[English] 日本語
 Yorodumi
Yorodumi- PDB-5df6: Crystal structure of PTPN11 tandem SH2 domains in complex with a ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 5df6 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Crystal structure of PTPN11 tandem SH2 domains in complex with a TXNIP peptide | |||||||||
 Components Components |
| |||||||||
 Keywords Keywords | HYDROLASE / Structural Genomics / Structural Genomics Consortium / SGC | |||||||||
| Function / homology |  Function and homology information Function and homology informationcellular response to tumor cell / negative regulation of cortisol secretion / intestinal epithelial cell migration / microvillus organization / negative regulation of growth hormone secretion / cellular response to oxidised low-density lipoprotein particle stimulus / genitalia development / atrioventricular canal development / negative regulation of cell division / negative regulation of cell adhesion mediated by integrin ...cellular response to tumor cell / negative regulation of cortisol secretion / intestinal epithelial cell migration / microvillus organization / negative regulation of growth hormone secretion / cellular response to oxidised low-density lipoprotein particle stimulus / genitalia development / atrioventricular canal development / negative regulation of cell division / negative regulation of cell adhesion mediated by integrin / STAT5 Activation / Co-inhibition by BTLA / Netrin mediated repulsion signals / cerebellar cortex formation / negative regulation of neutrophil activation / positive regulation of hormone secretion / regulation of protein export from nucleus / positive regulation of ossification / positive regulation of lipopolysaccharide-mediated signaling pathway / Interleukin-37 signaling / Signaling by Leptin / hormone metabolic process / MET activates PTPN11 / Regulation of RUNX1 Expression and Activity / negative regulation of chondrocyte differentiation / Regulation of FOXO transcriptional activity by acetylation / Signal regulatory protein family interactions / face morphogenesis / ERBB signaling pathway / platelet formation / triglyceride metabolic process / megakaryocyte development / organ growth / negative regulation of type I interferon production / Interleukin-20 family signaling / PI-3K cascade:FGFR3 / Interleukin-6 signaling / Co-inhibition by CTLA4 / Platelet sensitization by LDL / STAT5 activation downstream of FLT3 ITD mutants / peptide hormone receptor binding / PI-3K cascade:FGFR2 / PI-3K cascade:FGFR4 / MAPK3 (ERK1) activation / PI-3K cascade:FGFR1 / Prolactin receptor signaling / neurotrophin TRK receptor signaling pathway / regulation of cell adhesion mediated by integrin / regulation of type I interferon-mediated signaling pathway / platelet-derived growth factor receptor signaling pathway / MAPK1 (ERK2) activation / PECAM1 interactions / inner ear development / Bergmann glial cell differentiation / peptidyl-tyrosine dephosphorylation / positive regulation of intracellular signal transduction / non-membrane spanning protein tyrosine phosphatase activity / phosphoprotein phosphatase activity / RET signaling / Regulation of IFNA/IFNB signaling / The NLRP3 inflammasome / Interleukin-3, Interleukin-5 and GM-CSF signaling / PI3K Cascade / enzyme inhibitor activity / Co-inhibition by PD-1 / fibroblast growth factor receptor signaling pathway / GAB1 signalosome / positive regulation of insulin receptor signaling pathway / response to mechanical stimulus / ephrin receptor signaling pathway / regulation of protein-containing complex assembly / Activated NTRK2 signals through FRS2 and FRS3 / Regulation of IFNG signaling / response to glucose / Purinergic signaling in leishmaniasis infection / keratinocyte differentiation / cell adhesion molecule binding / FRS-mediated FGFR3 signaling / Signaling by CSF3 (G-CSF) / GPVI-mediated activation cascade / Signaling by FLT3 ITD and TKD mutants / negative regulation of T cell proliferation / T cell costimulation / FRS-mediated FGFR2 signaling / hormone-mediated signaling pathway / FRS-mediated FGFR4 signaling / Activation of IRF3, IRF7 mediated by TBK1, IKKε (IKBKE) / FRS-mediated FGFR1 signaling / Tie2 Signaling / phosphotyrosine residue binding / FLT3 Signaling / protein-tyrosine-phosphatase / homeostasis of number of cells within a tissue / Downstream signal transduction / axonogenesis / positive regulation of mitotic cell cycle / positive regulation of interferon-beta production / protein tyrosine phosphatase activity / protein tyrosine kinase binding / Negative regulation of FGFR3 signaling Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / Resolution: 1.78 Å SYNCHROTRON / Resolution: 1.78 Å | |||||||||
