[English] 日本語
 Yorodumi
Yorodumi- PDB-7est: Interaction of the peptide CF3-LEU-ALA-NH-C6H4-CF3(TFLA) with por... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7est | ||||||
|---|---|---|---|---|---|---|---|
| Title | Interaction of the peptide CF3-LEU-ALA-NH-C6H4-CF3(TFLA) with porcine pancreatic elastase. X-ray studies at 1.8 Angstroms | ||||||
 Components Components | ELASTASE | ||||||
 Keywords Keywords | HYDROLASE/HYDROLASE INHIBITOR / SERINE PROTEINASE / HYDROLASE-HYDROLASE INHIBITOR complex | ||||||
| Function / homology |  Function and homology information Function and homology informationpancreatic elastase / serine-type endopeptidase activity / proteolysis / : / metal ion binding Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / Resolution: 1.8 Å X-RAY DIFFRACTION / Resolution: 1.8 Å | ||||||
 Authors Authors | Li De Lasierra, I. / Prange, T. | ||||||
 Citation Citation |  Journal: J.Mol.Recog. / Year: 1990 Journal: J.Mol.Recog. / Year: 1990Title: Interaction of the peptide CF3-Leu-Ala-NH-C6H4-CF3 (TFLA) with porcine pancreatic elastase. X-ray studies at 1.8 A. Authors: de la Sierra, I.L. / Papamichael, E. / Sakarellos, C. / Dimicoli, J.L. / Prange, T. #1:  Journal: J.Mol.Biol. / Year: 1978 Journal: J.Mol.Biol. / Year: 1978Title: The Atomic Structure of Crystalline Porcine Pancreatic Elastase at 2.5 Angstroms Resolution. Comparisons with the Structure of Alpha-Chymotrypsin Authors: Sawyer, L. / Shotton, C.M. / Campbell, J.W. / Wendell, P.L. / Muirhead, H. / Watson, H.C. / Diamond, R. / Ladner, R.C. #2:  Journal: Eur.J.Biochem. / Year: 1980 Journal: Eur.J.Biochem. / Year: 1980Title: The Indirect Mechanism of Action of the Trifluoroacetyl Peptides on Elastase Authors: Dimicoli, J.-L. / Renaud, A. / Bieth, J. #3:  Journal: J.Mol.Biol. / Year: 1982 Journal: J.Mol.Biol. / Year: 1982Title: Crystallographic Study of the Binding of a Tri-Fluoroacetyl Dipeptide Anilide Inhibitor with Elastase Authors: Hughes, D.L. / Diecker, L.C. / Bieth, L.C. / Dimicoli, J.-L. #4:  Journal: Acta Crystallogr B / Year: 1988 Journal: Acta Crystallogr B / Year: 1988Title: Structure of native porcine pancreatic elastase at 1.65 A resolutions. Authors: Meyer, E. / Cole, G. / Radhakrishnan, R. / Epp, O. #5:  Journal: J.Mol.Biol. / Year: 1986 Journal: J.Mol.Biol. / Year: 1986Title: Structure of the Product Complex of Acetyl-Ala-Pro-Ala with Porcine Pancreatic Elastase at 1.65 A Resolution Authors: Meyer, E. / Radhakrishnan, R. / Cole, G. / Presta, L.G. | ||||||
| History |
| ||||||
| Remark 700 | SHEET THE TWO SEVEN STRANDED SHEETS IN THIS STRUCTURE ARE REALLY SIX STRANDED BETA BARRELS. THIS IS ...SHEET THE TWO SEVEN STRANDED SHEETS IN THIS STRUCTURE ARE REALLY SIX STRANDED BETA BARRELS. THIS IS DENOTED BY THE FIRST STRAND RECURRING AS THE LAST STRAND. |
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7est.cif.gz 7est.cif.gz | 65.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7est.ent.gz pdb7est.ent.gz | 46.8 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7est.json.gz 7est.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/es/7est https://data.pdbj.org/pub/pdb/validation_reports/es/7est ftp://data.pdbj.org/pub/pdb/validation_reports/es/7est ftp://data.pdbj.org/pub/pdb/validation_reports/es/7est | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  6estC  2estS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 1 molecules E
| #1: Protein | Mass: 25928.031 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  |
|---|
-Non-polymers , 5 types, 111 molecules 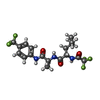








| #2: Chemical | ChemComp-0Z2 / | ||
|---|---|---|---|
| #3: Chemical | ChemComp-SO4 / | ||
| #4: Chemical | ChemComp-CA / | ||
| #5: Chemical | ChemComp-DMF / #6: Water | ChemComp-HOH / | |
-Details
| Has protein modification | Y |
|---|---|
| Sequence details | THE RESIDUE NUMBERING SCHEME USED FOR ELASTASE WAS CHOSEN TO MAXIMIZE HOMOLOGY WITH THE NUMBERING ...THE RESIDUE NUMBERING SCHEME USED FOR ELASTASE WAS CHOSEN TO MAXIMIZE HOMOLOGY WITH THE NUMBERING FOR CHYMOTRYPS |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION X-RAY DIFFRACTION |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.15 Å3/Da / Density % sol: 42.88 % | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | pH: 5.7 / Details: 10% DMF SOLUTION BUFFERED AT PH 5.7 WITH ACETATE | ||||||||||||||||||||||||||||||
| Crystal grow | *PLUS pH: 5.7 / Method: vapor diffusion, sitting drop | ||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Radiation | Scattering type: x-ray |
|---|---|
| Radiation wavelength | Relative weight: 1 |
| Reflection | *PLUS Highest resolution: 2 Å / Num. all: 19661 / Num. obs: 11021 / Num. measured all: 10569 / Rmerge(I) obs: 0.039 |
- Processing
Processing
| Software | Name:  X-PLOR / Classification: refinement X-PLOR / Classification: refinement | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Starting model: ELASTASE COMPLEXED WITH A TRIFLUOROACETYL DIPEPTIDE INHIBITOR, (PDB ENTRY 2EST) Rfactor Rwork: 0.19 / Highest resolution: 1.8 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Highest resolution: 1.8 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement | *PLUS Highest resolution: 2.2 Å / Rfactor all: 0.169 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints | *PLUS Type: x_angle_d / Dev ideal: 2 |
 Movie
Movie Controller
Controller













 PDBj
PDBj




