[English] 日本語
 Yorodumi
Yorodumi- PDB-5ni9: Crystal structure of HLA-DRB1*04:01 with the alpha-enolase peptid... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5ni9 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal structure of HLA-DRB1*04:01 with the alpha-enolase peptide 326-340 | ||||||
 Components Components |
| ||||||
 Keywords Keywords | IMMUNE SYSTEM / HLA-DR / enolase / arthritis / antigen presentation | ||||||
| Function / homology |  Function and homology information Function and homology informationManipulation of host energy metabolism / positive regulation of plasminogen activation / regulation of interleukin-4 production / regulation of interleukin-10 production / phosphopyruvate hydratase / phosphopyruvate hydratase complex / phosphopyruvate hydratase activity / negative regulation of hypoxia-induced intrinsic apoptotic signaling pathway / autolysosome membrane / myeloid dendritic cell antigen processing and presentation ...Manipulation of host energy metabolism / positive regulation of plasminogen activation / regulation of interleukin-4 production / regulation of interleukin-10 production / phosphopyruvate hydratase / phosphopyruvate hydratase complex / phosphopyruvate hydratase activity / negative regulation of hypoxia-induced intrinsic apoptotic signaling pathway / autolysosome membrane / myeloid dendritic cell antigen processing and presentation / antigen processing and presentation of endogenous peptide antigen via MHC class II / regulation of T-helper cell differentiation / positive regulation of CD4-positive, CD25-positive, alpha-beta regulatory T cell differentiation / positive regulation of CD4-positive, alpha-beta T cell activation / positive regulation of muscle contraction / antigen processing and presentation of peptide or polysaccharide antigen via MHC class II / positive regulation of T cell mediated immune response to tumor cell / positive regulation of memory T cell differentiation / positive regulation of monocyte differentiation / inflammatory response to antigenic stimulus / CD4 receptor binding / Gluconeogenesis / Glycolysis / M band / T-helper 1 type immune response / intermediate filament / canonical glycolysis / transport vesicle membrane / Translocation of ZAP-70 to Immunological synapse / Phosphorylation of CD3 and TCR zeta chains / polysaccharide binding / negative regulation of type II interferon production / humoral immune response / positive regulation of ATP biosynthetic process / nuclear outer membrane / Generation of second messenger molecules / macrophage differentiation / immunological synapse / Co-inhibition by PD-1 / epidermis development / detection of bacterium / negative regulation of T cell proliferation / T cell receptor binding / MHC class II antigen presentation / positive regulation of insulin secretion involved in cellular response to glucose stimulus / transcription corepressor binding / trans-Golgi network membrane / glycolytic process / gluconeogenesis / RNA polymerase II transcription regulatory region sequence-specific DNA binding / lumenal side of endoplasmic reticulum membrane / protein tetramerization / ER to Golgi transport vesicle membrane / negative regulation of inflammatory response to antigenic stimulus / clathrin-coated endocytic vesicle membrane / peptide antigen assembly with MHC class II protein complex / negative regulation of cell growth / MHC class II protein complex / positive regulation of T cell mediated cytotoxicity / structural constituent of cytoskeleton / DNA-binding transcription repressor activity, RNA polymerase II-specific / antigen processing and presentation of exogenous peptide antigen via MHC class II / positive regulation of immune response / peptide antigen binding / cognition / positive regulation of T cell activation / Interferon gamma signaling / response to virus / endocytic vesicle membrane / MHC class II protein complex binding / transcription corepressor activity / Downstream TCR signaling / late endosome membrane / T cell receptor signaling pathway / GTPase binding / early endosome membrane / cell cortex / adaptive immune response / lysosome / immune response / cadherin binding / Golgi membrane / external side of plasma membrane / lysosomal membrane / negative regulation of DNA-templated transcription / magnesium ion binding / cell surface / negative regulation of transcription by RNA polymerase II / signal transduction / protein homodimerization activity / : / RNA binding / extracellular exosome / membrane / nucleus / plasma membrane / cytoplasm / cytosol Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.33 Å MOLECULAR REPLACEMENT / Resolution: 1.33 Å | ||||||
