[English] 日本語
 Yorodumi
Yorodumi- EMDB-3656: Structure of the Bacillus subtilis hibernating 100S ribosome reve... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-3656 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of the Bacillus subtilis hibernating 100S ribosome reveals the basis for 70S dimerization. | |||||||||
 Map data Map data | Cryo-EM reconstruction of the Bacillus subtilis 100s subcomplex (70s-30s) | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | 100S / Bacillus subtilis / cryo-EM / Hibernation / HPF / RMF / rRNA / YvyD / translation | |||||||||
| Function / homology |  Function and homology information Function and homology informationprimary metabolic process / negative regulation of translational elongation / positive regulation of rRNA processing / nucleoid / ribosomal small subunit binding / rRNA processing / large ribosomal subunit / ribosome binding / ribosomal small subunit biogenesis / ribosomal small subunit assembly ...primary metabolic process / negative regulation of translational elongation / positive regulation of rRNA processing / nucleoid / ribosomal small subunit binding / rRNA processing / large ribosomal subunit / ribosome binding / ribosomal small subunit biogenesis / ribosomal small subunit assembly / small ribosomal subunit / small ribosomal subunit rRNA binding / transferase activity / 5S rRNA binding / large ribosomal subunit rRNA binding / cytosolic small ribosomal subunit / cytosolic large ribosomal subunit / cytoplasmic translation / tRNA binding / rRNA binding / negative regulation of translation / ribosome / structural constituent of ribosome / translation / ribonucleoprotein complex / response to antibiotic / mRNA binding / DNA binding / RNA binding / zinc ion binding / metal ion binding / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.8 Å | |||||||||
 Authors Authors | Beckert B / Abdelshahid M / Schaefer H / Steinchen W / Arenz S / Berninghausen O / Beckmann R / Bange G / Turgay K / Wilson DN | |||||||||
| Funding support |  Germany, 1 items Germany, 1 items
| |||||||||
 Citation Citation |  Journal: EMBO J / Year: 2017 Journal: EMBO J / Year: 2017Title: Structure of the hibernating 100S ribosome reveals the basis for 70S dimerization. Authors: Bertrand Beckert / Maha Abdelshahid / Heinrich Schäfer / Wieland Steinchen / Stefan Arenz / Otto Berninghausen / Roland Beckmann / Gert Bange / Kürşad Turgay / Daniel N Wilson /  Abstract: Under stress conditions, such as nutrient deprivation, bacteria enter into a hibernation stage, which is characterized by the appearance of 100S ribosomal particles. In , dimerization of 70S ...Under stress conditions, such as nutrient deprivation, bacteria enter into a hibernation stage, which is characterized by the appearance of 100S ribosomal particles. In , dimerization of 70S ribosomes into 100S requires the action of the ribosome modulation factor (RMF) and the hibernation-promoting factor (HPF). Most other bacteria lack RMF and instead contain a long form HPF (LHPF), which is necessary and sufficient for 100S formation. While some structural information exists as to how RMF and HPF mediate formation of 100S (100S), structural insight into 100S formation by LHPF has so far been lacking. Here we present a cryo-EM structure of the hibernating 100S (100S), revealing that the C-terminal domain (CTD) of the LHPF occupies a site on the 30S platform distinct from RMF Moreover, unlike RMF, the HPF-CTD is directly involved in forming the dimer interface, thereby illustrating the divergent mechanisms by which 100S formation is mediated in the majority of bacteria that contain LHPF, compared to some γ-proteobacteria, such as . | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_3656.map.gz emd_3656.map.gz | 47.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-3656-v30.xml emd-3656-v30.xml emd-3656.xml emd-3656.xml | 69.9 KB 69.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_3656.png emd_3656.png | 191.5 KB | ||
| Filedesc metadata |  emd-3656.cif.gz emd-3656.cif.gz | 13.1 KB | ||
| Others |  emd_3656_half_map_1.map.gz emd_3656_half_map_1.map.gz emd_3656_half_map_2.map.gz emd_3656_half_map_2.map.gz | 324.5 MB 324.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-3656 http://ftp.pdbj.org/pub/emdb/structures/EMD-3656 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-3656 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-3656 | HTTPS FTP |
-Validation report
| Summary document |  emd_3656_validation.pdf.gz emd_3656_validation.pdf.gz | 428.5 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_3656_full_validation.pdf.gz emd_3656_full_validation.pdf.gz | 427.6 KB | Display | |
| Data in XML |  emd_3656_validation.xml.gz emd_3656_validation.xml.gz | 15.4 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-3656 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-3656 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-3656 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-3656 | HTTPS FTP |
-Related structure data
| Related structure data |  5njtMC  3664C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_3656.map.gz / Format: CCP4 / Size: 347.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_3656.map.gz / Format: CCP4 / Size: 347.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM reconstruction of the Bacillus subtilis 100s subcomplex (70s-30s) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.084 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Half map: Half Map 1 of the Bacillus subtilis 100s subcomplex (70s-30s)
| File | emd_3656_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half Map 1 of the Bacillus subtilis 100s subcomplex (70s-30s) | ||||||||||||
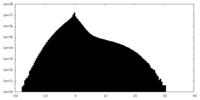
| Projections & Slices |
| ||||||||||||
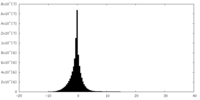
| Density Histograms |
-Half map: Half Map 2 of the Bacillus subtilis 100s subcomplex (70s-30s)
| File | emd_3656_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half Map 2 of the Bacillus subtilis 100s subcomplex (70s-30s) | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : Structure of the Bacillus subtilis hibernating 100S ribosome reve...
