supercomplexes are largely formed by 'protein-lipids-protein' interactions, which in turn have a substantial impact on the local geometry of the surrounding membranes. Our in situ structures also capture numerous reactive intermediates within these respiratory supercomplexes, shedding light on the dynamic processes of the ubiquinone/ubiquinol exchange mechanism in complex I and the Q-cycle in complex III. Structural comparison of supercomplexes from mitochondria treated under different conditions indicates a possible correlation between conformational states of complexes I and III, probably in response to environmental changes. By preserving the native membrane environment, our approach enables structural studies of mitochondrial respiratory supercomplexes in reaction at high resolution across multiple scales, from atomic-level details to the broader subcellular context. |
 リンク リンク |  Nature / Nature /  PubMed:38811722 / PubMed:38811722 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 2.0 - 6.5 Å |
| 構造データ | EMDB-42143, PDB-8ud1: EMDB-42165, PDB-8ueo: EMDB-42166, PDB-8uep: EMDB-42167, PDB-8ueq: EMDB-42168, PDB-8uer: EMDB-42169, PDB-8ues: EMDB-42170, PDB-8uet: EMDB-42171, PDB-8ueu: EMDB-42172, PDB-8uev: EMDB-42173, PDB-8uew: EMDB-42174, PDB-8uex: EMDB-42175, PDB-8uey: EMDB-42176, PDB-8uez: EMDB-42221, PDB-8ugd: EMDB-42222, PDB-8uge: EMDB-42223, PDB-8ugf: EMDB-42224, PDB-8ugg: EMDB-42225, PDB-8ugh: EMDB-42226, PDB-8ugi: EMDB-42227, PDB-8ugj: EMDB-42228, PDB-8ugk: EMDB-42229, PDB-8ugl: EMDB-42230, PDB-8ugn: EMDB-42231, PDB-8ugp: EMDB-42233, PDB-8ugr: |
| 化合物 |  ChemComp-3PE:  ChemComp-PC1:  ChemComp-SF4:  ChemComp-FES:  ChemComp-FMN:  ChemComp-K:  ChemComp-CDL:  ChemComp-GTP:  ChemComp-MG:  ChemComp-NDP:  ChemComp-ZN:  ChemComp-EHZ:  ChemComp-AME:  ChemComp-MYR:  ChemComp-HOH:  ChemComp-PGT:  ChemComp-HEM:  ChemComp-HEC:  ChemComp-AYA:  ChemComp-PEK:  ChemComp-HEA:  ChemComp-CU:  ChemComp-NA:  ChemComp-PGV:  ChemComp-CUA:  ChemComp-PSC:  ChemComp-PO4:  ChemComp-CHD: |
| 由来 |
|
 キーワード キーワード | ELECTRON TRANSPORT / in-situ cryo-EM structure / mammalian / mitochondria / respiratory supercomplex / proton pumping / membrane protein / In-situ / In-situ cryo-EM structure mitochondria / complex I / Active-Q10(half occupied) |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について