[English] 日本語
 Yorodumi
Yorodumi- PDB-5fs5: Breaking down the wall: mutation of the tyrosine gate of the univ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5fs5 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
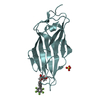
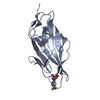
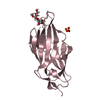
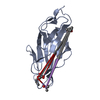
| Title | Breaking down the wall: mutation of the tyrosine gate of the universal Escherichia coli fimbrial adhesin FimH | |||||||||
 Components Components | FIMH | |||||||||
 Keywords Keywords | CELL ADHESION / BACTERIAL ADHESIN / TYPE 1 FIMBRIAE / URINARY TRACT INFECTION / VARIABLE IMMUNOGLOBULIN FOLD | |||||||||
| Function / homology |  Function and homology information Function and homology informationpilus tip / mechanosensory behavior / Attachment of bacteria to epithelial cells / cell adhesion involved in single-species biofilm formation / pilus / cell-substrate adhesion / D-mannose binding / host cell membrane / cell adhesion Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.42 Å MOLECULAR REPLACEMENT / Resolution: 1.42 Å | |||||||||
 Authors Authors | Bouckaert, J. | |||||||||
 Citation Citation |  Journal: IUCrJ / Year: 2017 Journal: IUCrJ / Year: 2017Title: Mutation of Tyr137 of the universal Escherichia coli fimbrial adhesin FimH relaxes the tyrosine gate prior to mannose binding. Authors: Rabbani, S. / Krammer, E.M. / Roos, G. / Zalewski, A. / Preston, R. / Eid, S. / Zihlmann, P. / Prevost, M. / Lensink, M.F. / Thompson, A. / Ernst, B. / Bouckaert, J. | |||||||||
| History |
|
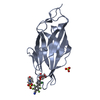
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5fs5.cif.gz 5fs5.cif.gz | 118.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5fs5.ent.gz pdb5fs5.ent.gz | 94 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5fs5.json.gz 5fs5.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/fs/5fs5 https://data.pdbj.org/pub/pdb/validation_reports/fs/5fs5 ftp://data.pdbj.org/pub/pdb/validation_reports/fs/5fs5 ftp://data.pdbj.org/pub/pdb/validation_reports/fs/5fs5 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  4ca4C  5fwrC  5fx3C  4auuS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||
| Unit cell |
| |||||||||
| Components on special symmetry positions |
|
- Components
Components
| #1: Protein | Mass: 16824.732 Da / Num. of mol.: 1 / Fragment: LECTIN DOMAIN, UNP RESIDUES 10-167 / Mutation: YES Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
|---|---|
| #2: Sugar | ChemComp-KGM / |
| #3: Chemical | ChemComp-NA / |
| #4: Water | ChemComp-HOH / |
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 1.93 Å3/Da / Density % sol: 36.19 % / Description: NONE |
|---|---|
| Crystal grow | pH: 6.5 Details: 5% PGA-LM; 100 MM NA CACODYLATE PH 6.5; 12 % W/V PEG 8K; 10 MM N-HEPTYL ALPHA-D-MANNOPYRANOSIDE |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  BESSY BESSY  / Beamline: 14.2 / Wavelength: 0.91841 / Beamline: 14.2 / Wavelength: 0.91841 |
| Detector | Type: RAYONIX / Detector: CCD / Date: Apr 19, 2012 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.91841 Å / Relative weight: 1 |
| Reflection | Resolution: 1.42→16.24 Å / Num. obs: 25691 / % possible obs: 98.4 % / Observed criterion σ(I): 1.34 / Redundancy: 6.64 % / Biso Wilson estimate: 8.64 Å2 / Rmerge(I) obs: 0.09 / Net I/σ(I): 17.03 |
| Reflection shell | Resolution: 1.42→1.48 Å / Redundancy: 91.8 % / Rmerge(I) obs: 0.47 / Mean I/σ(I) obs: 4.15 / % possible all: 89 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 4AUU Resolution: 1.42→16.246 Å / SU ML: 0.12 / σ(F): 1.34 / Phase error: 14.21 / Stereochemistry target values: ML
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å / Solvent model: FLAT BULK SOLVENT MODEL | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.42→16.246 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
|
 Movie
Movie Controller
Controller
















 PDBj
PDBj