+Supramolecule #1: Structure of the Bacillus subtilis hibernating 100S ribosome reve...
+Macromolecule #1: 16S ribosomal RNA
+Macromolecule #21: 23S ribosomal RNA
+Macromolecule #22: 5S ribosomal RNA
+Macromolecule #2: 30S ribosomal protein S2
+Macromolecule #3: 30S ribosomal protein S3
+Macromolecule #4: 30S ribosomal protein S4
+Macromolecule #5: 30S ribosomal protein S5
+Macromolecule #6: 30S ribosomal protein S6
+Macromolecule #7: 30S ribosomal protein S7
+Macromolecule #8: 30S ribosomal protein S8
+Macromolecule #9: 30S ribosomal protein S9
+Macromolecule #10: 30S ribosomal protein S10
+Macromolecule #11: 30S ribosomal protein S11
+Macromolecule #12: 30S ribosomal protein S12
+Macromolecule #13: 30S ribosomal protein S13
+Macromolecule #14: 30S ribosomal protein S14
+Macromolecule #15: 30S ribosomal protein S15
+Macromolecule #16: 30S ribosomal protein S16
+Macromolecule #17: 30S ribosomal protein S17
+Macromolecule #18: 30S ribosomal protein S18
+Macromolecule #19: 30S ribosomal protein S19
+Macromolecule #20: 30S ribosomal protein S20
+Macromolecule #23: 50S ribosomal protein L2
+Macromolecule #24: 50S ribosomal protein L3
+Macromolecule #25: 50S ribosomal protein L4
+Macromolecule #26: 50S ribosomal protein L5
+Macromolecule #27: 50S ribosomal protein L6
+Macromolecule #28: 50S ribosomal protein L10
+Macromolecule #29: 50S ribosomal protein L13
+Macromolecule #30: 50S ribosomal protein L14
+Macromolecule #31: 50S ribosomal protein L15
+Macromolecule #32: 50S ribosomal protein L16
+Macromolecule #33: 50S ribosomal protein L17
+Macromolecule #34: 50S ribosomal protein L18
+Macromolecule #35: 50S ribosomal protein L19
+Macromolecule #36: 50S ribosomal protein L20
+Macromolecule #37: 50S ribosomal protein L21
+Macromolecule #38: 50S ribosomal protein L22
+Macromolecule #39: 50S ribosomal protein L23
+Macromolecule #40: 50S ribosomal protein L24
+Macromolecule #41: 50S ribosomal protein L27
+Macromolecule #42: 50S ribosomal protein L32
+Macromolecule #43: 50S ribosomal protein L33 1
+Macromolecule #44: 50S ribosomal protein L34
+Macromolecule #45: 50S ribosomal protein L35
+Macromolecule #46: 50S ribosomal protein L36
+Macromolecule #47: 50S ribosomal protein L28
+Macromolecule #48: 50S ribosomal protein L29
+Macromolecule #49: 50S ribosomal protein L30
+Macromolecule #50: 50S ribosomal protein L31
+Macromolecule #51: Ribosome hibernation promotion factor
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 Component:
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Grid | Model: Quantifoil R3/3 / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 30 sec. | ||||||||||||
| Vitrification | Cryogen name: ETHANE | ||||||||||||
| Details | 4 OD260/ml ml Bs100S sample were applied to 2 nm pre-coated Quantifoil R3/3 holey carbon supported grids and vitrified using Vitrobot Mark IV (FEI Company) |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON II (4k x 4k) / Detector mode: COUNTING / Average electron dose: 2.5 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller




















 Z
Z Y
Y X
X