 Authors Authors | Dong, A. / Li, W. / Tempel, W. / Liu, Y. / Bountra, C. / Arrowsmith, C.H. / Edwards, A.M. / Min, J. / Structural Genomics Consortium (SGC) | |||||||||
 Citation Citation |  Journal: Biochem.J. / Year: 2016 Journal: Biochem.J. / Year: 2016Title: Structural basis for the regulatory role of the PPxY motifs in the thioredoxin-interacting protein TXNIP. Authors: Liu, Y. / Lau, J. / Li, W. / Tempel, W. / Li, L. / Dong, A. / Narula, A. / Qin, S. / Min, J. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  5df6.cif.gz 5df6.cif.gz | 63.4 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb5df6.ent.gz pdb5df6.ent.gz | 44.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  5df6.json.gz 5df6.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  5df6_validation.pdf.gz 5df6_validation.pdf.gz | 445.1 KB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  5df6_full_validation.pdf.gz 5df6_full_validation.pdf.gz | 445.2 KB | Display | |
| Data in XML |  5df6_validation.xml.gz 5df6_validation.xml.gz | 11.9 KB | Display | |
| Data in CIF |  5df6_validation.cif.gz 5df6_validation.cif.gz | 16.5 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/df/5df6 https://data.pdbj.org/pub/pdb/validation_reports/df/5df6 ftp://data.pdbj.org/pub/pdb/validation_reports/df/5df6 ftp://data.pdbj.org/pub/pdb/validation_reports/df/5df6 | HTTPS FTP |
-Related structure data
| Related structure data |  5cq2C 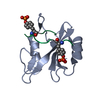 3tkzS  4xz0S S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 28750.146 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: PTPN11, PTP2C, SHPTP2 / Plasmid: pET28-SacB-AP / Production host: Homo sapiens (human) / Gene: PTPN11, PTP2C, SHPTP2 / Plasmid: pET28-SacB-AP / Production host:  | ||||||
|---|---|---|---|---|---|---|---|
| #2: Protein/peptide | Mass: 1503.610 Da / Num. of mol.: 2 / Source method: obtained synthetically / Source: (synth.)  Homo sapiens (human) / References: UniProt: Q9H3M7*PLUS Homo sapiens (human) / References: UniProt: Q9H3M7*PLUS#3: Chemical | ChemComp-UNX / #4: Water | ChemComp-HOH / | Has protein modification | Y | |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 1.98 Å3/Da / Density % sol: 37.9 % |
|---|---|
| Crystal grow | Temperature: 291 K / Method: vapor diffusion / Details: 20% PEG-3350, 0.2 M ammonium formate |
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  APS APS  / Beamline: 19-ID / Wavelength: 0.97929 Å / Beamline: 19-ID / Wavelength: 0.97929 Å |
| Detector | Type: ADSC QUANTUM 315r / Detector: CCD / Date: Jun 19, 2015 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength: 0.97929 Å / Relative weight: 1 |
| Reflection | Resolution: 1.78→46.69 Å / Num. obs: 23333 / % possible obs: 98.5 % / Redundancy: 3.9 % / Rmerge(I) obs: 0.076 / Net I/σ(I): 11.2 |
| Reflection shell | Resolution: 1.78→1.82 Å / Redundancy: 3.3 % / Rmerge(I) obs: 0.526 / Mean I/σ(I) obs: 1.4 / % possible all: 95.9 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Starting model: 3TKZ AND 4XZ0 Resolution: 1.78→46.69 Å / SU B: 3.725 / SU ML: 0.112 / Cross valid method: THROUGHOUT / σ(F): 0 / ESU R: 0.129 / ESU R Free: 0.133 Details: ARP/WARP WAS USED FOR AUTOMATED MODEL BUILDING. COOT WAS USED FOR INTERACTIVE MODEL BUILDING. PHENIX.MOLPROBITY WAS USED FOR GEOMETRY VALIDATION. JLIGAND AND THE GRADE SERVER WERE USED IN RESTRAINT PREPARATION.
| ||||||||||||||||||||||||
| Displacement parameters | Biso mean: 30.51 Å2
| ||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.78→46.69 Å
|
 Movie
Movie Controller
Controller












 PDBj
PDBj






































