[English] 日本語
 Yorodumi
Yorodumi- PDB-7rnu: nSH2 domain of p85-beta subunit of phosphatidylinositol 3-kinase ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7rnu | ||||||
|---|---|---|---|---|---|---|---|
| Title | nSH2 domain of p85-beta subunit of phosphatidylinositol 3-kinase in complex with an actin peptide with phosphorylated tyrosine 53 | ||||||
 Components Components |
| ||||||
 Keywords Keywords | PEPTIDE BINDING PROTEIN / phosphorylated tyrosine binding protein / actin peptide | ||||||
| Function / homology |  Function and homology information Function and homology informationregulation of actin filament polymerization / 1-phosphatidylinositol-3-kinase regulator activity / IRS-mediated signalling / phosphatidylinositol 3-kinase regulatory subunit binding / Formation of the dystrophin-glycoprotein complex (DGC) / regulation of stress fiber assembly / Co-stimulation by ICOS / Striated Muscle Contraction / RHOD GTPase cycle / phosphatidylinositol 3-kinase complex, class IA ...regulation of actin filament polymerization / 1-phosphatidylinositol-3-kinase regulator activity / IRS-mediated signalling / phosphatidylinositol 3-kinase regulatory subunit binding / Formation of the dystrophin-glycoprotein complex (DGC) / regulation of stress fiber assembly / Co-stimulation by ICOS / Striated Muscle Contraction / RHOD GTPase cycle / phosphatidylinositol 3-kinase complex, class IA / Nephrin family interactions / RHOF GTPase cycle / Signaling by LTK in cancer / Signaling by LTK / RND1 GTPase cycle / PI3K/AKT activation / RND2 GTPase cycle / RND3 GTPase cycle / Signaling by ALK / RHOB GTPase cycle / myosin binding / RHOJ GTPase cycle / mesenchyme migration / Synthesis of PIPs at the plasma membrane / intracellular glucose homeostasis / RHOU GTPase cycle / CDC42 GTPase cycle / RET signaling / striated muscle thin filament / skeletal muscle thin filament assembly / Interleukin-3, Interleukin-5 and GM-CSF signaling / PI3K Cascade / T cell differentiation / CD28 dependent PI3K/Akt signaling / Role of LAT2/NTAL/LAB on calcium mobilization / RHOA GTPase cycle / RAC2 GTPase cycle / RAC3 GTPase cycle / Interleukin receptor SHC signaling / Role of phospholipids in phagocytosis / regulation of protein localization to plasma membrane / Signaling by PDGFRA transmembrane, juxtamembrane and kinase domain mutants / Signaling by PDGFRA extracellular domain mutants / negative regulation of MAPK cascade / skeletal muscle fiber development / GPVI-mediated activation cascade / stress fiber / Tie2 Signaling / phosphotyrosine residue binding / RAC1 GTPase cycle / positive regulation of cell adhesion / Downstream signal transduction / Interleukin-7 signaling / B cell differentiation / muscle contraction / Signaling by phosphorylated juxtamembrane, extracellular and kinase domain KIT mutants / response to endoplasmic reticulum stress / sarcomere / actin filament / filopodium / Regulation of signaling by CBL / phosphatidylinositol 3-kinase/protein kinase B signal transduction / Signaling by SCF-KIT / ADP binding / receptor tyrosine kinase binding / positive regulation of protein import into nucleus / regulation of autophagy / structural constituent of cytoskeleton / VEGFA-VEGFR2 Pathway / Hydrolases; Acting on acid anhydrides; Acting on acid anhydrides to facilitate cellular and subcellular movement / cellular response to insulin stimulus / Constitutive Signaling by Aberrant PI3K in Cancer / Signaling by ALK fusions and activated point mutants / insulin receptor signaling pathway / DAP12 signaling / Downstream TCR signaling / PIP3 activates AKT signaling / actin cytoskeleton / lamellipodium / protein transport / cell body / PI5P, PP2A and IER3 