[English] 日本語
 Yorodumi
Yorodumi- PDB-7yxj: Drosophila melanogaster JMJD7 (dmJMJD7) in complex with Mn and 2,... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7yxj | ||||||
|---|---|---|---|---|---|---|---|
| Title | Drosophila melanogaster JMJD7 (dmJMJD7) in complex with Mn and 2,4-PDCA | ||||||
 Components Components | GH14974p | ||||||
 Keywords Keywords | OXIDOREDUCTASE / NON-HEME / IRON / 2-OXOGLUTARATE / DIOXYGENASE / OXYGENASE / JMJC / JMJC DOMAIN / JMJC DOMAIN-CONTAINING PROTEIN 7 / JMJD7 / JMJC HYDROXYLASE / JMJC DEMETHYLASE / KDMS / POST-TRANSLATIONAL MODIFICATIONS / PTM / HYDROXYLATION / LYSYL HYDROXYLATION / DIMERISATION / TRANSLATION FACTOR / DEVELOPMENTALLY REGULATED GTP BINDING PROTEINS / DRG1 / DRG2 / TRAFAC GTPASE / HYPOXIA / NUCLEIC ACID- BINDING / METAL-BINDING / TRANSLATION / HELIX-LOOP-HELIX-BETA / DSBH / FACIAL TRIAD / DEVELOPMENT / CANCER / RIBOSOME BIOGENESIS | ||||||
| Function / homology |  Function and homology information Function and homology informationpeptidyl-lysine (3S)-dioxygenase / peptidyl-lysine 3-dioxygenase activity / Protein hydroxylation / 2-oxoglutarate-dependent dioxygenase activity / circadian behavior / Hydrolases; Acting on peptide bonds (peptidases) / ferrous iron binding / endopeptidase activity / identical protein binding / nucleus ...peptidyl-lysine (3S)-dioxygenase / peptidyl-lysine 3-dioxygenase activity / Protein hydroxylation / 2-oxoglutarate-dependent dioxygenase activity / circadian behavior / Hydrolases; Acting on peptide bonds (peptidases) / ferrous iron binding / endopeptidase activity / identical protein binding / nucleus / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.45 Å MOLECULAR REPLACEMENT / Resolution: 2.45 Å | ||||||
 Authors Authors | Chowdhury, R. / Schofield, C.J. | ||||||
| Funding support |  United Kingdom, 1items United Kingdom, 1items
| ||||||
 Citation Citation |  Journal: Sci Rep / Year: 2022 Journal: Sci Rep / Year: 2022Title: Conservation of the unusual dimeric JmjC fold of JMJD7 from Drosophila melanogaster to humans. Authors: Chowdhury, R. / Abboud, M.I. / Wiley, J. / Tumber, A. / Markolovic, S. / Schofield, C.J. #1:  Journal: Nat Chem Biol / Year: 2018 Journal: Nat Chem Biol / Year: 2018Title: The Jumonji-C oxygenase JMJD7 catalyzes (3S)-lysyl hydroxylation of TRAFAC GTPases. Authors: Markolovic, S. / Zhuang, Q. / Wilkins, S.E. / Eaton, C.D. / Abboud, M.I. / Katz, M.J. / McNeil, H.E. / Lesniak, R.K. / Hall, C. / Struwe, W.B. / Konietzny, R. / Davis, S. / Yang, M. / Ge, W. ...Authors: Markolovic, S. / Zhuang, Q. / Wilkins, S.E. / Eaton, C.D. / Abboud, M.I. / Katz, M.J. / McNeil, H.E. / Lesniak, R.K. / Hall, C. / Struwe, W.B. / Konietzny, R. / Davis, S. / Yang, M. / Ge, W. / Benesch, J.L.P. / Kessler, B.M. / Ratcliffe, P.J. / Cockman, M.E. / Fischer, R. / Wappner, P. / Chowdhury, R. / Coleman, M.L. / Schofield, C.J. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7yxj.cif.gz 7yxj.cif.gz | 463.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7yxj.ent.gz pdb7yxj.ent.gz | 382.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7yxj.json.gz 7yxj.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/yx/7yxj https://data.pdbj.org/pub/pdb/validation_reports/yx/7yxj ftp://data.pdbj.org/pub/pdb/validation_reports/yx/7yxj ftp://data.pdbj.org/pub/pdb/validation_reports/yx/7yxj | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  7yxgSC  7yxhC  7yxiC  7yxkC  7yxlC S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 4 molecules ABCD
| #1: Protein | Mass: 36714.309 Da / Num. of mol.: 4 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   References: UniProt: Q9VU77, Oxidoreductases; Acting on paired donors, with incorporation or reduction of molecular oxygen; With 2-oxoglutarate as one donor, and incorporation of one atom of oxygen into each donor |
|---|
-Non-polymers , 5 types, 218 molecules 


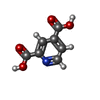





| #2: Chemical | ChemComp-MN / #3: Chemical | ChemComp-PEG / | #4: Chemical | ChemComp-EDO / #5: Chemical | ChemComp-PD2 / #6: Water | ChemComp-HOH / | |
|---|
-Details
| Has ligand of interest | Y |
|---|---|
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.56 Å3/Da / Density % sol: 51.97 % |
|---|---|
| Crystal grow | Temperature: 298 K / Method: vapor diffusion, sitting drop / pH: 5.4 Details: Sample: 16.0 mg/ml dmJMJD7 (in 50 mM Hepes-Na pH 7.5, 200 mM NaCl, 5% glycerol), 2.0 mM MnCl2 and 4.0 mM compound. Reservoir: 0.1 M Bis-Tris pH 5.4, 0.3 M magnesium chloride, 0.1 M ammonium ...Details: Sample: 16.0 mg/ml dmJMJD7 (in 50 mM Hepes-Na pH 7.5, 200 mM NaCl, 5% glycerol), 2.0 mM MnCl2 and 4.0 mM compound. Reservoir: 0.1 M Bis-Tris pH 5.4, 0.3 M magnesium chloride, 0.1 M ammonium acetate (or alternatively, 0.1 M barium chloride), and 0.002 M manganese chloride. Cryo-protection: 20% v/v ethylene glycol. |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  Diamond Diamond  / Beamline: I24 / Wavelength: 0.96863 Å / Beamline: I24 / Wavelength: 0.96863 Å | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Detector | Type: DECTRIS PILATUS 6M-F / Detector: PIXEL / Date: Aug 5, 2017 / Details: MIRRORS | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation | Monochromator: SI 111 / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Radiation wavelength | Wavelength: 0.96863 Å / Relative weight: 1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection | Resolution: 2.45→205.054 Å / Num. obs: 52754 / % possible obs: 99.8 % / Redundancy: 4.9 % / Biso Wilson estimate: 50.1 Å2 / CC1/2: 0.995 / Rpim(I) all: 0.062 / Rrim(I) all: 0.14 / Rsym value: 0.125 / Net I/av σ(I): 4.7 / Net I/σ(I): 7.8 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Reflection shell | Diffraction-ID: 1
|
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 7YXG Resolution: 2.45→55.354 Å / SU ML: 0.28 / Cross valid method: THROUGHOUT / σ(F): 1.34 / Phase error: 24.65 / Stereochemistry target values: ML
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 153.47 Å2 / Biso mean: 65.058 Å2 / Biso min: 22.42 Å2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: final / Resolution: 2.45→55.354 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Rfactor Rfree error: 0
|
 Movie
Movie Controller
Controller


 PDBj
PDBj





