+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1hbm | ||||||
|---|---|---|---|---|---|---|---|
| Title | METHYL-COENZYME M REDUCTASE ENZYME PRODUCT COMPLEX | ||||||
 Components Components | (METHYL-COENZYME M REDUCTASE I ... Coenzyme-B sulfoethylthiotransferase) x 3 Coenzyme-B sulfoethylthiotransferase) x 3 | ||||||
 Keywords Keywords |  METHANOGENESIS / BIOLOGICAL METHANOGENESIS / NI-ENZYME / METHANOGENESIS / BIOLOGICAL METHANOGENESIS / NI-ENZYME /  OXIDOREDUCTASE OXIDOREDUCTASE | ||||||
| Function / homology |  Function and homology information Function and homology information coenzyme-B sulfoethylthiotransferase / coenzyme-B sulfoethylthiotransferase /  coenzyme-B sulfoethylthiotransferase activity / coenzyme-B sulfoethylthiotransferase activity /  methanogenesis / methanogenesis /  metal ion binding / metal ion binding /  cytoplasm cytoplasmSimilarity search - Function | ||||||
| Biological species |    METHANOTHERMOBACTER THERMAUTOTROPHICUS (archaea) METHANOTHERMOBACTER THERMAUTOTROPHICUS (archaea) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  MOLECULAR REPLACEMENT / Resolution: 1.8 Å MOLECULAR REPLACEMENT / Resolution: 1.8 Å | ||||||
 Authors Authors | Ermler, U. / Grabarse, W. | ||||||
 Citation Citation |  Journal: J.Mol.Biol. / Year: 2001 Journal: J.Mol.Biol. / Year: 2001Title: On the Mechanism of Biological Methane Formation: Structural Evidence for Conformational Changes in Methyl-Coenzyme M Reductase Upon Substrate Binding Authors: Grabarse, W. / Mahlert, F. / Duin, E.C. / Goubeaud, M. / Shima, S. / Thauer, R.K. / Lamzin, V. / Ermler, U. | ||||||
| History |
|
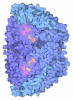
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1hbm.cif.gz 1hbm.cif.gz | 548.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1hbm.ent.gz pdb1hbm.ent.gz | 437.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1hbm.json.gz 1hbm.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/hb/1hbm https://data.pdbj.org/pub/pdb/validation_reports/hb/1hbm ftp://data.pdbj.org/pub/pdb/validation_reports/hb/1hbm ftp://data.pdbj.org/pub/pdb/validation_reports/hb/1hbm | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  1hbnC  1hboC  1hbuC  1mroS C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
| ||||||||
| Noncrystallographic symmetry (NCS) | NCS oper: (Code: given Matrix: (0.6268, -0.7779, 0.0458), Vector  : : |
- Components
Components
-METHYL-COENZYME M REDUCTASE I ... , 3 types, 6 molecules ADBECF
| #1: Protein | Mass: 60508.469 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)    METHANOTHERMOBACTER THERMAUTOTROPHICUS (archaea) METHANOTHERMOBACTER THERMAUTOTROPHICUS (archaea)Cellular location: CYTOPLASM  / Strain: MARBURG / References: UniProt: P11558 / Strain: MARBURG / References: UniProt: P11558#2: Protein | Mass: 47148.477 Da / Num. of mol.: 2 / Source method: isolated from a natural source Source: (natural)    METHANOTHERMOBACTER THERMAUTOTROPHICUS (archaea) METHANOTHERMOBACTER THERMAUTOTROPHICUS (archaea)Cellular location: CYTOPLASM  / Strain: MARBURG / References: UniProt: P11560 / Strain: MARBURG / References: UniProt: P11560#3: Protein | Mass: 28666.039 Da / Num. of mol.: 2 / Source method: isolated from a natural source / Details: MCR-OX-SILENT STATE, ENZYME-SUBSTRATE COMPLEX Source: (natural)    METHANOTHERMOBACTER THERMAUTOTROPHICUS (archaea) METHANOTHERMOBACTER THERMAUTOTROPHICUS (archaea)Cellular location: CYTOPLASM  / Strain: MARBURG / References: UniProt: P11562 / Strain: MARBURG / References: UniProt: P11562 |
|---|
-Non-polymers , 8 types, 2124 molecules 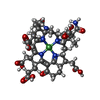













| #4: Chemical |  Cofactor F430 Cofactor F430#5: Chemical | #6: Chemical | ChemComp-GOL /  Glycerol Glycerol#7: Chemical | ChemComp-NA / #8: Chemical |  Chloride Chloride#9: Chemical | ChemComp-MG / #10: Chemical | ChemComp-ZN / | #11: Water | ChemComp-HOH / |  Water Water |
|---|
-Details
| Compound details | CATALYZES THE FINAL STEP IN METHANOGEN| Sequence details | REFERENCE: 1 RESIDUE IS LACKING IN THE C-TERMINUS AND THE N-TERMINUS, C AND N-TERMINUS ARE VISIBLE ...REFERENCE: 1 RESIDUE IS LACKING IN THE C-TERMINUS AND THE N-TERMINUS, C AND N-TERMINUS ARE VISIBLE IN THE CRYSTAL STRUCTURE. MET 1 WAS CLEARLY SHOWN NOT TO BE PRESENT. | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.9 Å3/Da / Density % sol: 31 % | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Crystal grow | pH: 7.5 Details: 25% PEG 400, 0.2 M NACL, 20 MG/ML FINAL PROTEIN CONC, 0.2 M MGCL, 0.1 M HEPES PH 7.5 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Temperature: 4 ℃ / Method: vapor diffusion, hanging drop | ||||||||||||||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  ROTATING ANODE / Type: RIGAKU / Wavelength: 1.54 ROTATING ANODE / Type: RIGAKU / Wavelength: 1.54 |
| Detector | Type: MAR scanner 345 mm plate / Detector: IMAGE PLATE / Date: Apr 15, 1998 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 1.54 Å / Relative weight: 1 : 1.54 Å / Relative weight: 1 |
| Reflection | Resolution: 1.8→10 Å / Num. obs: 198635 / % possible obs: 91.8 % / Redundancy: 4.2 % / Rmerge(I) obs: 0.088 / Rsym value: 0.08 / Net I/σ(I): 8.21 |
| Reflection shell | Resolution: 1.8→1.82 Å / Redundancy: 2 % / Rmerge(I) obs: 0.18 / Mean I/σ(I) obs: 3.9 / Rsym value: 0.19 / % possible all: 74.9 |
| Reflection shell | *PLUS Highest resolution: 1.8 Å / % possible obs: 74.9 % / Rmerge(I) obs: 0.183 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 1MRO Resolution: 1.8→10 Å / Cross valid method: THROUGHOUT / σ(F): 0
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.8→10 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Software | *PLUS Name: REFMAC / Classification: refinement | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS Rfactor obs: 0.164 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS |
 Movie
Movie Controller
Controller













 PDBj
PDBj