Regulate PI3K/AKT Signaling / RAF/MAP kinase cascade / protein phosphatase binding / High laminar flow shear stress activates signaling by PIEZO1 and PECAM1:CDH5:KDR in endothelial cells / blood microparticle / G alpha (q) signalling events / Extra-nuclear estrogen signaling / immune response / protein heterodimerization activity / hydrolase activity / focal adhesion / positive regulation of gene expression / positive regulation of transcription by RNA polymerase II / extracellular space / extracellular exosome / ATP binding / nucleus / cytosol Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.45 Å MOLECULAR REPLACEMENT / Resolution: 1.45 Å | ||||||
 Authors Authors | Dai, S. / Horton, J.R. / Cheng, X. | ||||||
| Funding support |  United States, 1items United States, 1items
| ||||||
 Citation Citation |  Journal: To Be Published Journal: To Be PublishedTitle: The Functional Analysis of a Major Tyrosine Phosphorylation Site on Actin Authors: Amelie, A. / Dai, S. / Shen, X. / Horton, J.R. / Zhang, X. / Cheng, X. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7rnu.cif.gz 7rnu.cif.gz | 141.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7rnu.ent.gz pdb7rnu.ent.gz | 89.4 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7rnu.json.gz 7rnu.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/rn/7rnu https://data.pdbj.org/pub/pdb/validation_reports/rn/7rnu ftp://data.pdbj.org/pub/pdb/validation_reports/rn/7rnu ftp://data.pdbj.org/pub/pdb/validation_reports/rn/7rnu | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  7rnsC  7rnvC  2iuiS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 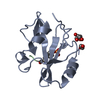
| ||||||||||||
| 2 | 
| ||||||||||||
| 3 | 
| ||||||||||||
| 4 | 
| ||||||||||||
| Unit cell |
| ||||||||||||
| Components on special symmetry positions |
|
- Components
Components
| #1: Protein | Mass: 13029.506 Da / Num. of mol.: 4 / Fragment: nSH2 domain (UNP residues 318-428) Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: PIK3R2 / Production host: Homo sapiens (human) / Gene: PIK3R2 / Production host:  #2: Protein/peptide | Mass: 1063.975 Da / Num. of mol.: 4 / Fragment: UNP residues 52-60 / Source method: obtained synthetically / Source: (synth.)  Homo sapiens (human) / References: UniProt: P68133 Homo sapiens (human) / References: UniProt: P68133#3: Chemical | ChemComp-EDO / #4: Water | ChemComp-HOH / | Has ligand of interest | Y | Has protein modification | Y | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.33 Å3/Da / Density % sol: 47.27 % |
|---|---|
| Crystal grow | Temperature: 292 K / Method: vapor diffusion, sitting drop / pH: 5 / Details: 25% w/v PEG1500, 100 mM MIB, pH 5.0 |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  APS APS  / Beamline: 22-ID / Wavelength: 1 Å / Beamline: 22-ID / Wavelength: 1 Å |
| Detector | Type: DECTRIS EIGER X 16M / Detector: PIXEL / Date: Sep 28, 2020 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 1 Å / Relative weight: 1 |
| Reflection | Resolution: 1.45→34.92 Å / Num. obs: 79846 / % possible obs: 89.7 % / Redundancy: 4.5 % / Biso Wilson estimate: 15.07 Å2 / CC1/2: 0.991 / Net I/σ(I): 11.8 |
| Reflection shell | Resolution: 1.45→1.5 Å / Num. unique obs: 4489 / CC1/2: 0.69 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB entry 2IUI Resolution: 1.45→34.92 Å / SU ML: 0.1734 / Cross valid method: FREE R-VALUE / σ(F): 1.34 / Phase error: 25.4433 Stereochemistry target values: GeoStd + Monomer Library + CDL v1.2
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 23.19 Å2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.45→34.92 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller











 PDBj
PDBj