 Authors Authors | Gerstner, C. / Dubnovitsky, A. | ||||||
 Citation Citation |  Journal: J. Autoimmun. / Year: 2018 Journal: J. Autoimmun. / Year: 2018Title: Memory T cells specific to citrullinated alpha-enolase are enriched in the rheumatic joint Authors: Pieper, J. / Dubnovitsky, A. / Gerstner, C. / James, E.A. / Rieck, M. / Kozhukh, G. / Tandre, K. / Pellegrino, S. / Gebe, J.A. / Ronnblom, L. / Sandalova, T. / Kwok, W.W. / Klareskog, L. / ...Authors: Pieper, J. / Dubnovitsky, A. / Gerstner, C. / James, E.A. / Rieck, M. / Kozhukh, G. / Tandre, K. / Pellegrino, S. / Gebe, J.A. / Ronnblom, L. / Sandalova, T. / Kwok, W.W. / Klareskog, L. / Buckner, J.H. / Achour, A. / Malmstrom, V. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5ni9.cif.gz 5ni9.cif.gz | 190.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5ni9.ent.gz pdb5ni9.ent.gz | 151.5 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5ni9.json.gz 5ni9.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ni/5ni9 https://data.pdbj.org/pub/pdb/validation_reports/ni/5ni9 ftp://data.pdbj.org/pub/pdb/validation_reports/ni/5ni9 ftp://data.pdbj.org/pub/pdb/validation_reports/ni/5ni9 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  5nigC  4mcyS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
-HLA class II histocompatibility antigen, ... , 2 types, 2 molecules AB
| #1: Protein | Mass: 21911.750 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: HLA-DRA, HLA-DRA1 / Production host: Homo sapiens (human) / Gene: HLA-DRA, HLA-DRA1 / Production host:  |
|---|---|
| #2: Protein | Mass: 23102.672 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: HLA-DRB1 / Production host: Homo sapiens (human) / Gene: HLA-DRB1 / Production host:  |
-Protein/peptide , 1 types, 1 molecules C
| #3: Protein/peptide | Mass: 1681.033 Da / Num. of mol.: 1 / Fragment: UNP residues 326-340 / Source method: obtained synthetically / Details: synthetic peptide from human alpha-enolase / Source: (synth.)  Homo sapiens (human) / References: UniProt: P06733, phosphopyruvate hydratase Homo sapiens (human) / References: UniProt: P06733, phosphopyruvate hydratase |
|---|
-Non-polymers , 4 types, 314 molecules 
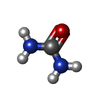





| #4: Chemical | ChemComp-MRD / ( | ||||
|---|---|---|---|---|---|
| #5: Chemical | | #6: Chemical | #7: Water | ChemComp-HOH / | |
-Details
| Has protein modification | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.52 Å3/Da / Density % sol: 51.11 % / Description: big rod-like crystals |
|---|---|
| Crystal grow | Temperature: 293 K / Method: vapor diffusion, hanging drop / pH: 6.5 Details: 0.1 M pH 6.5 10% (vol/vol) MPD 15% (vol/vol) PEG3350 |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  ESRF ESRF  / Beamline: MASSIF-1 / Wavelength: 0.966 Å / Beamline: MASSIF-1 / Wavelength: 0.966 Å |
| Detector | Type: DECTRIS PILATUS3 2M / Detector: PIXEL / Date: May 13, 2016 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.966 Å / Relative weight: 1 |
| Reflection | Resolution: 1.33→49.43 Å / Num. obs: 108263 / % possible obs: 99.3 % / Redundancy: 4.17 % / Biso Wilson estimate: 26 Å2 / CC1/2: 1 / Net I/σ(I): 13.7 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 4MCY Resolution: 1.33→49.43 Å / Cor.coef. Fo:Fc: 0.977 / Cor.coef. Fo:Fc free: 0.971 / SU B: 1.965 / SU ML: 0.034 / Cross valid method: THROUGHOUT / ESU R: 0.043 / ESU R Free: 0.043 / Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 24.919 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: 1 / Resolution: 1.33→49.43 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller






















 PDBj
PDBj















