[English] 日本語
 Yorodumi
Yorodumi- PDB-2sbt: A COMPARISON OF THE THREE-DIMENSIONAL STRUCTURES OF SUBTILISIN BP... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2sbt | ||||||
|---|---|---|---|---|---|---|---|
| Title | A COMPARISON OF THE THREE-DIMENSIONAL STRUCTURES OF SUBTILISIN BPN AND SUBTILISIN NOVO | ||||||
 Components Components | SUBTILISIN NOVO | ||||||
 Keywords Keywords | HYDROLASE (SERINE PROTEINASE) | ||||||
| Function / homology |  Function and homology information Function and homology information subtilisin / sporulation resulting in formation of a cellular spore / subtilisin / sporulation resulting in formation of a cellular spore /  fibrinolysis / serine-type endopeptidase activity / fibrinolysis / serine-type endopeptidase activity /  proteolysis / extracellular region / proteolysis / extracellular region /  metal ion binding metal ion bindingSimilarity search - Function | ||||||
| Biological species |   Bacillus amyloliquefaciens (bacteria) Bacillus amyloliquefaciens (bacteria) | ||||||
| Method |  X-RAY DIFFRACTION / Resolution: 2.8 Å X-RAY DIFFRACTION / Resolution: 2.8 Å | ||||||
 Authors Authors | Drenth, J. / Hol, W.G.J. / Jansonius, J.N. / Koekoek, R. | ||||||
 Citation Citation |  Journal: Cold Spring Harbor Symp.Quant.Biol. / Year: 1972 Journal: Cold Spring Harbor Symp.Quant.Biol. / Year: 1972Title: A comparison of the three-dimensional structures of subtilisin BPN' and subtilisin novo. Authors: Drenth, J. / Hol, W.G. / Jansonius, J.N. / Koekoek, R. #1:  Journal: Eur.J.Biochem. / Year: 1972 Journal: Eur.J.Biochem. / Year: 1972Title: Subtilisin Novo,the Three-Dimensional Structure and its Comparison with Subtilisin Bpn Authors: Drenth, J. / Hol, W.G.J. / Jansonius, J.N. / Koekoek, R. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2sbt.cif.gz 2sbt.cif.gz | 61.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2sbt.ent.gz pdb2sbt.ent.gz | 35.2 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2sbt.json.gz 2sbt.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/sb/2sbt https://data.pdbj.org/pub/pdb/validation_reports/sb/2sbt ftp://data.pdbj.org/pub/pdb/validation_reports/sb/2sbt ftp://data.pdbj.org/pub/pdb/validation_reports/sb/2sbt | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 27552.525 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Bacillus amyloliquefaciens (bacteria) / References: UniProt: P00782, 3.4.21.14 Bacillus amyloliquefaciens (bacteria) / References: UniProt: P00782, 3.4.21.14 |
|---|---|
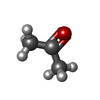
| #2: Chemical | ChemComp-ACN /  Acetone Acetone |
| #3: Water | ChemComp-HOH /  Water Water |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION X-RAY DIFFRACTION |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 1.98 Å3/Da / Density % sol: 37.84 % |
|---|---|
Crystal grow | *PLUS Method: otherDetails: Birktoft, J.J., (1969) Biophys. Res. Commun., 36, 131. |
- Processing
Processing
| Refinement | Highest resolution: 2.8 Å | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement step | Cycle: LAST / Highest resolution: 2.8 Å
|
 Movie
Movie Controller
Controller












 PDBj
PDBj